Volume 8, Issue 4 (July & August 2017 -- 2017)
BCN 2017, 8(4): 279-284 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Heidari M M, Khatami M, Tahamtan Y. Molecular Analysis of rs2070744 and rs1799983 Polymorphisms of NOS3 Gene in Iranian Patients With Multiple Sclerosis. BCN 2017; 8 (4) :279-284
URL: http://bcn.iums.ac.ir/article-1-665-en.html
URL: http://bcn.iums.ac.ir/article-1-665-en.html
1- Department of Biology, Faculty of Science, Yazd University, Yazd, Iran.
2- Department of Biology, Ashkezar Branch, Islamic Azad University, Yazd, Iran.
2- Department of Biology, Ashkezar Branch, Islamic Azad University, Yazd, Iran.
Full-Text [PDF 561 kb]
| Abstract (HTML)
Full-Text:
1. Introduction
Multiple Sclerosis (MS) is a neuroimmunological disease, causing severe neurological disabilities as a result of demyelination (Goris, Pauwels, & Dubois, 2012). It is mainly defined as a heterogeneous, complex, and multifactorial disease. The disease becomes progressive after several episodes in the majority of affected individuals (Hoffjan & Akkad, 2010; McElroy & Oksenberg, 2011). Although twin and sibling pair studies point to a genetic component, the etiology of MS is mostly unknown. However, there is a seminal effort to detect the responsible genes in increased susceptibility and affecting clinical consequence (Hoffjan & Akkad, 2010).
An important role for Nitric Oxide (NO) in the pathogenesis of MS and its influence on the various aspects of the disorder, including changes in synaptic transmission, inflammation, and neuronal death were pointed by abundant evidence (Gerzanich, Ivanova, Zhou, & Simard, 2003; Jagnandan, Sessa, & Fulton, 2005). Several studies are shown that nitrate, NO degradation product, is found with increased concentration in cerebrospinal fluid of patients with MS (Giovannoni, 1998; Tajouri et al., 2004).
Oxidation of L-arginine to Nitric Oxide (NO) is catalyzed by Nitric Oxide Synthases (NOSs). An isoform of NO producing enzymes is endothelial Nitric Oxide Synthase (NOS3) that is constitutively expressed in endothelial cells. This enzyme has been found to play a prominent role in both vasculogenesis and angiogenesis. The nitric oxide synthases is encoded by NOS3 gene located on chromosome 7q35-q36 (Forstermann & Sessa, 2011). There are two common polymorphisms of NOS3 gene in many populations that are associated with NOS3 enzyme activity and production. In the seventh exon of the NOS3 gene, the c.894G>T SNP substitutes glutamic acid at codon 298 to aspartic acid (Glu298Asp).
This polymorphism causes diminished NOS3 enzyme activity and the c.-813C>T (rs2070744) polymorphism in the promoter region, where it has been found with reduced NOS3 promoter activity in vitro (Miyamoto et al., 1998; Veldman et al., 2002). The genetic association of these NOS3 gene polymorphisms and susceptibility to MS have identified in several studies (AlFadhli, Mohammed & Al Shubaili, 2013; Blanco, Yague, Graus, & Saiz, 2003; Modin et al., 2001; Tajouri et al., 2004). The purpose of the present research was to determine a relation between c.-813C>T (rs2070744) and c.894G>T (rs1799983) polymorphisms of NOS3 gene with susceptibility to MS in Iranian population.
2. Methods
2.1. Study subjects
After a neurological examination, 78 patients with MS (60 females and 18 males) and the mean age of 33.74 years (age range from 24-57 years) were recruited from MS Clinic (Yazd, Iran). Healthy volunteers (80 subjects) were randomly selected from their families with no history of autoimmune and neurological diseases. In the present study, the experimental and control groups were ethnically matched. All individuals gave informed consent for DNA test. DNA extraction was performed from whole blood with salting out method according to the manufacturer’s instructions. Patients and controls’ blood and DNA samples were isolated and stored at -20°C.
2.2. Genotyping of NOS3 (rs2070744) by Multiplex-ARMS PCR
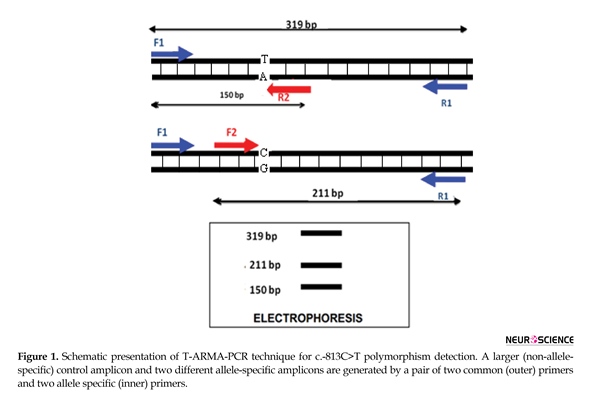
Genotyping of c.-813C>T NOS3 SNP (rs2070744) was detected by tetra-primer ARMS-PCR (T-ARMS-PCR). PCR amplification of c.-813C>T polymorphism in the 5′ flanking region of the NOS3 gene was performed using four primers. The online website Primer1 was used for design of our primers (Table 1). The primers specificity were tested with ‘BLAST’ program in NCBI server. To improve the specificity of T-ARMS-PCR, the 3rd nucleotide from the 3´-terminus of inner primers was changed to destabilize mismatch.
PCR was achieved in 25 µL volume, containing 50 ng of DNA, 1X PCR Master Mix, and 250 nM of each primer (Yekta Tajhiz Azma, Tehran, Iran). Touchdown PCR was performed at 95ºC for 2 min, denaturation at 95ºC for 20 s, the first 10 cycles annealing temperature at 68ºC to 60ºC and remaining 25 cycles at annealing at 59ºC for 60 s and extension at 72ºC for 50 s. The PCR products were run on 2% agarose gel electrophoresis. In this method, the gel electrophoresis revealed 3 bands in AG heterozygotes (319, 150, and 211bp), 2 bands in AA homozygotes (319 and 150 bp), and 2 bands in GG homozygotes (319 and 211 bp) (Figures 1 and 2).
2.3. Genotyping NOS3 (rs1799983) by PCR-RFLP
The NOS3 polymorphism c.894G>T introduces the restriction site for MboI endonuclease. PCR was achieved in 25 µL volume; containing 50 ng of DNA, 1X PCR Master Mix, and 250 nM of each primer (Yekta Tajhiz Azma, Tehran, Iran). The PCR reaction was according to the following stages: 35 cycles; including denaturation, annealing, and extension at 95°C, 64°C, and 72°C for 35 s, respectively; and a final extension at 72°C for 5 min. According to Figure 2, a 292 bp fragment was detected in GG homozygote genotypes (MboI- undigested), also 197 and 95 bp fragments in TT homozygote genotypes (MboI- digested), finally 292, 197, and 95 bp fragments in GT heterozygote genotypes (MboI- digested and undigested fragments).
2.4. Statistical analysis
Statistical analysis was performed using SPSS (IBM SPSS 22, SPSS Inc., Chicago, IL., USA). Allelic and genotypic frequencies were calculated for each SNPs. Multiple logistic regression models (co-dominant, dominant, recessive, and over-dominant) were employed to analyze the genetic data. P value less than 0.05 was considered statistically significant.
3. Results
The genotype distributions and allele frequencies of two types of polymorphism in NOS3 gene, c.-813C>T, and c.894G>T were analyzed in Iranian patients with MS and healthy controls. Table 2 presents genotypic and allelic frequencies of c.-813C>T polymorphism among the patients and controls group. In the patients group, the frequencies of TT, CT and CC genotypes were 53.84%, 41.02%, and 5.1%, respectively. In the control group, the frequencies of TT, CT, and CC genotypes were 80.0%, 18.8% and 1.2%, respectively. Statistical analysis showed that c.-813C>T polymorphism is associated with risk of MS according to co-dominant, dominant, and over-dominant model (co-dominant model, P=0.001; dominant model, P=0.004; and over-dominant, P=0.002). The prevalence of C allele was significantly higher in patients compared to the control group (OR: 0.345, 95%CI: 0.18-0.64; P=0.001).
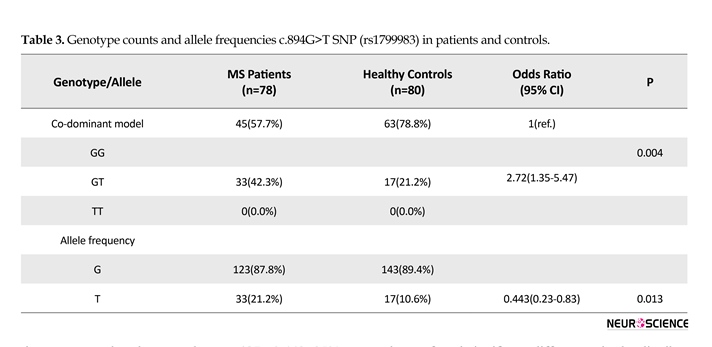
Genotypic and allelic frequencies of c.894G>T polymorphism among the patients and controls group were shown in Table 3. In the patients group, the frequencies of GG, GT, and TT genotypes were 57.7%, 42.3%, and 0.0%, respectively. In the control group, the frequencies of GG, GT, and TT genotypes were 78.7%, 21.2%, and 0.0%, respectively. Statistical analysis showed that according to co-dominant model, c.894G>T polymorphism is associated with risk of MS (P=0.004). The prevalence of T allele was significantly higher in patients compared to the control group (OR: 0.443, 95% CI: 0.23-0.83; P=0.013).
Multiple Sclerosis (MS) is a neuroimmunological disease, causing severe neurological disabilities as a result of demyelination (Goris, Pauwels, & Dubois, 2012). It is mainly defined as a heterogeneous, complex, and multifactorial disease. The disease becomes progressive after several episodes in the majority of affected individuals (Hoffjan & Akkad, 2010; McElroy & Oksenberg, 2011). Although twin and sibling pair studies point to a genetic component, the etiology of MS is mostly unknown. However, there is a seminal effort to detect the responsible genes in increased susceptibility and affecting clinical consequence (Hoffjan & Akkad, 2010).
An important role for Nitric Oxide (NO) in the pathogenesis of MS and its influence on the various aspects of the disorder, including changes in synaptic transmission, inflammation, and neuronal death were pointed by abundant evidence (Gerzanich, Ivanova, Zhou, & Simard, 2003; Jagnandan, Sessa, & Fulton, 2005). Several studies are shown that nitrate, NO degradation product, is found with increased concentration in cerebrospinal fluid of patients with MS (Giovannoni, 1998; Tajouri et al., 2004).
Oxidation of L-arginine to Nitric Oxide (NO) is catalyzed by Nitric Oxide Synthases (NOSs). An isoform of NO producing enzymes is endothelial Nitric Oxide Synthase (NOS3) that is constitutively expressed in endothelial cells. This enzyme has been found to play a prominent role in both vasculogenesis and angiogenesis. The nitric oxide synthases is encoded by NOS3 gene located on chromosome 7q35-q36 (Forstermann & Sessa, 2011). There are two common polymorphisms of NOS3 gene in many populations that are associated with NOS3 enzyme activity and production. In the seventh exon of the NOS3 gene, the c.894G>T SNP substitutes glutamic acid at codon 298 to aspartic acid (Glu298Asp).
This polymorphism causes diminished NOS3 enzyme activity and the c.-813C>T (rs2070744) polymorphism in the promoter region, where it has been found with reduced NOS3 promoter activity in vitro (Miyamoto et al., 1998; Veldman et al., 2002). The genetic association of these NOS3 gene polymorphisms and susceptibility to MS have identified in several studies (AlFadhli, Mohammed & Al Shubaili, 2013; Blanco, Yague, Graus, & Saiz, 2003; Modin et al., 2001; Tajouri et al., 2004). The purpose of the present research was to determine a relation between c.-813C>T (rs2070744) and c.894G>T (rs1799983) polymorphisms of NOS3 gene with susceptibility to MS in Iranian population.
2. Methods
2.1. Study subjects
After a neurological examination, 78 patients with MS (60 females and 18 males) and the mean age of 33.74 years (age range from 24-57 years) were recruited from MS Clinic (Yazd, Iran). Healthy volunteers (80 subjects) were randomly selected from their families with no history of autoimmune and neurological diseases. In the present study, the experimental and control groups were ethnically matched. All individuals gave informed consent for DNA test. DNA extraction was performed from whole blood with salting out method according to the manufacturer’s instructions. Patients and controls’ blood and DNA samples were isolated and stored at -20°C.
2.2. Genotyping of NOS3 (rs2070744) by Multiplex-ARMS PCR
Genotyping of c.-813C>T NOS3 SNP (rs2070744) was detected by tetra-primer ARMS-PCR (T-ARMS-PCR). PCR amplification of c.-813C>T polymorphism in the 5′ flanking region of the NOS3 gene was performed using four primers. The online website Primer1 was used for design of our primers (Table 1). The primers specificity were tested with ‘BLAST’ program in NCBI server. To improve the specificity of T-ARMS-PCR, the 3rd nucleotide from the 3´-terminus of inner primers was changed to destabilize mismatch.
PCR was achieved in 25 µL volume, containing 50 ng of DNA, 1X PCR Master Mix, and 250 nM of each primer (Yekta Tajhiz Azma, Tehran, Iran). Touchdown PCR was performed at 95ºC for 2 min, denaturation at 95ºC for 20 s, the first 10 cycles annealing temperature at 68ºC to 60ºC and remaining 25 cycles at annealing at 59ºC for 60 s and extension at 72ºC for 50 s. The PCR products were run on 2% agarose gel electrophoresis. In this method, the gel electrophoresis revealed 3 bands in AG heterozygotes (319, 150, and 211bp), 2 bands in AA homozygotes (319 and 150 bp), and 2 bands in GG homozygotes (319 and 211 bp) (Figures 1 and 2).
2.3. Genotyping NOS3 (rs1799983) by PCR-RFLP
The NOS3 polymorphism c.894G>T introduces the restriction site for MboI endonuclease. PCR was achieved in 25 µL volume; containing 50 ng of DNA, 1X PCR Master Mix, and 250 nM of each primer (Yekta Tajhiz Azma, Tehran, Iran). The PCR reaction was according to the following stages: 35 cycles; including denaturation, annealing, and extension at 95°C, 64°C, and 72°C for 35 s, respectively; and a final extension at 72°C for 5 min. According to Figure 2, a 292 bp fragment was detected in GG homozygote genotypes (MboI- undigested), also 197 and 95 bp fragments in TT homozygote genotypes (MboI- digested), finally 292, 197, and 95 bp fragments in GT heterozygote genotypes (MboI- digested and undigested fragments).
2.4. Statistical analysis
Statistical analysis was performed using SPSS (IBM SPSS 22, SPSS Inc., Chicago, IL., USA). Allelic and genotypic frequencies were calculated for each SNPs. Multiple logistic regression models (co-dominant, dominant, recessive, and over-dominant) were employed to analyze the genetic data. P value less than 0.05 was considered statistically significant.
3. Results
The genotype distributions and allele frequencies of two types of polymorphism in NOS3 gene, c.-813C>T, and c.894G>T were analyzed in Iranian patients with MS and healthy controls. Table 2 presents genotypic and allelic frequencies of c.-813C>T polymorphism among the patients and controls group. In the patients group, the frequencies of TT, CT and CC genotypes were 53.84%, 41.02%, and 5.1%, respectively. In the control group, the frequencies of TT, CT, and CC genotypes were 80.0%, 18.8% and 1.2%, respectively. Statistical analysis showed that c.-813C>T polymorphism is associated with risk of MS according to co-dominant, dominant, and over-dominant model (co-dominant model, P=0.001; dominant model, P=0.004; and over-dominant, P=0.002). The prevalence of C allele was significantly higher in patients compared to the control group (OR: 0.345, 95%CI: 0.18-0.64; P=0.001).
Genotypic and allelic frequencies of c.894G>T polymorphism among the patients and controls group were shown in Table 3. In the patients group, the frequencies of GG, GT, and TT genotypes were 57.7%, 42.3%, and 0.0%, respectively. In the control group, the frequencies of GG, GT, and TT genotypes were 78.7%, 21.2%, and 0.0%, respectively. Statistical analysis showed that according to co-dominant model, c.894G>T polymorphism is associated with risk of MS (P=0.004). The prevalence of T allele was significantly higher in patients compared to the control group (OR: 0.443, 95% CI: 0.23-0.83; P=0.013).
4. Discussion
The involvement of Nitric Oxide (NO) in MS had been suggested after the discovery of the critical role of NO in inflammation. However, due to the opposing roles of NO in cellular processes, the extent of NO contribution to MS is not yet fully understood. In this study, we found significant differences in the distribution of c.-813C>T and c.894G>T polymorphisms of the NOS3 gene in Iranian patients with MS compared with healthy population. Several reports have been showed that c.-813C>T (rs2070744) polymorphism in the 5′ flanking (promoter) region of the NOS3 gene is associated with coronary spasm and vascular disease (Casas et al., 2006; Kajiyama et al., 2000; Nakayama et al., 1999; Nakayama et al., 2000). Also, previous studies have demonstrated the association between this polymorphism and recurrent abortions and male infertility (Luo et al., 2013; Safarinejad, Shafiei, & Safarinejad, 2010).
The involvement of Nitric Oxide (NO) in MS had been suggested after the discovery of the critical role of NO in inflammation. However, due to the opposing roles of NO in cellular processes, the extent of NO contribution to MS is not yet fully understood. In this study, we found significant differences in the distribution of c.-813C>T and c.894G>T polymorphisms of the NOS3 gene in Iranian patients with MS compared with healthy population. Several reports have been showed that c.-813C>T (rs2070744) polymorphism in the 5′ flanking (promoter) region of the NOS3 gene is associated with coronary spasm and vascular disease (Casas et al., 2006; Kajiyama et al., 2000; Nakayama et al., 1999; Nakayama et al., 2000). Also, previous studies have demonstrated the association between this polymorphism and recurrent abortions and male infertility (Luo et al., 2013; Safarinejad, Shafiei, & Safarinejad, 2010).
This SNP significantly reduces NOS3 promoter transcription activity, inhibiting NO production which ends in endothelial dysfunction. The present study demonstrated that this polymorphism was associated with the MS in Iranian population and might play an important role in our patients. The frequency of C allele in our patients was significantly higher than control subjects (P=0.001). To our knowledge, the current research is the first to study the correlation of rs2070744 with MS. The physiological and pathological effects of c.-813C>T polymorphism, however, requires further research.
The present study is the first to reveal an association between the c.894G>T SNP (rs1799983) of the NOS3 (Glu298Asp variant) gene and susceptibility to MS (P=0.004). We aimed to elucidate the missense Glu298Asp polymorphism within exon 7 of the NOS3 gene in 78 Iranian MS patients and 80 healthy controls. Some authors have suggested that Glu298Asp polymorphism in NOS3 gene plays a role in various diseases such as idiopathic male infertility, myocardial infraction, and placental abruption (Gad et al., 2012; Luo, Wen, Zhou, Chen, & Zhang, 2014; Safarinejad et al., 2010; Yoshimura, Yoshimura, Tabata, Yasue, & Okamura, 2001). The c.894G>T SNP alters the stability, biological half-life, and activity of the enzyme and is associated with reduced NO production (Wang et al., 1997).
Several findings have revealed that this mutation was the result of a conformational change in the eNOS protein from helix form to tight turn (Joshi, Mineo, Shaul, & Bauer, 2007). Based on our expectation, the statistical difference in this SNP indicates the correlation of G allele of rs1799983 with MS patients compared with controls. Thus, G allele had the greatest effects in Iranian patients. Our results show that NOS3 polymorphisms may be associated with MS in Iranian patients and so these variants might influence the risk of MS, specifically in the Iranian population.
Acknowledgments
This research was financialy supported by Yazd University. The authors thank the patients for their cooperation as well as MS Clinic (Yazd, Iran) and Shahid Sadoughi University of Medical Sciences.
Conflict of Interest
The authors declared no conflicts of interest.
References
AlFadhli, S., Mohammed, E. M. A., & Al Shubaili, A. (2013). Association analysis of nitric oxide synthases: NOS1, NOS2A and NOS3 genes, with multiple sclerosis. Annals of Human Biology, 40(4), 368–375. doi: 10.3109/03014460.2013.786756
Blanco, Y., Yague, J., Graus, F., & Saiz, A. (2003). No association of inducible nitric oxide synthase gene (NOS2A) to multiple sclerosis. Journal of Neurology, 250(5), 598–600. doi: 10.1007/s00415-003-1047-7
Casas, J. P., Cavalleri, G. L., Bautista, L. E., Smeeth, L., Humphries, S. E., & Hingorani, A. D. (2006). Endothelial nitric oxide synthase gene polymorphisms and cardiovascular disease: A HuGE review. American Journal of Epidemiology, 164(10), 921-35. doi: 10.1093/aje/kwj302
Forstermann, U., & Sessa, W. C. (2011). Nitric oxide synthases: Regulation and function. European Heart Journal, 33(7), 829–37. doi: 10.1093/eurheartj/ehr304
The present study is the first to reveal an association between the c.894G>T SNP (rs1799983) of the NOS3 (Glu298Asp variant) gene and susceptibility to MS (P=0.004). We aimed to elucidate the missense Glu298Asp polymorphism within exon 7 of the NOS3 gene in 78 Iranian MS patients and 80 healthy controls. Some authors have suggested that Glu298Asp polymorphism in NOS3 gene plays a role in various diseases such as idiopathic male infertility, myocardial infraction, and placental abruption (Gad et al., 2012; Luo, Wen, Zhou, Chen, & Zhang, 2014; Safarinejad et al., 2010; Yoshimura, Yoshimura, Tabata, Yasue, & Okamura, 2001). The c.894G>T SNP alters the stability, biological half-life, and activity of the enzyme and is associated with reduced NO production (Wang et al., 1997).
Several findings have revealed that this mutation was the result of a conformational change in the eNOS protein from helix form to tight turn (Joshi, Mineo, Shaul, & Bauer, 2007). Based on our expectation, the statistical difference in this SNP indicates the correlation of G allele of rs1799983 with MS patients compared with controls. Thus, G allele had the greatest effects in Iranian patients. Our results show that NOS3 polymorphisms may be associated with MS in Iranian patients and so these variants might influence the risk of MS, specifically in the Iranian population.
Acknowledgments
This research was financialy supported by Yazd University. The authors thank the patients for their cooperation as well as MS Clinic (Yazd, Iran) and Shahid Sadoughi University of Medical Sciences.
Conflict of Interest
The authors declared no conflicts of interest.
References
AlFadhli, S., Mohammed, E. M. A., & Al Shubaili, A. (2013). Association analysis of nitric oxide synthases: NOS1, NOS2A and NOS3 genes, with multiple sclerosis. Annals of Human Biology, 40(4), 368–375. doi: 10.3109/03014460.2013.786756
Blanco, Y., Yague, J., Graus, F., & Saiz, A. (2003). No association of inducible nitric oxide synthase gene (NOS2A) to multiple sclerosis. Journal of Neurology, 250(5), 598–600. doi: 10.1007/s00415-003-1047-7
Casas, J. P., Cavalleri, G. L., Bautista, L. E., Smeeth, L., Humphries, S. E., & Hingorani, A. D. (2006). Endothelial nitric oxide synthase gene polymorphisms and cardiovascular disease: A HuGE review. American Journal of Epidemiology, 164(10), 921-35. doi: 10.1093/aje/kwj302
Forstermann, U., & Sessa, W. C. (2011). Nitric oxide synthases: Regulation and function. European Heart Journal, 33(7), 829–37. doi: 10.1093/eurheartj/ehr304
Gad, M. Z., Abdel Rahman, M. F., Hashad, I. M., Abdel-Maksoud, S. M., Farag, N. M., & Abou-Aisha, K. (2012). Endothelial nitric oxide synthase (G894T) gene polymorphism in a random sample of the Egyptian population: Comparison with myocardial infarction patients. Genetic Testing and Molecular Biomarkers, 16(7), 695–700. doi: 10.1089/gtmb.2011.0342
Gerzanich, V., Ivanova, S., Zhou, H., & Simard, J. M. (2003). Mislocalization of eNOS and upregulation of cerebral vascular Ca2+ channel activity in angiotensin-hypertension. Hypertension, 41(5), 1124-30. doi: 10.1161/01.HYP.0000066288.20169.21
Giovannoni, G. (1998). Cerebrospinal fluid and serum nitric oxide metabolites in patients with multiple sclerosis. Multiple Sclerosis Journal, 4(1), 27–30. doi: 10.1177/135245859800400107
Goris, A., Pauwels, I., & Dubois, B. (2012). Progress in multiple sclerosis genetics. Current Genomics, 13(8), 646-63. doi: 10.2174/138920212803759695
Hoffjan, S., & Akkad, D. A. (2010). The genetics of multiple sclerosis: An update 2010. Molecular and Cellular Probes, 24(5), 237–43. doi: 10.1016/j.mcp.2010.04.006
Jagnandan, D., Sessa, W. C., & Fulton, D. (2005). Intracellular location regulates calcium-calmodulin-dependent activation of organelle-restricted eNOS. AJP: Cell Physiology, 289(4), 1024–33. doi: 10.1152/ajpcell.00162.2005
Joshi, M. S., Mineo, C., Shaul, P. W., & Bauer, J. A. (2007). Biochemical consequences of the NOS3 Glu298Asp variation in human endothelium: Altered caveolar localization and impaired response to shear. The FASEB Journal, 21(11), 2655–63. doi: 10.1096/fj.06-7088com
Kajiyama, N., Saito, Y., Miyamoto, Y., Yoshimura, M., Nakayama, M., Harada, M., et al. (2000). Lack of association between T-786.RAR.C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene and essential hypertension. Hypertension Research, 23(6), 561-5. doi: 10.1291/hypres.23.561
Luo, J. Q., Wen, J. G., Zhou, H. H., Chen, X. P., & Zhang, W. (2014). Endothelial nitric oxide synthase gene G894T polymorphism and myocardial infarction: A meta-analysis of 34 studies involving 21,068 subjects. PLoS ONE, 9, 87196. doi: 10.1371/journal.pone.0087196
Luo, L., Li, D. H., Wei, S. G., Zhang, H. B., Li, S. B., & Zhao, J. (2013). Polymorphisms in the endothelial nitric oxide synthase gene associated with recurrent miscarriage. Genetics and Molecular Research, 12(3), 3879–86. doi: 10.4238/2013.september.23.6
McElroy, J. P., & Oksenberg, J. R. (2011). Multiple sclerosis genetics 2010. Neurologic Clinics, 29(2), 219-31. doi: 10.1016/j.ncl.2010.12.002
Miyamoto, Y., Saito, Y., Kajiyama, N., Yoshimura, M., Shimasaki, Y., Nakayama, M., et al. (1998). Endothelial nitric oxide synthase gene is positively associated with essential hypertension. Hypertension, 32(1), 3-8. doi: 10.1161/01.hyp.32.1.3
Modin, H., Dai, Y., Masterman, T., Svejgaard, A., Sørensen, P. S., Oturai, A., et al. (2001). No linkage or association of the nitric oxide synthase genes to multiple sclerosis. Journal of Neuroimmunology, 119(1), 95–100. doi: 10.1016/s0165-5728(01)00366-6
Nakayama, M., Yasue, H., Yoshimura, M., Shimasaki, Y., Kugiyama, K., Ogawa, H., et al. (1999). T−786→C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation, 99(22), 2864-70. doi: 10.1161/01.cir.99.22.2864
Nakayama, M., Yasue, H., Yoshimura, M., Shimasaki, Y., Ogawa, H., Kugiyama, K., et al. (2000). T−786→C mutation in the 5′-flanking region of the endothelial nitric oxide synthase gene is associated with myocardial infarction, especially without coronary organic stenosis. The American Journal of Cardiology, 86(6), 628-34. doi: 10.1016/s0002-9149(00)01041-9
Safarinejad, M. R., Shafiei, N., & Safarinejad, S. (2010). The role of endothelial nitric oxide synthase (eNOS) T-786C, G894T, and 4a/b gene polymorphisms in the risk of idiopathic male infertility. Molecular Reproduction and Development, 77(8), 720–7. doi: 10.1002/mrd.21210
Tajouri, L., Ferreira, L., Ovcaric, M., Curtain, R., Lea, R., Csurhes, P., et al (2004). Investigation of a neuronal nitric oxide synthase gene (NOS1) polymorphism in a multiple sclerosis population. Journal of the Neurological Sciences, 218(1-2), 25–8. doi: 10.1016/j.jns.2003.10.006
Veldman, B. A., Spiering, W., Doevendans, P. A., Vervoort, G., Kroon, A. A., de Leeuw, P. W., et al. (2002). The Glu298Asp polymorphism of the NOS 3 gene as a determinant of the baseline production of nitric oxide. Journal of Hypertension, 20(10), 2023–7. doi: 10.1097/00004872-200210000-00022
Wang, X. L., Mahaney, M. C., Sim, A. S., Wang, J., Wang, J., Blangero, J., et al. (1997). Genetic contribution of the endothelial constitutive nitric oxide synthase gene to plasma nitric oxide levels. Arteriosclerosis, Thrombosis, and Vascular Biology, 17(11), 3147-53. doi: 10.1161/01.atv.17.11.3147
Yoshimura, T., Yoshimura, M., Tabata, A., Yasue, H., & Okamura, H. (2001). The missense Glu298Asp variant of the endothelial nitric oxide synthase gene is strongly associated with placental abruption. Human Genetics, 108(3), 181–3. doi: 10.1007/s004390000454
Gerzanich, V., Ivanova, S., Zhou, H., & Simard, J. M. (2003). Mislocalization of eNOS and upregulation of cerebral vascular Ca2+ channel activity in angiotensin-hypertension. Hypertension, 41(5), 1124-30. doi: 10.1161/01.HYP.0000066288.20169.21
Giovannoni, G. (1998). Cerebrospinal fluid and serum nitric oxide metabolites in patients with multiple sclerosis. Multiple Sclerosis Journal, 4(1), 27–30. doi: 10.1177/135245859800400107
Goris, A., Pauwels, I., & Dubois, B. (2012). Progress in multiple sclerosis genetics. Current Genomics, 13(8), 646-63. doi: 10.2174/138920212803759695
Hoffjan, S., & Akkad, D. A. (2010). The genetics of multiple sclerosis: An update 2010. Molecular and Cellular Probes, 24(5), 237–43. doi: 10.1016/j.mcp.2010.04.006
Jagnandan, D., Sessa, W. C., & Fulton, D. (2005). Intracellular location regulates calcium-calmodulin-dependent activation of organelle-restricted eNOS. AJP: Cell Physiology, 289(4), 1024–33. doi: 10.1152/ajpcell.00162.2005
Joshi, M. S., Mineo, C., Shaul, P. W., & Bauer, J. A. (2007). Biochemical consequences of the NOS3 Glu298Asp variation in human endothelium: Altered caveolar localization and impaired response to shear. The FASEB Journal, 21(11), 2655–63. doi: 10.1096/fj.06-7088com
Kajiyama, N., Saito, Y., Miyamoto, Y., Yoshimura, M., Nakayama, M., Harada, M., et al. (2000). Lack of association between T-786.RAR.C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene and essential hypertension. Hypertension Research, 23(6), 561-5. doi: 10.1291/hypres.23.561
Luo, J. Q., Wen, J. G., Zhou, H. H., Chen, X. P., & Zhang, W. (2014). Endothelial nitric oxide synthase gene G894T polymorphism and myocardial infarction: A meta-analysis of 34 studies involving 21,068 subjects. PLoS ONE, 9, 87196. doi: 10.1371/journal.pone.0087196
Luo, L., Li, D. H., Wei, S. G., Zhang, H. B., Li, S. B., & Zhao, J. (2013). Polymorphisms in the endothelial nitric oxide synthase gene associated with recurrent miscarriage. Genetics and Molecular Research, 12(3), 3879–86. doi: 10.4238/2013.september.23.6
McElroy, J. P., & Oksenberg, J. R. (2011). Multiple sclerosis genetics 2010. Neurologic Clinics, 29(2), 219-31. doi: 10.1016/j.ncl.2010.12.002
Miyamoto, Y., Saito, Y., Kajiyama, N., Yoshimura, M., Shimasaki, Y., Nakayama, M., et al. (1998). Endothelial nitric oxide synthase gene is positively associated with essential hypertension. Hypertension, 32(1), 3-8. doi: 10.1161/01.hyp.32.1.3
Modin, H., Dai, Y., Masterman, T., Svejgaard, A., Sørensen, P. S., Oturai, A., et al. (2001). No linkage or association of the nitric oxide synthase genes to multiple sclerosis. Journal of Neuroimmunology, 119(1), 95–100. doi: 10.1016/s0165-5728(01)00366-6
Nakayama, M., Yasue, H., Yoshimura, M., Shimasaki, Y., Kugiyama, K., Ogawa, H., et al. (1999). T−786→C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation, 99(22), 2864-70. doi: 10.1161/01.cir.99.22.2864
Nakayama, M., Yasue, H., Yoshimura, M., Shimasaki, Y., Ogawa, H., Kugiyama, K., et al. (2000). T−786→C mutation in the 5′-flanking region of the endothelial nitric oxide synthase gene is associated with myocardial infarction, especially without coronary organic stenosis. The American Journal of Cardiology, 86(6), 628-34. doi: 10.1016/s0002-9149(00)01041-9
Safarinejad, M. R., Shafiei, N., & Safarinejad, S. (2010). The role of endothelial nitric oxide synthase (eNOS) T-786C, G894T, and 4a/b gene polymorphisms in the risk of idiopathic male infertility. Molecular Reproduction and Development, 77(8), 720–7. doi: 10.1002/mrd.21210
Tajouri, L., Ferreira, L., Ovcaric, M., Curtain, R., Lea, R., Csurhes, P., et al (2004). Investigation of a neuronal nitric oxide synthase gene (NOS1) polymorphism in a multiple sclerosis population. Journal of the Neurological Sciences, 218(1-2), 25–8. doi: 10.1016/j.jns.2003.10.006
Veldman, B. A., Spiering, W., Doevendans, P. A., Vervoort, G., Kroon, A. A., de Leeuw, P. W., et al. (2002). The Glu298Asp polymorphism of the NOS 3 gene as a determinant of the baseline production of nitric oxide. Journal of Hypertension, 20(10), 2023–7. doi: 10.1097/00004872-200210000-00022
Wang, X. L., Mahaney, M. C., Sim, A. S., Wang, J., Wang, J., Blangero, J., et al. (1997). Genetic contribution of the endothelial constitutive nitric oxide synthase gene to plasma nitric oxide levels. Arteriosclerosis, Thrombosis, and Vascular Biology, 17(11), 3147-53. doi: 10.1161/01.atv.17.11.3147
Yoshimura, T., Yoshimura, M., Tabata, A., Yasue, H., & Okamura, H. (2001). The missense Glu298Asp variant of the endothelial nitric oxide synthase gene is strongly associated with placental abruption. Human Genetics, 108(3), 181–3. doi: 10.1007/s004390000454
Type of Study: Original |
Subject:
Cellular and molecular Neuroscience
Received: 2016/12/13 | Accepted: 2017/05/5 | Published: 2017/07/1
Received: 2016/12/13 | Accepted: 2017/05/5 | Published: 2017/07/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |