Volume 16, Issue 1 (January & February 2025)
BCN 2025, 16(1): 65-80 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Mollajani R, Taghi Joghataei M, Tehrani-Doost M, Khosrowabadi R. Effect of Bumetanide on Neural Correlates of Emotion Recognition in Youth With Autism Spectrum Disorder: An Event-related Potential Study. BCN 2025; 16 (1) :65-80
URL: http://bcn.iums.ac.ir/article-1-2405-en.html
URL: http://bcn.iums.ac.ir/article-1-2405-en.html
1- Neuroscience Research Center, Iran University of Medical Sciences, Tehran, Iran.
2- Department of Cognitive Neuroscience, Institute for Cognitive Science Studies, Tehran, Iran.
2- Department of Cognitive Neuroscience, Institute for Cognitive Science Studies, Tehran, Iran.
Keywords: Bumetanide, Autism spectrum disorder (ASD), Facial emotion recognition, Event-related potentials (ERPs)
Full-Text [PDF 1634 kb]
| Abstract (HTML)
N170
Regarding the peak amplitude (PA), the results of ANOVA-RM in the PA of this component showed that the main effect of interaction between emotion and treatment was significant in the P7 electrode (P=0.008, F=5.720), and according to the t-test result, it increased significantly in sad faces (P=0.05, T=2.056). However, this electrode’s main effects on emotion and treatment were insignificant (P>0.05). The PAs of the other electrodes were not significant (P>0.05).
Regarding the peak latency (PL), this component was not significant in any electrode (P>0.05), the details of which are provided in Table 4.
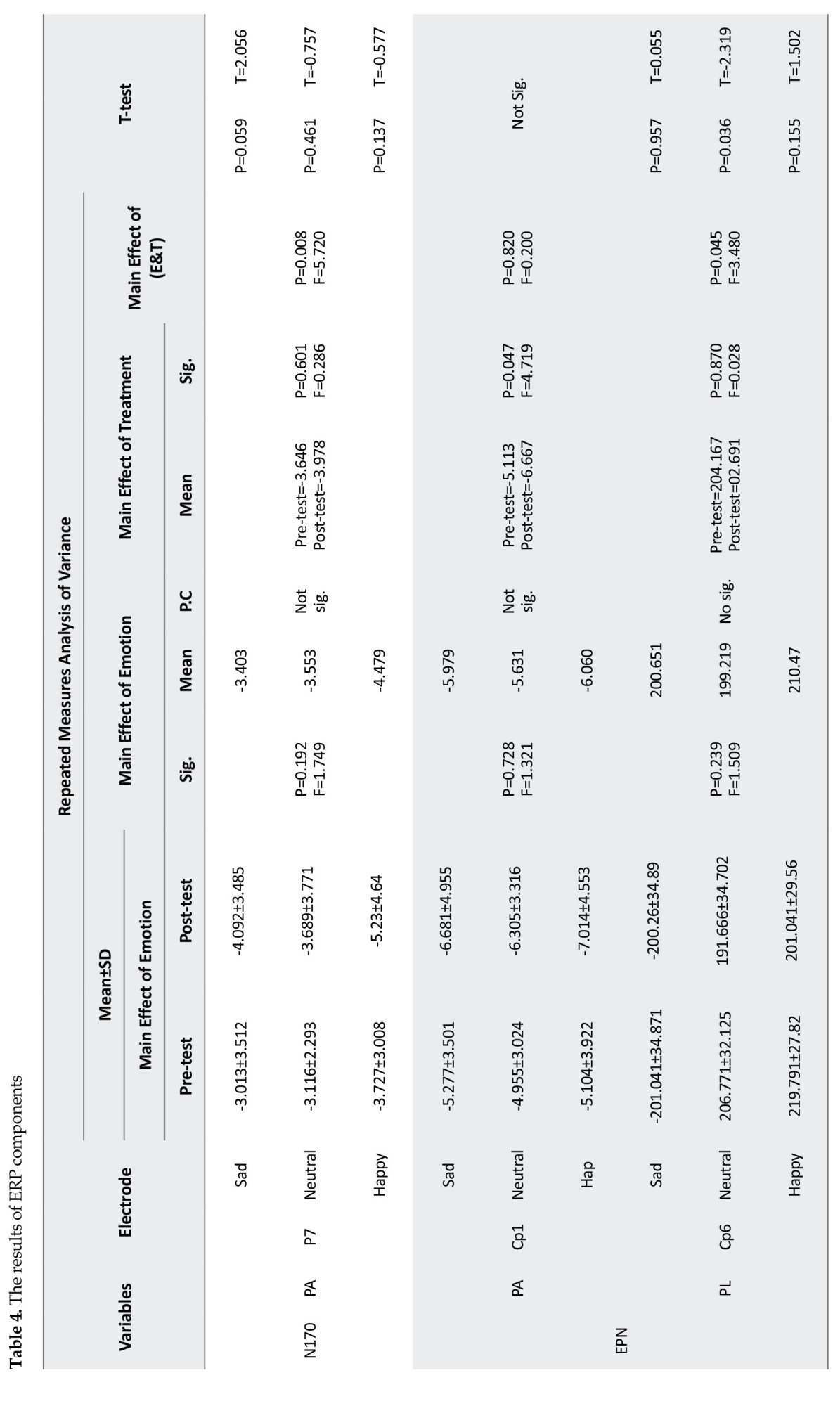
EPN
Based on the ANOVA-RM for PA, the main effect of treatment was significant for Cp1 (P=0.047, F=4.719), and its mean difference in the post-test was greater than the pre-test. Contrarily, the main effects of emotion and the interaction between emotion and treatment were not significant (P>0.05) in this electrode. Further, the PAs of the other electrodes were not significant.
Regarding the peak latencies (PL), the main effect of emotion was significant (P=0.000, F=495.591) for Cp6, and the latency decreased in the post-test compared to the pre-test in all emotional states, but the main effects of treatment and interaction between emotion and treatment were not significant in this electrode (P>0.05). Eventually, the PL of this component was not significant in the other electrodes either (P>0.05). The related data are presented in Table 4.
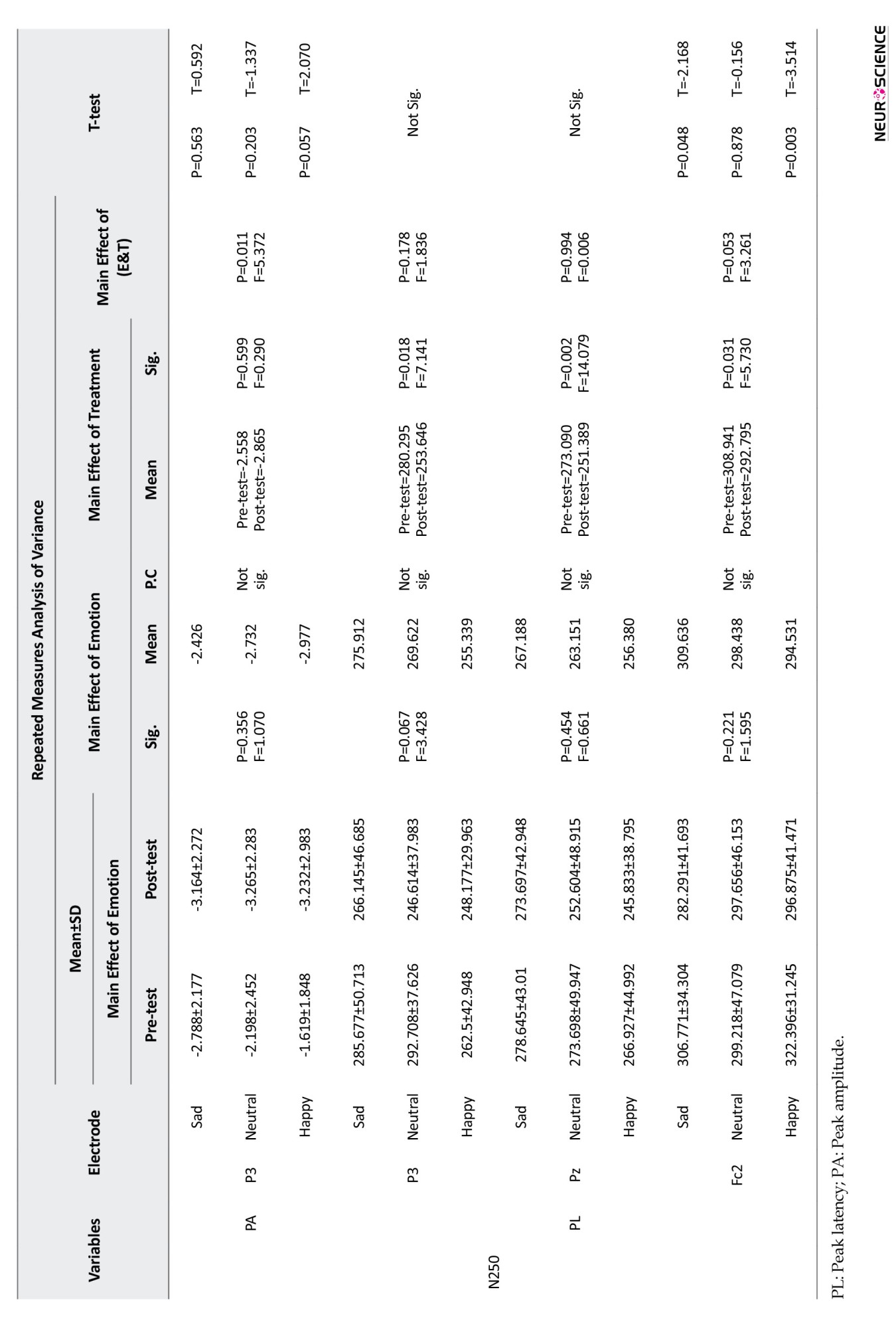
N250
Regarding the PA, the analysis of the results on this component revealed that the main effect of interaction between emotion and the treatment in P3 was significant in terms of PA based on ANOVA results (P=0.011, F=5.372). The results of t-test showed that it increased significantly in happy faces (P=0.05, T=2.070). However, the main effects of emotion and treatment were not significant (P>0.05) in this electrode. Furthermore, the PA of the other electrodes was not significant concerning this component (P>0.05).
Regarding the PL, according to the ANOVA results, the main effects of treatment in P3 (P=0.018, F=7.141) and Pz (P=0.002, F=14.079) were significant and decreased in the post-test, but the main effects of emotion and interaction between emotion and treatment were not significant in these channels (P>0.05). As regards Fc2, the main effects of treatment (P=0.031, F=5.730) and interaction between emotion and treatment (P=0.05 F=3.261) were significant, and based on the results of the t-test (Table 3), there was a decrease in the mean differences in the sad (P=0.048, T=-2.168) and happy (P=0.003, T=3.514) faces.
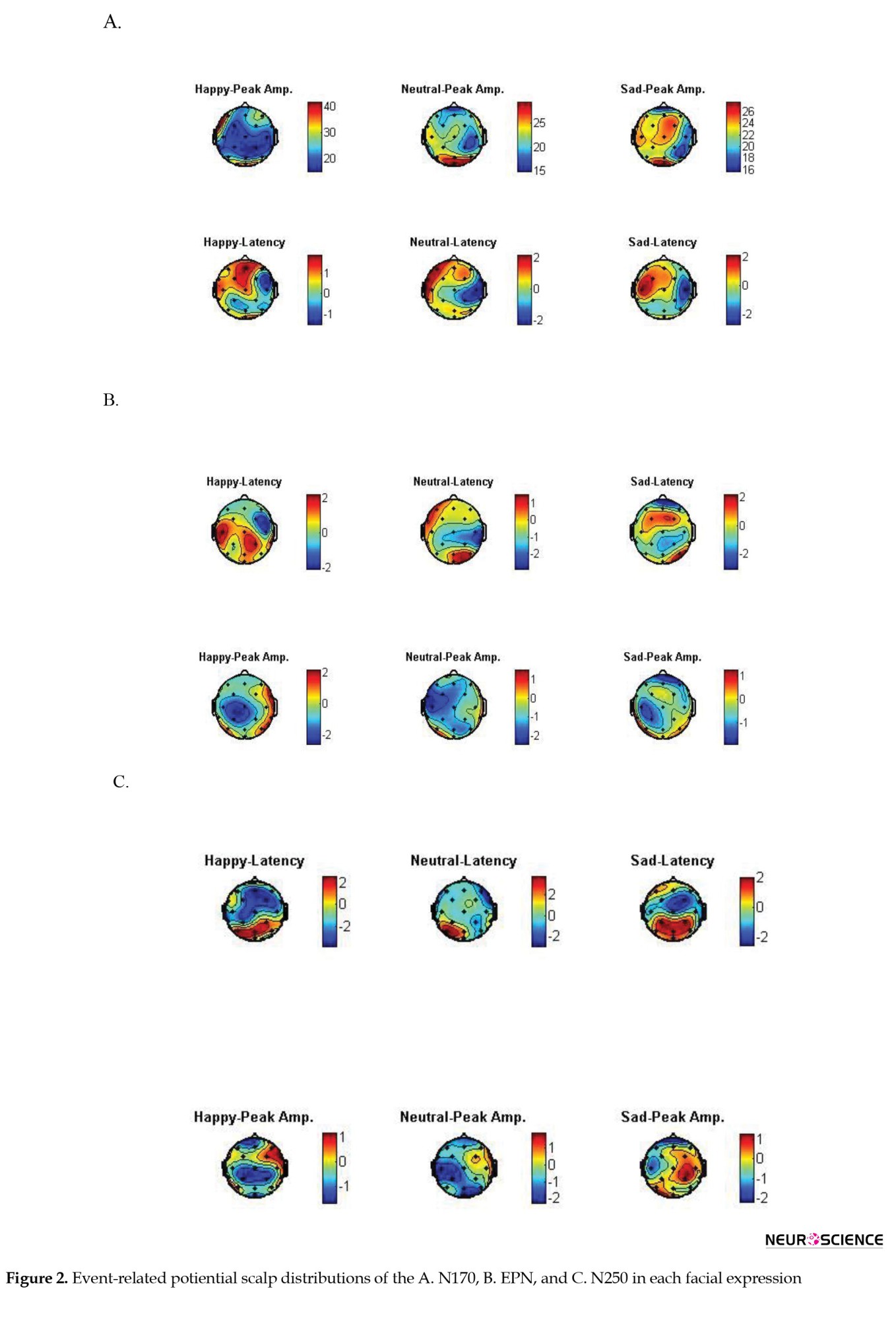
Figure 2, parts A, B, and C show ERP scalp distributions of the N170, EPN, and N250 components, respectively.
Full-Text:
1. Introduction
Autism spectrum disorder (ASD) is a neuro-developmental disorder identified by impaired social communication and repetitive behaviors (American Psychiatric Association [APA], 2013). In recent years, the number of children recognized with ASD has increased, and reports indicate that ASD affects nearly 1% of the population (Baio, 2012). This disorder is characterized by an inability to interact socially, including difficulty making friendships and understanding others' emotions and thoughts (Kaland et al., 2007). Impairment in comprehending others' mental states plays a significant role in social and communicative deficits in ASD (Baron-Cohen et al., 1996). According to Haviland and Lelwica, the ability to identify and distinguish emotional expressions grows in typically developing infants from 10 weeks of age (Haviland & Lelwica, 1987) and continues during childhood (Herba & Phillips, 2004). This ability is delayed in children with ASD and is evaluated using tasks that assess facial emotion recognition, vocal intonation, and body language (Baron-Cohen at al., 2001; Yirmiy et al., 1992). Behavioral studies report a failure in this ability and reduced accuracy in detecting facial emotions, especially negative ones (Ashwin et al., 2006; Corden et al., 2008; Wallace et al., 2008; Rump et al., 2009) as well as voices (Baker et al., 2010). This impairment is supposed to be an essential deficit in ASD (Philip et al., 2010).
In event-related potentials (ERPs) studies, smaller amplitudes, and increased latencies are similar to emotional expressions in patients with ASD compared to those of the control group and confirm the facial emotion processing deficits in ASD. According to these studies, statistically significant differences are found between the groups mainly in P1 and N170 amplitudes and latencies, in which those with ASD represent delayed latencies and lower amplitudes to emotional stimuli compared to typically developing individuals (Apicellaa et al., 2012; Batty et al., 2011; O'Connor et al., 2005).
Accordingly, emotion recognition, especially in the face, demonstrates an ordinary intervention target, which highlights the need for explaining sensory and cognitive processes that uphold social cognition, especially emotion recognition in ASD (Akechi et al., 2010; Lerner et al., 2013).
Social cognition deficiency in this population has remained the main treatment challenge (Weiss & Harris, 2001). On the other hand, the European Medicine Agency (EMA) or Food and Drug Administration (FDA) has not found an approved and effective medication for ASD that significantly improves the core symptoms, especially social interaction and emotion recognition (Lemonnier et al., 2017). Only two approved drugs are available for treating autism-related irritability: Risperidone (5-16 years old) which is regarded as a serotonin type 2A (5-HT2A) and dopamine type 2 (D2) receptor antagonist (McCracken, 2002) and aripiprazole (6-17 years old) which is regarded as partial dopamine (DA) D2 and 5-HT1A agonist, and a 5-HT2A antagonist (Wink et al., 2010). Regardless of their side effects (Fung et al., 2016; Lemonnier et al., 2017), none of these drugs seem to reduce the core symptoms of ASD (Lemonnier et al., 2017).
Bumetanide, a diuretic and chloride cotransporter antagonist, has been recently proposed for treating ASD (Lemonnier et al., 2017; Mollajan et al., 2019). Based on previous studies, the level of chloride and gamma-aminobutyric acid (GABA) ergic signaling changes in ASD and some other developmental disorders (Ben-Ari, 2015; Blaesse et al., 2009; Nardou et al., 2011). Bumetanide has been widely used in adults since 1975 and children since 1986 for treating some disorders such as broncho-pulmonary dysplasia, hypertension, congestive heart failure, and nephrotic syndrome. Bumetanide restores low (Cl−)I levels and shifts GABA from excitation to inhibition (Ben-Ari, 2015). According to a previous study, the maternal infusion of bumetanide in ASD animal models reduced the children's physiological levels (Cl-)I and regulated electrical and behavioral parameters (Eftekhari et al., 2014; Tyzio et al., 2014). Based on these observations, the effects of bumetanide on 5 neonates with autism were evaluated, and the results showed an improvement in the behavioral symptoms of infantile autistic syndrome with no serious side effects (Lemonnier & Ben-Ari, 2010). The results of another study confirm these findings and demonstrate that bumetanide significantly ameliorates the symptoms of ASD. The side effects of the drug in this study are limited to mild hypokalemia, which is managed by a potassium supplement (Lemonnier et al., 2012). Moreover, an open trial study on 7 adolescents and young adults with autism indicates improved facial emotion recognition and the activation of involved brain regions (Hadjikhani et al., 2015). Using functional magnetic resonance imaging (fMRI) and eye-tracking, Hadjikhani et al. found that the administration of bumetanide normalized the amygdala activation level during constrained eye contact with dynamic emotional face stimuli in individuals with ASD. Furthermore, data on eye-tracking revealed that the amount of time allocated to the spontaneous eye gaze increased during a free-viewing mode of identical face stimuli by administering bumetanide. These results show that bumetanide probably improves the specific aspects of social processing in autism (Hadjikhani et al., 2018).
Based on the results of recent studies on the effect of bumetanide, especially at the behavioral level, the present study aimed to evaluate the effect of this drug on facial emotion recognition and emotion processing, as well as changes in neuronal functions using ERPs. We wanted to see if the medication could improve emotion recognition and whether the neural correlates of emotion processing would be changed after the treatment.
2. Materials and Methods
Study participants
Twenty boys were recruited from the child and adolescent psychiatry clinics for this study. They were diagnosed with ASD level 1, needing support based on DSM-5 (high-functioning ASD [ASD-HF], pre-DSM-5 nomenclature). They were within the age range of 7-16 years. Among the participants, 5 cases were excluded from the study before the study because of some behavioral disturbance such as aggression and restlessness due to discontinuing their previous medications (n=2) and before the follow-up phase because of lacking enough cooperation for using the drug regularly and continuing the intervention (n=3). All participants were right-handed, possessed normal or corrected to normal vision, and reported no history of critical neurological and medical problems such as epilepsy, brain injury, or metabolic disorders. Moreover, they had no liver dysfunction, renal disease, cardiac problems, and allergy to sulfonamides. All participants had intelligence quotient (IQ) scores above 70 based on the Wechsler intelligence scale for children (WISC-R).
Study measures
Childhood autism rating scale (CARS)
The CARS contains 15 items, each scored 1-4, ranging from normal, mildly abnormal, moderately abnormal to severely abnormal. The scores collectively add up to 15-60, and 30 is the cutoff point for diagnosing mild autism. Additionally, scores within the range of 30-37 represent mild to moderate autism, whereas those between 38 and 60 demonstrate severe autism (Schopler et al., 1980).
The schedule for affective disorders and schizophrenia for school-age children-present and lifetime version
This schedule for affective disorders and schizophrenia for school-age children and lifetime version (K-SADS-PL) is a semi-structured diagnostic interview used to evaluate current, past, and lifetime diagnostic status in children and adolescents within the age range of 6-18 years (Kaufman et al., 1997). Kaufman et al. (1997) reported various components of the K-SADS, and the psychometric characteristics of its Persian version have well been confirmed for most psychiatric disorders (Shahrivar et al., 2010).
Social responsive scale (SRS)
The SRS was designed to determine the presence of impairments in reciprocal social behaviors (Constantino et al., 2006). This 65-item rating scale measures the severity of each behavior, ranging from mild to severe, producing a total of 5 subscale scores. Parents, teachers, or other frequent care providers can complete this scale in 15-20 minutes. It can be applied in various ways, such as a measure of the severity of social impairment in children at risk of ASD or a general population screening instrument. In addition, SRS can be utilized for behavioral, genetic, and intervention evaluation studies. The Persian version of this questionnaire was validated in a community sample of Farsi-speaking individuals (Tehrani‑Doost et al., 2018).
WISC-revised (WISC-R)
The WISC-R is individually administered to children aged 6-16 years to evaluate their intelligence levels (Wechsler, 1949). It includes verbal (i.e. information, comprehension, mathematics, numbers, similarities, and vocabulary) and non-verbal (i.e. picture arrangement, picture completion, object assembly, block design, coding, and Maze) intelligence scales. The Persian version of this questionnaire was validated in a community sample of Farsi-speaking individuals (Shahym, 1991). The verbal intelligence scale was used in this study.
ERP task
The ERP task was developed using different faces showing sad, happy, and neutral expressions (4 men and 4 women) selected from the Radboud Faces Database (Lang, 1997). The selected faces were validated in terms of their emotions in 60 normal individuals with the same age range and gender. All the face stimuli covered a visual angle of about 5.5×7.5º and were presented on a computer screen in front of a gray background. The task in this study was designed by the eevokeTM software, with 144 stimuli, and each facial expression (i.e. happy, sad, and neutral) was repeated 48 times. The task included 3 blocks, 48 stimuli per block, which were randomly presented to avoid the lack of interest in the subjects. The presentation time of each stimulus included 2000 ms, followed by continuously presenting a blank screen with a white fixation cross at the center for 1000±300 ms. The participants were requested to look at the stimuli while maintaining fixation carefully and to avoid other body movements or deep breathing.
ERP recording
ERPs were recorded using the ANT (advanced neuron technology) system with 32 Ag/AgCl sintered electrodes mounted on an elastic cap following the 10/20 international system. The averaged mastoids were employed as the reference and grounded to the Fz channel. Furthermore, two electrodes were applied to monitor vertical eye movements to remove offline artifacts, followed by keeping the impedance below 5 kΩ throughout the procedure. Then, raw data were filtered online with a band-pass from 0.01 to 100 Hz and sampled at 250 Hz. Next, the acquired data resembled offline at a 250-Hz sampling rate. Moreover, baseline corrections were performed approximately 200 ms before the stimulus onset in order to confirm the quality of the recording. The recording condition was in line with the required standards (i.e. under the electrical shield and in a sound-attenuated and dim-lighted room).
Common blood test
These tests included alkaline phosphatase, uric acid, creatine, sodium, potassium, and the kidney and liver functions of the participants.
Study procedure
First, the participants were interviewed by a child and adolescent psychiatrist and diagnosed as ASD-HF (level 1-needing support) based on DSM-5 criteria. Then, they were evaluated using CARS, K-SADS-PL, and SRS. The WISC-R was utilized to evaluate intellectual abilities as well. The blood tests were also performed on the participants. The participants were off medication one week before the study except for melatonin at a dose of 3 mg. To perform the ERP experiment, each participant was asked to sit at a 60-cm distance from the monitor screen (a 19-w monitor, LG F900P with a 100-Hz screen refresh rate) and to fix his head on a chin rest and watch the monitor screen. After the evaluations, bumetanide was prescribed at 1 mg daily (0.5 mg twice a day) for 3 months based on the protocol of previous studies (Lemonnier et al., 2012; Lemonnier et al., 2017). The medication adherence and probable adverse effects were assessed by asking the parents based on the number of pills used and using common blood tests periodically at weeks 1, 6, and 12 of the trial in case of the adverse effects. The CARS, SRS, and blood tests were repeated at weeks 6 and 12 again. The ERP experiment was repeated less than one week after the intervention.
Data measure and analysis
The latencies and amplitudes of traditionally analyzed channels for N170 (P7, P8) (Eimer, 2000; Eimer & Holmes, 2007; Luo et al., 2010; Pesciarelli, 2011), early posterior negativity (EPN) (Cp5, Cp6, P3, P4) (Junghöfer et al., 2001; Rellecke et al., 2013; Schupp et al., 2004), and N250 (C3, C4, Fc1, Fc2, F3, F4) (Eimer, 2000; Harms et al., 2010; Luo et al., 2010; Pesciarelli, 2011) as the primary variables and other exploratory electrode regions for these components as the secondary variables evaluated in this study. The time epochs selected for these components included 130-250 ms for N170, 150-250 ms for EPN, and 215-350 ms for N250. The data were analyzed using the MATLAB software, version 2014b integrated with EEGLAB and ERPLAB. Then, the acquired data were approached offline at a 250-Hz sampling rate and digitally filtered offline from 0.1 to 30 Hz. The continuous electroencephalographic data were divided into 1000-ms epochs starting 200 ms before and 800 ms after the stimulus onset. The eye movements were corrected using independent component analysis (Makeig & Enghof, 2000). Next, a semi-automatic rejection further eliminated the remaining artifacts, removing epochs exceeding ±100 μV in each channel. The mean rejection rate of the epochs was 7.66%, and there were no significant differences between the conditions.
Statical analysis
The paired t-test and the repeated measures analysis of variance (ANOVA), including two factors, emotion (3 levels: Happy, sad, and neutral) and treatment (2 levels: Pre-test and post-test), were conducted to find the main effect of emotion, treatment, and their interactions on latencies and amplitude for each electrode separately. The P were corrected using the Bonferroni correction.
3. Results
Characteristics of the participants are shown in Tables 1 and 2.

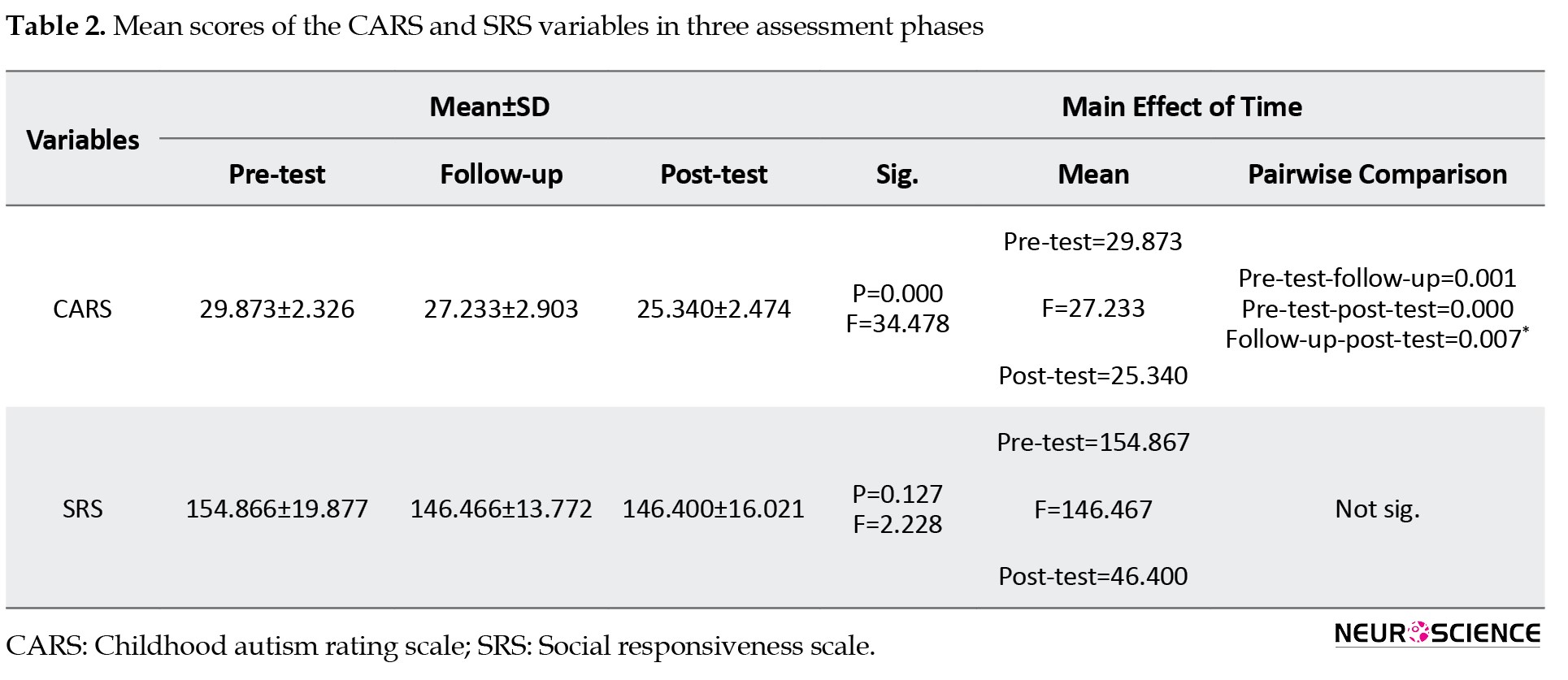
The results of the CARS and SRS
With regard to CARS variables, the repeated measures analysis revealed a significant amelioration of the total score during and after the intervention (P<0.05). The total score of SRS failed to differ markedly during and after treatment with bumetanide (P>0.05), although it decreased due to the intervention (Table 2).
Behavioral data of ERP recording
Response accuracy (RA)
The repeated measures analysis showed that the main effect of emotion on the correct response was significant (P=0.000, F=13.121). Based on the pairwise comparisons, happy faces were detected more significantly than sad (P=0.000) and neutral faces (P=0.003). Additionally, the main impact of treatment was significant on RA, while the interaction between emotion and treatment demonstrated no significant effect (Table 3).
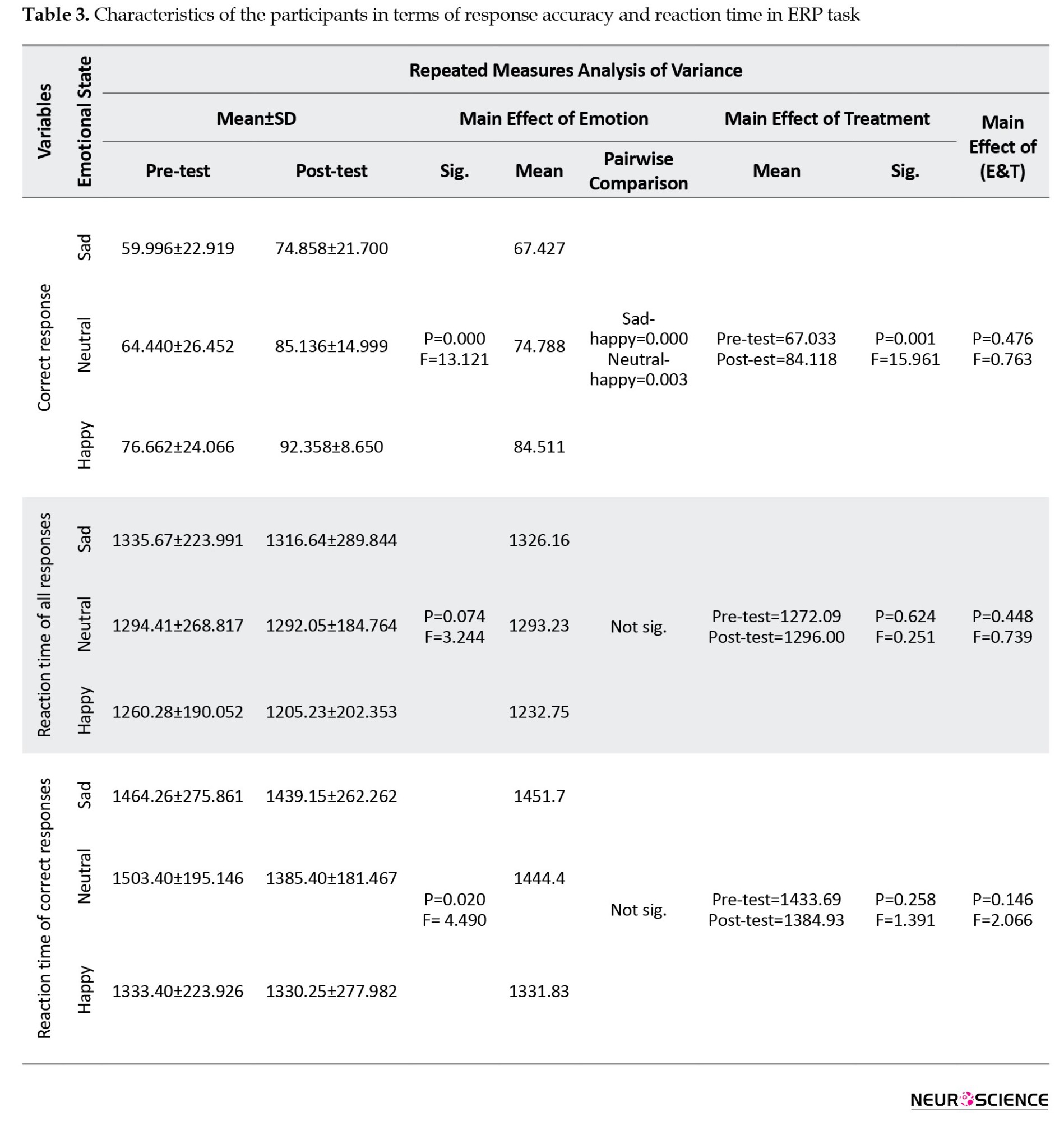
Reaction time (RT)
Regarding the RT of all responses (RT_AR), the main effects in RT_AR were not significant in any emotional state (P>0.05) according to the repeated measures ANOVA (ANOVA-RM) results.
Regarding the RT of correct responses (RT_CR), the main effect of emotion in RT_CR was significant (P=0.020, F=4.490) based on ANOVA results. However, nothing was significant in the pairwise comparison with Bonferroni correction between the emotional states. In addition, the main effects of treatment (P=0.258, F=1.391) and interaction between emotion and treatment (P=0.146, F=2.066) were not significant regarding this variable (Table 3).
ERP components
Figures 1A, and 1B show the grand averages of ERP waves of the three emotional states pre- and post-treatment.
Autism spectrum disorder (ASD) is a neuro-developmental disorder identified by impaired social communication and repetitive behaviors (American Psychiatric Association [APA], 2013). In recent years, the number of children recognized with ASD has increased, and reports indicate that ASD affects nearly 1% of the population (Baio, 2012). This disorder is characterized by an inability to interact socially, including difficulty making friendships and understanding others' emotions and thoughts (Kaland et al., 2007). Impairment in comprehending others' mental states plays a significant role in social and communicative deficits in ASD (Baron-Cohen et al., 1996). According to Haviland and Lelwica, the ability to identify and distinguish emotional expressions grows in typically developing infants from 10 weeks of age (Haviland & Lelwica, 1987) and continues during childhood (Herba & Phillips, 2004). This ability is delayed in children with ASD and is evaluated using tasks that assess facial emotion recognition, vocal intonation, and body language (Baron-Cohen at al., 2001; Yirmiy et al., 1992). Behavioral studies report a failure in this ability and reduced accuracy in detecting facial emotions, especially negative ones (Ashwin et al., 2006; Corden et al., 2008; Wallace et al., 2008; Rump et al., 2009) as well as voices (Baker et al., 2010). This impairment is supposed to be an essential deficit in ASD (Philip et al., 2010).
In event-related potentials (ERPs) studies, smaller amplitudes, and increased latencies are similar to emotional expressions in patients with ASD compared to those of the control group and confirm the facial emotion processing deficits in ASD. According to these studies, statistically significant differences are found between the groups mainly in P1 and N170 amplitudes and latencies, in which those with ASD represent delayed latencies and lower amplitudes to emotional stimuli compared to typically developing individuals (Apicellaa et al., 2012; Batty et al., 2011; O'Connor et al., 2005).
Accordingly, emotion recognition, especially in the face, demonstrates an ordinary intervention target, which highlights the need for explaining sensory and cognitive processes that uphold social cognition, especially emotion recognition in ASD (Akechi et al., 2010; Lerner et al., 2013).
Social cognition deficiency in this population has remained the main treatment challenge (Weiss & Harris, 2001). On the other hand, the European Medicine Agency (EMA) or Food and Drug Administration (FDA) has not found an approved and effective medication for ASD that significantly improves the core symptoms, especially social interaction and emotion recognition (Lemonnier et al., 2017). Only two approved drugs are available for treating autism-related irritability: Risperidone (5-16 years old) which is regarded as a serotonin type 2A (5-HT2A) and dopamine type 2 (D2) receptor antagonist (McCracken, 2002) and aripiprazole (6-17 years old) which is regarded as partial dopamine (DA) D2 and 5-HT1A agonist, and a 5-HT2A antagonist (Wink et al., 2010). Regardless of their side effects (Fung et al., 2016; Lemonnier et al., 2017), none of these drugs seem to reduce the core symptoms of ASD (Lemonnier et al., 2017).
Bumetanide, a diuretic and chloride cotransporter antagonist, has been recently proposed for treating ASD (Lemonnier et al., 2017; Mollajan et al., 2019). Based on previous studies, the level of chloride and gamma-aminobutyric acid (GABA) ergic signaling changes in ASD and some other developmental disorders (Ben-Ari, 2015; Blaesse et al., 2009; Nardou et al., 2011). Bumetanide has been widely used in adults since 1975 and children since 1986 for treating some disorders such as broncho-pulmonary dysplasia, hypertension, congestive heart failure, and nephrotic syndrome. Bumetanide restores low (Cl−)I levels and shifts GABA from excitation to inhibition (Ben-Ari, 2015). According to a previous study, the maternal infusion of bumetanide in ASD animal models reduced the children's physiological levels (Cl-)I and regulated electrical and behavioral parameters (Eftekhari et al., 2014; Tyzio et al., 2014). Based on these observations, the effects of bumetanide on 5 neonates with autism were evaluated, and the results showed an improvement in the behavioral symptoms of infantile autistic syndrome with no serious side effects (Lemonnier & Ben-Ari, 2010). The results of another study confirm these findings and demonstrate that bumetanide significantly ameliorates the symptoms of ASD. The side effects of the drug in this study are limited to mild hypokalemia, which is managed by a potassium supplement (Lemonnier et al., 2012). Moreover, an open trial study on 7 adolescents and young adults with autism indicates improved facial emotion recognition and the activation of involved brain regions (Hadjikhani et al., 2015). Using functional magnetic resonance imaging (fMRI) and eye-tracking, Hadjikhani et al. found that the administration of bumetanide normalized the amygdala activation level during constrained eye contact with dynamic emotional face stimuli in individuals with ASD. Furthermore, data on eye-tracking revealed that the amount of time allocated to the spontaneous eye gaze increased during a free-viewing mode of identical face stimuli by administering bumetanide. These results show that bumetanide probably improves the specific aspects of social processing in autism (Hadjikhani et al., 2018).
Based on the results of recent studies on the effect of bumetanide, especially at the behavioral level, the present study aimed to evaluate the effect of this drug on facial emotion recognition and emotion processing, as well as changes in neuronal functions using ERPs. We wanted to see if the medication could improve emotion recognition and whether the neural correlates of emotion processing would be changed after the treatment.
2. Materials and Methods
Study participants
Twenty boys were recruited from the child and adolescent psychiatry clinics for this study. They were diagnosed with ASD level 1, needing support based on DSM-5 (high-functioning ASD [ASD-HF], pre-DSM-5 nomenclature). They were within the age range of 7-16 years. Among the participants, 5 cases were excluded from the study before the study because of some behavioral disturbance such as aggression and restlessness due to discontinuing their previous medications (n=2) and before the follow-up phase because of lacking enough cooperation for using the drug regularly and continuing the intervention (n=3). All participants were right-handed, possessed normal or corrected to normal vision, and reported no history of critical neurological and medical problems such as epilepsy, brain injury, or metabolic disorders. Moreover, they had no liver dysfunction, renal disease, cardiac problems, and allergy to sulfonamides. All participants had intelligence quotient (IQ) scores above 70 based on the Wechsler intelligence scale for children (WISC-R).
Study measures
Childhood autism rating scale (CARS)
The CARS contains 15 items, each scored 1-4, ranging from normal, mildly abnormal, moderately abnormal to severely abnormal. The scores collectively add up to 15-60, and 30 is the cutoff point for diagnosing mild autism. Additionally, scores within the range of 30-37 represent mild to moderate autism, whereas those between 38 and 60 demonstrate severe autism (Schopler et al., 1980).
The schedule for affective disorders and schizophrenia for school-age children-present and lifetime version
This schedule for affective disorders and schizophrenia for school-age children and lifetime version (K-SADS-PL) is a semi-structured diagnostic interview used to evaluate current, past, and lifetime diagnostic status in children and adolescents within the age range of 6-18 years (Kaufman et al., 1997). Kaufman et al. (1997) reported various components of the K-SADS, and the psychometric characteristics of its Persian version have well been confirmed for most psychiatric disorders (Shahrivar et al., 2010).
Social responsive scale (SRS)
The SRS was designed to determine the presence of impairments in reciprocal social behaviors (Constantino et al., 2006). This 65-item rating scale measures the severity of each behavior, ranging from mild to severe, producing a total of 5 subscale scores. Parents, teachers, or other frequent care providers can complete this scale in 15-20 minutes. It can be applied in various ways, such as a measure of the severity of social impairment in children at risk of ASD or a general population screening instrument. In addition, SRS can be utilized for behavioral, genetic, and intervention evaluation studies. The Persian version of this questionnaire was validated in a community sample of Farsi-speaking individuals (Tehrani‑Doost et al., 2018).
WISC-revised (WISC-R)
The WISC-R is individually administered to children aged 6-16 years to evaluate their intelligence levels (Wechsler, 1949). It includes verbal (i.e. information, comprehension, mathematics, numbers, similarities, and vocabulary) and non-verbal (i.e. picture arrangement, picture completion, object assembly, block design, coding, and Maze) intelligence scales. The Persian version of this questionnaire was validated in a community sample of Farsi-speaking individuals (Shahym, 1991). The verbal intelligence scale was used in this study.
ERP task
The ERP task was developed using different faces showing sad, happy, and neutral expressions (4 men and 4 women) selected from the Radboud Faces Database (Lang, 1997). The selected faces were validated in terms of their emotions in 60 normal individuals with the same age range and gender. All the face stimuli covered a visual angle of about 5.5×7.5º and were presented on a computer screen in front of a gray background. The task in this study was designed by the eevokeTM software, with 144 stimuli, and each facial expression (i.e. happy, sad, and neutral) was repeated 48 times. The task included 3 blocks, 48 stimuli per block, which were randomly presented to avoid the lack of interest in the subjects. The presentation time of each stimulus included 2000 ms, followed by continuously presenting a blank screen with a white fixation cross at the center for 1000±300 ms. The participants were requested to look at the stimuli while maintaining fixation carefully and to avoid other body movements or deep breathing.
ERP recording
ERPs were recorded using the ANT (advanced neuron technology) system with 32 Ag/AgCl sintered electrodes mounted on an elastic cap following the 10/20 international system. The averaged mastoids were employed as the reference and grounded to the Fz channel. Furthermore, two electrodes were applied to monitor vertical eye movements to remove offline artifacts, followed by keeping the impedance below 5 kΩ throughout the procedure. Then, raw data were filtered online with a band-pass from 0.01 to 100 Hz and sampled at 250 Hz. Next, the acquired data resembled offline at a 250-Hz sampling rate. Moreover, baseline corrections were performed approximately 200 ms before the stimulus onset in order to confirm the quality of the recording. The recording condition was in line with the required standards (i.e. under the electrical shield and in a sound-attenuated and dim-lighted room).
Common blood test
These tests included alkaline phosphatase, uric acid, creatine, sodium, potassium, and the kidney and liver functions of the participants.
Study procedure
First, the participants were interviewed by a child and adolescent psychiatrist and diagnosed as ASD-HF (level 1-needing support) based on DSM-5 criteria. Then, they were evaluated using CARS, K-SADS-PL, and SRS. The WISC-R was utilized to evaluate intellectual abilities as well. The blood tests were also performed on the participants. The participants were off medication one week before the study except for melatonin at a dose of 3 mg. To perform the ERP experiment, each participant was asked to sit at a 60-cm distance from the monitor screen (a 19-w monitor, LG F900P with a 100-Hz screen refresh rate) and to fix his head on a chin rest and watch the monitor screen. After the evaluations, bumetanide was prescribed at 1 mg daily (0.5 mg twice a day) for 3 months based on the protocol of previous studies (Lemonnier et al., 2012; Lemonnier et al., 2017). The medication adherence and probable adverse effects were assessed by asking the parents based on the number of pills used and using common blood tests periodically at weeks 1, 6, and 12 of the trial in case of the adverse effects. The CARS, SRS, and blood tests were repeated at weeks 6 and 12 again. The ERP experiment was repeated less than one week after the intervention.
Data measure and analysis
The latencies and amplitudes of traditionally analyzed channels for N170 (P7, P8) (Eimer, 2000; Eimer & Holmes, 2007; Luo et al., 2010; Pesciarelli, 2011), early posterior negativity (EPN) (Cp5, Cp6, P3, P4) (Junghöfer et al., 2001; Rellecke et al., 2013; Schupp et al., 2004), and N250 (C3, C4, Fc1, Fc2, F3, F4) (Eimer, 2000; Harms et al., 2010; Luo et al., 2010; Pesciarelli, 2011) as the primary variables and other exploratory electrode regions for these components as the secondary variables evaluated in this study. The time epochs selected for these components included 130-250 ms for N170, 150-250 ms for EPN, and 215-350 ms for N250. The data were analyzed using the MATLAB software, version 2014b integrated with EEGLAB and ERPLAB. Then, the acquired data were approached offline at a 250-Hz sampling rate and digitally filtered offline from 0.1 to 30 Hz. The continuous electroencephalographic data were divided into 1000-ms epochs starting 200 ms before and 800 ms after the stimulus onset. The eye movements were corrected using independent component analysis (Makeig & Enghof, 2000). Next, a semi-automatic rejection further eliminated the remaining artifacts, removing epochs exceeding ±100 μV in each channel. The mean rejection rate of the epochs was 7.66%, and there were no significant differences between the conditions.
Statical analysis
The paired t-test and the repeated measures analysis of variance (ANOVA), including two factors, emotion (3 levels: Happy, sad, and neutral) and treatment (2 levels: Pre-test and post-test), were conducted to find the main effect of emotion, treatment, and their interactions on latencies and amplitude for each electrode separately. The P were corrected using the Bonferroni correction.
3. Results
Characteristics of the participants are shown in Tables 1 and 2.


The results of the CARS and SRS
With regard to CARS variables, the repeated measures analysis revealed a significant amelioration of the total score during and after the intervention (P<0.05). The total score of SRS failed to differ markedly during and after treatment with bumetanide (P>0.05), although it decreased due to the intervention (Table 2).
Behavioral data of ERP recording
Response accuracy (RA)
The repeated measures analysis showed that the main effect of emotion on the correct response was significant (P=0.000, F=13.121). Based on the pairwise comparisons, happy faces were detected more significantly than sad (P=0.000) and neutral faces (P=0.003). Additionally, the main impact of treatment was significant on RA, while the interaction between emotion and treatment demonstrated no significant effect (Table 3).

Reaction time (RT)
Regarding the RT of all responses (RT_AR), the main effects in RT_AR were not significant in any emotional state (P>0.05) according to the repeated measures ANOVA (ANOVA-RM) results.
Regarding the RT of correct responses (RT_CR), the main effect of emotion in RT_CR was significant (P=0.020, F=4.490) based on ANOVA results. However, nothing was significant in the pairwise comparison with Bonferroni correction between the emotional states. In addition, the main effects of treatment (P=0.258, F=1.391) and interaction between emotion and treatment (P=0.146, F=2.066) were not significant regarding this variable (Table 3).
ERP components
Figures 1A, and 1B show the grand averages of ERP waves of the three emotional states pre- and post-treatment.
N170
Regarding the peak amplitude (PA), the results of ANOVA-RM in the PA of this component showed that the main effect of interaction between emotion and treatment was significant in the P7 electrode (P=0.008, F=5.720), and according to the t-test result, it increased significantly in sad faces (P=0.05, T=2.056). However, this electrode’s main effects on emotion and treatment were insignificant (P>0.05). The PAs of the other electrodes were not significant (P>0.05).
Regarding the peak latency (PL), this component was not significant in any electrode (P>0.05), the details of which are provided in Table 4.


EPN
Based on the ANOVA-RM for PA, the main effect of treatment was significant for Cp1 (P=0.047, F=4.719), and its mean difference in the post-test was greater than the pre-test. Contrarily, the main effects of emotion and the interaction between emotion and treatment were not significant (P>0.05) in this electrode. Further, the PAs of the other electrodes were not significant.
Regarding the peak latencies (PL), the main effect of emotion was significant (P=0.000, F=495.591) for Cp6, and the latency decreased in the post-test compared to the pre-test in all emotional states, but the main effects of treatment and interaction between emotion and treatment were not significant in this electrode (P>0.05). Eventually, the PL of this component was not significant in the other electrodes either (P>0.05). The related data are presented in Table 4.
N250
Regarding the PA, the analysis of the results on this component revealed that the main effect of interaction between emotion and the treatment in P3 was significant in terms of PA based on ANOVA results (P=0.011, F=5.372). The results of t-test showed that it increased significantly in happy faces (P=0.05, T=2.070). However, the main effects of emotion and treatment were not significant (P>0.05) in this electrode. Furthermore, the PA of the other electrodes was not significant concerning this component (P>0.05).
Regarding the PL, according to the ANOVA results, the main effects of treatment in P3 (P=0.018, F=7.141) and Pz (P=0.002, F=14.079) were significant and decreased in the post-test, but the main effects of emotion and interaction between emotion and treatment were not significant in these channels (P>0.05). As regards Fc2, the main effects of treatment (P=0.031, F=5.730) and interaction between emotion and treatment (P=0.05 F=3.261) were significant, and based on the results of the t-test (Table 3), there was a decrease in the mean differences in the sad (P=0.048, T=-2.168) and happy (P=0.003, T=3.514) faces.
Figure 2, parts A, B, and C show ERP scalp distributions of the N170, EPN, and N250 components, respectively.
In this study, the results of the blood tests showed no significant changes in blood serum potassium before and after the intervention, indicating that bumetanide failed to increase the risk of hypokalemia. Moreover, the adverse events of bumetanide only included mild diuresis, which was not salient.
4. Discussion
Based on previous studies, bumetanide medication, which alters the GABAergic system from excitation to inhibition (Ben-Ari, 2015; Blaesse et al., 2009; Nardou et al., 2011), can reduce the core symptoms of ASD. One of the main outcomes of this disorder is the impairment of social interaction, which is caused by deficits in understanding others’ mental states, including their emotions through facial expressions (Baron-Cohen et al., 1996, Baron-Cohen et al., 2001; Kaland et al., 2007; Yirmiya et al., 1992). Moreover, based on Hadjikhani’s findings, bumetanide can improve emotion recognition and alter brain activation related to this function in people with ASD (Hadjikhani et al., 2018; Hadjikhani et al., 2015). According to these findings, we hypothesized that bumetanide could change neural responses based on action potentials related to facial emotion recognition in individuals with ASD. Therefore, we conducted the present study to evaluate the effect of bumetanide on facial emotion recognition and neural correlates of boys within the age range of 7-16 years old with ASD level 1 (needing support) according to DSM-5 classification. The evaluations included CARS, K-SADS-PL, WISC-R, SRS, ERP recording, and common blood tests. The CARS, SRS, and blood tests were repeated at weeks 6 and 12, and ERP recording was repeated 12 weeks after treatment. The participants’ blood tests had no significant changes during the intervention, and no obvious side effects were reported for medication.
Effects of bumetanide treatment on core symptoms of ASD
In this study, the total score of CARS showed a significant improvement in terms of clinical and general behavioral symptoms. These findings confirmed our hypothesis, indicating that bumetanide decreases the clinical and behavioral aspects of ASD and ameliorates its core symptoms. This finding is consistent with the results of Lemonnier and Ben-Ari (2010), Lemonnier et al. 2012, Lemonnier et al., 2013, and Lemonnier et al.. 2017), and Hadjikhani et al. (2015).
Effects of bumetanide on behavioral data of ERP recording
Regarding the response accuracy (RA) and based on the results, the main effect of emotion was significant on RA (without considering the effect of bumetanide) so that the happy faces were recognized more than the sad ones. This medication can improve the detection of facial emotions, especially happy ones, and confirm one of our hypotheses. This finding is consistent with the results of some studies that reported impaired detecting of negative emotions in individuals with ASD (Ashwin et al., 2006; Corden et al., 2008; Wallace et al., 2008). Considering feature-based processing in ASD (Behrmann, Thomas, & Humphreys, 2006), these individuals have more difficulty recognizing sadness than happiness, which can be recognized by the upturned mouth alone (Harms et al., 2010). It was interpreted that processing of the mouth region could be sufficient for detecting happiness compared to the other emotional states (Adolphs et al., 2005; Uljarevic & Hamilton, 2013).
It was found in this study that RA increased after treatment with bumetanide regardless of the type of emotion. This finding is consistent with the study results, which showed that bumetanide increased RA and reduced reaction time in an emotional matching task along with improving emotion recognition and increased activity of the brain regions involved in emotion processing (Hadjikhani et al., 2018; Hadjikhani et al. 2015; Vlaskamp, 2015).
Regarding the reaction time of all responses (RT_AR) and correct responses (RT_CR), according to our findings, the main effect of emotion was not significant among the emotional states in terms of the RT_AR and RT_CR. This finding can be interpreted so that bumetanide did not change the time spent recognizing emotions through faces. It was proposed that this duration would be increased after treatment because of the slow response time in recognizing emotions in individuals with ASD.
The main effects of treatment and the interaction between emotion and treatment were not significant regarding these variables, meaning that the intervention had no significant effect on the response time. This result may be due to the small sample size.
Effects of bumetanide on ERP components
Data analysis showed that the PA and PL of ERP components, including N170, EPN, and N250, significantly differ in some electrodes between pre- and post-treatment.
N170 component
Regarding the PA of N170, a significant increase was observed in the P7 electrode after treatment with bumetanide. It has been shown that individuals with ASD had smaller N170 amplitude compared to normal people (Lerner et al., 2013; O'Connor et al., 2005; Wong et al., 2008). Therefore, bumetanide seems to have led to more neural responses while recognizing sad expressions. This finding is in line with Hadjikhani's results when they studied brain activation in individuals with ASD after bumetanide treatment. They found increased brain activity in regions involved in processing emotional facial expressions, including the inferior occipital cortex and the fusiform cortex, equivalent to the P7 increased amplitude seen in our study (Hadjikhani et al., 2015).
The PL of N170 was not significant in any channels. It seems that bumetanide had no significant effect on the speed of neural response while watching facial emotions. The behavioral responses mentioned above are consistent with neural responses reflected in the ERPS's PL.
EPN components
Based on the result, the PA of EPN was significantly increased in Cp1 after treatment. This finding shows that bumetanide increased neural brain activity while recognizing emotions, increasing accuracy in detecting facial expressions.
The PL of this component was significantly decreased in the Cp6 channel just in neutral faces.
EPN is the emotion-sensitive early posterior negativity that arises over temporo-occipital electrodes about 150 to 300 ms after the stimulus onset. It is related to the visual cortex’s increased perceptual encoding of emotional stimuli (Rellecke et al., 2012). Studies on children with autism indicated a weak and delayed appearance of this component in these patients (Faja et al., 2016; Monteiro et al., 2017; Rellecke et al., 2013). Based on the current study, bumetanide can increase neural activity and cognitive effort in the temporo-occipital region while approaching emotions.
N250 component
The results showed that the PA of N250 significantly increased in the P3 channel while recognizing happy faces. It means that bumetanide can increase neural responses in individuals with ASD while detecting positive emotions, which leads to increased accuracy in detecting emotions.
Based on this study's results, bumetanide treatment significantly decreased the latency of N250 in P3, Pz, and Fc2 electrodes. Moreover, significant decreases were shown for happy and sad faces in the Fc2 channel based on the significant interaction between emotion and treatment in repeated measure analysis. This finding demonstrated that using bumetanide can increase the speed of neural response in youth with ASD while viewing happy and sad faces, which causes more rapid recognition of these emotions.
The N250 marks higher-order face processing, such as affect decoding. This ERP component reflects the earliest evaluation of emotion processing involved in facial emotion perception (Lerner et al., 2013). Other research studies showed that it is associated with decoding emotional concepts and is evoked by observing an emotionally expressive face (Balconi & Pozzoli, 2008; Carretié et al., 2001; Wynn et al., 2008). In the present study, the amplitude of this component increased in P3, especially in happy faces, while its latency significantly decreased for sad and happy faces in P3, Pz, and Fc2 after the intervention. It means that bumetanide can increase neural involvement and rapid response in reaction to emotional faces and recognizing the content of facial expressions. Based on previous studies, it has been found a decrease in amplitude and an increase in latency of this component in individuals with ASD (Jeste & Nelson, 2009; Lerner et al., 2013). Therefore, it seems that bumetanide can increase neural activity and its response speed in individuals with ASD while evaluating emotions.
5. Conclusion
To the best of our knowledge, this study was the first one to evaluate the effect of bumetanide on facial emotion recognition and its neural correlates in youth with ASD using ERP. The findings showed that using bumetanide improved the clinical and behavioral aspects of ASD according to the total score of the CARS. Furthermore, the results demonstrated an increase in the amplitude and a decrease in the latency of ERP components, including N170, EPN, and N250, in some channels while viewing happy and sad faces. Thus, it seems that using bumetanide can increase the ability of emotion recognition and alter the neural activity of involved brain areas. These results confirmed previous findings regarding the improvements in symptoms and brain function of individuals with ASD after bumetanide treatment.
The results of this study should be considered in light of some limitations, including the small sample size and the absence of a placebo control group. A multi-center, double-blind, placebo-controlled study with a larger sample size is recommended.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of the Iran University of Medical Sciences, Tehran, Iran (Code: IR.IUMS.REC.1395.4537), and a consent form was obtained from the participants’ parents before initiating the study.
Funding
The Cognitive Sciences and Technologies Council, Tehran, Iran, supported this study. The funder played no role in study design, data collection and analysis, decision for publication, or manuscript preparation.
Authors' contributions
All authors contributed equally to the conception and design of the study, data collection and analysis, interception of the results and drafting of the manuscript. Each author approved the final version of the manuscript for submission.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the Cognitive Sciences and Technologies Council, Tehran, Iran, for funding this research project.
References
Adolphs, R., Gosselin, F., Buchanan, T. W., Tranel, D., Schyns, P., & Damasio, A. R. (2005). A mechanism for impaired fear recognition after amygdala damage. Nature, 433(7021), 68–72. [DOI:10.1038/nature03086] [PMID]
Akechi, H., Senju, A., Kikuchi, Y., Tojo, Y., Osanai, H., & Hasegawa, T. (2010). The effect of gaze direction on the processing of facial expressions in children with autism spectrum disorder: An ERP study. Neuropsychologia, 48(10), 2841–2851. [DOI:10.1016/j.neuropsychologia.2010.05.026] [PMID]
American Psychiatric Association (APA). (2013). Diagnostic and statistical manual of mental disorders: DSM-5TM, 5th ed. Arlington: American Psychiatric Publishing. [DOI:10.1176/appi.books.9780890425596]
Apicella, F., Sicca, F., Federico, R. R., Campatelli, G., & Muratori, F. (2013). Fusiform gyrus responses to neutral and emotional faces in children with autism spectrum disorders: A high density ERP study. Behavioural Brain Research, 251, 155–162. [DOI:10.1016/j.bbr.2012.10.040] [PMID]
Ashwin, C., Chapman, E., Colle, L., & Baron-Cohen, S. (2006). Impaired recognition of negative basic emotions in autism: A test of the amygdala theory. Social Neuroscience, 1(3-4), 349–363. [DOI:10.1080/17470910601040772] [PMID]
Baio, J. (2012). Prevalence of autism spectrum disorders—autism and developmental disabilities monitoring network, 14 sites, United States, 2008. Morbidity and Mortality Weekly Report (MMWR), 61(3), 1-19. [Link]
Baker, K. F., Montgomery, A. A., & Abramson, R. (2010). Brief report: perception and lateralization of spoken emotion by youths with high-functioning forms of autism. Journal of Autism and Developmental Disorders, 40(1), 123–129. [DOI:10.1007/s10803-009-0841-1] [PMID]
Balconi, M., & Pozzoli, U. (2008). Event-related oscillations (ERO) and event-related potentials (ERP) in emotional face recognition. The International Journal of Neuroscience, 118(10), 1412–1424. [DOI:10.1080/00207450601047119] [PMID]
Baron-Cohen, S., Cox, A., Baird, G., Swettenham, J., Nightingale, N., & Morgan, K., et al. (1996). Psychological markers in the detection of autism in infancy in a large population. The British Journal of Psychiatry, 168(2), 158–163. [DOI:10.1192/bjp.168.2.158] [PMID]
Baron-Cohen, S., Wheelwright, S., Hill, J., Raste, Y., & Plumb, I. (2001). The “reading the mind in the eyes” test revised version: A study with normal adults, and adults with Asperger syndrome or high-functioning autism. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 42(2), 241-251. [DOI:10.1111/1469-7610.00715]
Batty, M., Meaux, E., Wittemeyer, K., Rogé, B., & Taylor, M. J. (2011). Early processing of emotional faces in children with autism: An event-related potential study. Journal of Experimental Child Psychology, 109(4), 430–444. [DOI:10.1016/j.jecp.2011.02.001] [PMID]
Behrmann, M., Thomas, C., & Humphreys, K. (2006). Seeing it differently: Visual processing in autism. Trends in Cognitive Sciences, 10(6), 258–264. [DOI:10.1016/j.tics.2006.05.001] [PMID]
Ben-Ari Y. (2015). Is birth a critical period in the pathogenesis of autism spectrum disorders? Nature Reviews. Neuroscience, 16(8), 498–505. [DOI:10.1038/nrn3956] [PMID]
Blaesse, P., Airaksinen, M. S., Rivera, C., & Kaila, K. (2009). Cation-chloride cotransporters and neuronal function. Neuron, 61(6), 820–838. [DOI:10.1016/j.neuron.2009.03.003] [PMID]
Carretié, L., Martín-Loeches, M., Hinojosa, J. A., & Mercado, F. (2001). Emotion and attention interaction studied through event-related potentials. Journal of Cognitive Neuroscience, 13(8), 1109–1128. [DOI:10.1162/089892901753294400] [PMID]
Constantino, J. N., Lajonchere, C., Lutz, M., Gray, T., Abbacchi, A., & McKenna, K., et al. (2006). Autistic social impairment in the siblings of children with pervasive developmental disorders. The American Journal of Psychiatry, 163(2), 294–296. [DOI:10.1176/appi.ajp.163.2.294] [PMID]
Corden, B., Chilvers, R., & Skuse, D. (2008). Avoidance of emotionally arousing stimuli predicts social-perceptual impairment in Asperger's syndrome. Neuropsychologia, 46(1), 137–147. [DOI:10.1016/j.neuropsychologia.2007.08.005] [PMID]
Eftekhari, S., Shahrokhi, A., Tsintsadze, V., Nardou, R., Brouchoud, C., & Conesa, M., et al. (2014). Response to comment on "oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science, 346(6206), 176. [DOI:10.1126/science.1256009] [PMID]
Eimer M. (2000). Effects of face inversion on the structural encoding and recognition of faces. Evidence from event-related brain potentials. Brain research. Cognitive Brain Research, 10(1-2), 145–158. [DOI:10.1016/S0926-6410(00)00038-0] [PMID]
Eimer, M., & Holmes, A. (2007). Event-related brain potential correlates of emotional face processing. Neuropsychologia, 45(1), 15–31. [DOI:10.1016/j.neuropsychologia.2006.04.022] [PMID]
Faja, S., Dawson, G., Aylward, E., Wijsman, E. M., & Webb, S. J. (2016). Early event-related potentials to emotional faces differ for adults with autism spectrum disorder and by serotonin transporter genotype. Clinical Neurophysiology, 127(6), 2436–2447. [DOI:10.1016/j.clinph.2016.02.022] [PMID]
Feng, C., Wang, L., Wang, N., Gu, R., & Luo, Y. J. (2012). The time course of implicit processing of erotic pictures: An event-related potential study. Brain Research, 1489, 48–55. [DOI:10.1016/j.brainres.2012.10.019] [PMID]
Fung, L. K., Mahajan, R., Nozzolillo, A., Bernal, P., Krasner, A., Jo, B., & Coury, D., et al. (2016). Pharmacologic treatment of severe irritability and problem behaviors in autism: A systematic review and meta-analysis. Pediatrics, 137(Suppl 2), S124–S135. [DOI:10.1542/peds.2015-2851K] [PMID]
Hadjikhani, N., Åsberg Johnels, J., Lassalle, A., Zürcher, N. R., Hippolyte, L., & Gillberg, C., et al. (2018). Bumetanide for autism: More eye contact, less amygdala activation. Scientific Reports, 8(1), 3602. [DOI:10.1038/s41598-018-21958-x] [PMID]
Hadjikhani, N., Zürcher, N. R., Rogier, O., Ruest, T., Hippolyte, L., & Ben-Ari, Y., et al. (2015). Improving emotional face perception in autism with diuretic bumetanide: A proof-of-concept behavioral and functional brain imaging pilot study. Autism, 19(2), 149–157. [DOI:10.1177/1362361313514141] [PMID]
Harms, M. B., Martin, A., & Wallace, G. L. (2010). Facial emotion recognition in autism spectrum disorders: A review of behavioral and neuroimaging studies. Neuropsychology Review, 20(3), 290–322. [DOI:10.1007/s11065-010-9138-6] [PMID]
Haviland, J. M., & Lelwica, M. (1987). The induced affect response: 10-week-old infants' responses to three emotion expressions. Developmental Psychology, 23(1), 97–104. [DOI:/10.1037/0012-1649.23.1.97]
Herba, C., & Phillips, M. (2004). Annotation: Development of facial expression recognition from childhood to adolescence: Behavioural and neurological perspectives. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 45(7), 1185–1198. [DOI:10.1111/j.1469-7610.2004.00316.x] [PMID]
Jeste, S. S., & Nelson, C. A., 3rd (2009). Event related potentials in the understanding of autism spectrum disorders: An analytical review. Journal of Autism and Developmental Disorders, 39(3), 495–510. [DOI:10.1007/s10803-008-0652-9] [PMID]
Junghöfer, M., Bradley, M. M., Elbert, T. R., & Lang, P. J. (2001). Fleeting images: A new look at early emotion discrimination. Psychophysiology, 38(2), 175-178. [DOI:10.1111/1469-8986.3820175]
Kaland, N., Smith, L., & Mortensen, E. L. (2007). Response times of children and adolescents with Asperger syndrome on an 'advanced' test of theory of mind. Journal of Autism and Developmental Disorders, 37(2), 197–209. [DOI:10.1007/s10803-006-0152-8] [PMID]
Kaufman, J., Birmaher, B., Brent, D., Rao, U., Flynn, C., & Moreci, P., et al. (1997). Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): Initial reliability and validity data. Journal of the American Academy of Child and Adolescent Psychiatry, 36(7), 980–988. [DOI:10.1097/00004583-199707000-00021] [PMID]
Lang, P., Bradley, M.M., & Cuthbert, B.N. (1997). International affective picture system (IAPS): Technical manual and affective ratings. International Affective Picture System, 1-5. [Link]
Lemonnier, E., Degrez, C., Phelep, M., Tyzio, R., Josse, F., & Grandgeorge, M., et al. (2012). A randomised controlled trial of bumetanide in the treatment of autism in children. Translational Psychiatry, 2(12), e202. [DOI:10.1038/tp.2012.124] [PMID]
Lemonnier, E., Villeneuve, N., Sonie, S., Serret, S., Rosier, A., & Roue, M., et al. (2017). Effects of bumetanide on neurobehavioral function in children and adolescents with autism spectrum disorders. Translational Psychiatry, 7(3), e1056. [DOI:10.1038/tp.2017.10] [PMID]
Lemonnier, E., & Ben-Ari, Y. (2010). The diuretic bumetanide decreases autistic behaviour in five infants treated during 3 months with no side effects. Acta paediatrica, 99(12), 1885–1888. [DOI:10.1111/j.1651-2227.2010.01933.x] [PMID]
Lerner, M. D., McPartland, J. C., & Morris, J. P. (2013). Multimodal emotion processing in autism spectrum disorders: An event-related potential study. Developmental Cognitive Neuroscience, 3, 11–21. [DOI:10.1016/j.dcn.2012.08.005] [PMID]
Luo, W., Feng, W., He, W., Wang, N. Y., & Luo, Y. J. (2010). Three stages of facial expression processing: ERP study with rapid serial visual presentation. NeuroImage, 49(2), 1857–1867. [DOI:10.1016/j.neuroimage.2009.09.018] [PMID]
Makeig, S., & Enghof, S. (2000). Moving-window ica decomposition of eeg data reveals event-related changes in oscillatory brain activity. Paper presented in: Proceedings of the 2nd International Workshop on ICA and Blind Signal Separation, Helsinki, Finland, 22 June 2000. [Link]
McCracken, J. T., McGough, J., Shah, B., Cronin, P., Hong, D., & Aman, M. G., et al (2002). Risperidone in children with autism and serious behavioral problems. The New England Journal of Medicine, 347(5), 314–321. [DOI:10.1056/NEJMoa013171] [PMID]
Mollajani, R., Joghataei, M. T., & Tehrani-Doost, M. (2019). Bumetanide therapeutic effect in children and adolescents with autism spectrum disorder: A review study. Basic and Clinical Neuroscience, 10(5), 433–441. [DOI:10.32598/bcn.9.10.380] [PMID]
Monteiro, R., Simões, M., Andrade, J., & Castelo Branco, M. (2017). Processing of facial expressions in autism: A systematic review of EEG/ERP evidence. Review Journal of Autism and Developmental Disorders, 4, 255-276. [DOI:10.1007/s40489-017-0112-6]
Nardou, R., Yamamoto, S., Chazal, G., Bhar, A., Ferrand, N., & Dulac, O., et al. (2011). Neuronal chloride accumulation and excitatory GABA underlie aggravation of neonatal epileptiform activities by phenobarbital. Brain, 134(Pt 4), 987–1002. [DOI:10.1093/brain/awr041] [PMID]
O'Connor, K., Hamm, J. P., & Kirk, I. J. (2005). The neurophysiological correlates of face processing in adults and children with Asperger's syndrome. Brain and Cognition, 59(1), 82–95. [DOI:10.1016/j.bandc.2005.05.004] [PMID]
Philip, R. C., Whalley, H. C., Stanfield, A. C., Sprengelmeyer, R., Santos, I. M., & Young, A. W., et al. (2010). Deficits in facial, body movement and vocal emotional processing in autism spectrum disorders. Psychological Medicine, 40(11), 1919–1929. [DOI:10.1017/S0033291709992364] [PMID]
Rellecke, J., Sommer, W., & Schacht, A. (2012). Does processing of emotional facial expressions depend on intention? Time-resolved evidence from event-related brain potentials. Biological Psychology, 90(1), 23–32. [DOI:10.1016/j.biopsycho.2012.02.002] [PMID]
Rellecke, J., Sommer, W., & Schacht, A. (2013). Emotion effects on the n170: A question of reference? Brain Topography, 26(1), 62–71. [DOI:10.1007/s10548-012-0261-y] [PMID]
Rump, K. M., Giovannelli, J. L., Minshew, N. J., & Strauss, M. S. (2009). The development of emotion recognition in individuals with autism. Child Development, 80(5), 1434–1447. [DOI:10.1111/j.1467-8624.2009.01343.x] [PMID]
Schopler, E., Reichler, R. J., DeVellis, R. F., & Daly, K. (1980). Toward objective classification of childhood autism: Childhood Autism Rating Scale (CARS). Journal of Autism and Developmental Disorders, 10(1), 91–103. [DOI:10.1007/BF02408436] [PMID]
Schupp, H. T., Ohman, A., Junghöfer, M., Weike, A. I., Stockburger, J., & Hamm, A. O. (2004). The facilitated processing of threatening faces: An ERP analysis. Emotion, 4(2), 189–200. [DOI:10.1037/1528-3542.4.2.189] [PMID]
Shahrivar, Z., Kousha, M., Moallemi, S., Tehrani-Doost, M., & Alaghband-Rad, J. (2010). The reliability and validity of kiddie-schedule for affective disorders and schizophrenia-present and life-time version-Persian version. Child and adolescent Mental Health, 15(2), 97–102. [DOI:10.1111/j.1475-3588.2008.00518.x] [PMID]
Shahym, S. (1991). [The standardization of the wechsler intelligence scale for children (Persian)]. Journal of Social Sciences and Humanities, Shiraz University, 7-13-14. [Link]
Tehrani-Doost, M., Shahrivar, Z., Torabi, N., Ansari, S., Haji-Esmaeelzadeh, M., & Saeed-Ahmadi, S. (2020). Cross-cultural validation and normative data of the social responsiveness scale in a group of iranian general child population. Journal of Autism and Developmental Disorders, 50(7), 2389–2396. [DOI:10.1007/s10803-018-3773-9] [PMID]
Tyzio, R., Nardou, R., Ferrari, D. C., Tsintsadze, T., Shahrokhi, A., & Eftekhari, S., et al. (2014). Oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science, 343(6171), 675–679. [DOI:10.1126/science.1247190] [PMID]
Uljarevic, M., & Hamilton, A. (2013). Recognition of emotions in autism: a formal meta-analysis. Journal of Autism and Developmental Disorders, 43(7), 1517–1526. [DOI:10.1007/s10803-012-1695-5] [PMID]
Vlaskamp, C., Poil, S. S., Jansen, F., Linkenkaer-Hansen, K., Durston, S., & Oranje, B., et al. (2017). Bumetanide as a candidate treatment for behavioral problems in tuberous sclerosis complex. Frontiers in Neurology, 8, 469. [DOI:10.3389/fneur.2017.00469] [PMID]
Wallace, S., Coleman, M., & Bailey, A. (2008). An investigation of basic facial expression recognition in autism spectrum disorders. Cognition and Emotion, 22(7), 1353-1380. [DOI:10.1080/02699930701782153]
Wechsler, D. (1949). Wechsler intelligence scale for children. wechsler intelligence scale for children. San Antonio: Psychological Corporation. [Link]
Weiss, M. J., & Harris, S. L. (2001). Teaching social skills to people with autism. Behavior Modification, 25(5), 785–802. [DOI:10.1177/0145445501255007] [PMID]
Wink, L. K., Erickson, C. A., & McDougle, C. J. (2010). Pharmacologic treatment of behavioral symptoms associated with autism and other pervasive developmental disorders. Current treatment Options in Neurology, 12(6), 529–538. [DOI:10.1007/s11940-010-0091-8] [PMID]
Wong, T. K., Fung, P. C., Chua, S. E., & McAlonan, G. M. (2008). Abnormal spatiotemporal processing of emotional facial expressions in childhood autism: Dipole source analysis of event-related potentials. The European Journal of Neuroscience, 28(2), 407–416. [DOI:10.1111/j.1460-9568.2008.06328.x] [PMID]
Wynn, J. K., Lee, J., Horan, W. P., & Green, M. F. (2008). Using event related potentials to explore stages of facial affect recognition deficits in schizophrenia. Schizophrenia Bulletin, 34(4), 679–687. [DOI:10.1093/schbul/sbn047] [PMID]
Yirmiya, N., Sigman, M. D., Kasari, C., & Mundy, P. (1992). Empathy and cognition in high-functioning children with autism. Child Development, 63(1), 150–160. [DOI:10.2307/1130909] [PMID]
4. Discussion
Based on previous studies, bumetanide medication, which alters the GABAergic system from excitation to inhibition (Ben-Ari, 2015; Blaesse et al., 2009; Nardou et al., 2011), can reduce the core symptoms of ASD. One of the main outcomes of this disorder is the impairment of social interaction, which is caused by deficits in understanding others’ mental states, including their emotions through facial expressions (Baron-Cohen et al., 1996, Baron-Cohen et al., 2001; Kaland et al., 2007; Yirmiya et al., 1992). Moreover, based on Hadjikhani’s findings, bumetanide can improve emotion recognition and alter brain activation related to this function in people with ASD (Hadjikhani et al., 2018; Hadjikhani et al., 2015). According to these findings, we hypothesized that bumetanide could change neural responses based on action potentials related to facial emotion recognition in individuals with ASD. Therefore, we conducted the present study to evaluate the effect of bumetanide on facial emotion recognition and neural correlates of boys within the age range of 7-16 years old with ASD level 1 (needing support) according to DSM-5 classification. The evaluations included CARS, K-SADS-PL, WISC-R, SRS, ERP recording, and common blood tests. The CARS, SRS, and blood tests were repeated at weeks 6 and 12, and ERP recording was repeated 12 weeks after treatment. The participants’ blood tests had no significant changes during the intervention, and no obvious side effects were reported for medication.
Effects of bumetanide treatment on core symptoms of ASD
In this study, the total score of CARS showed a significant improvement in terms of clinical and general behavioral symptoms. These findings confirmed our hypothesis, indicating that bumetanide decreases the clinical and behavioral aspects of ASD and ameliorates its core symptoms. This finding is consistent with the results of Lemonnier and Ben-Ari (2010), Lemonnier et al. 2012, Lemonnier et al., 2013, and Lemonnier et al.. 2017), and Hadjikhani et al. (2015).
Effects of bumetanide on behavioral data of ERP recording
Regarding the response accuracy (RA) and based on the results, the main effect of emotion was significant on RA (without considering the effect of bumetanide) so that the happy faces were recognized more than the sad ones. This medication can improve the detection of facial emotions, especially happy ones, and confirm one of our hypotheses. This finding is consistent with the results of some studies that reported impaired detecting of negative emotions in individuals with ASD (Ashwin et al., 2006; Corden et al., 2008; Wallace et al., 2008). Considering feature-based processing in ASD (Behrmann, Thomas, & Humphreys, 2006), these individuals have more difficulty recognizing sadness than happiness, which can be recognized by the upturned mouth alone (Harms et al., 2010). It was interpreted that processing of the mouth region could be sufficient for detecting happiness compared to the other emotional states (Adolphs et al., 2005; Uljarevic & Hamilton, 2013).
It was found in this study that RA increased after treatment with bumetanide regardless of the type of emotion. This finding is consistent with the study results, which showed that bumetanide increased RA and reduced reaction time in an emotional matching task along with improving emotion recognition and increased activity of the brain regions involved in emotion processing (Hadjikhani et al., 2018; Hadjikhani et al. 2015; Vlaskamp, 2015).
Regarding the reaction time of all responses (RT_AR) and correct responses (RT_CR), according to our findings, the main effect of emotion was not significant among the emotional states in terms of the RT_AR and RT_CR. This finding can be interpreted so that bumetanide did not change the time spent recognizing emotions through faces. It was proposed that this duration would be increased after treatment because of the slow response time in recognizing emotions in individuals with ASD.
The main effects of treatment and the interaction between emotion and treatment were not significant regarding these variables, meaning that the intervention had no significant effect on the response time. This result may be due to the small sample size.
Effects of bumetanide on ERP components
Data analysis showed that the PA and PL of ERP components, including N170, EPN, and N250, significantly differ in some electrodes between pre- and post-treatment.
N170 component
Regarding the PA of N170, a significant increase was observed in the P7 electrode after treatment with bumetanide. It has been shown that individuals with ASD had smaller N170 amplitude compared to normal people (Lerner et al., 2013; O'Connor et al., 2005; Wong et al., 2008). Therefore, bumetanide seems to have led to more neural responses while recognizing sad expressions. This finding is in line with Hadjikhani's results when they studied brain activation in individuals with ASD after bumetanide treatment. They found increased brain activity in regions involved in processing emotional facial expressions, including the inferior occipital cortex and the fusiform cortex, equivalent to the P7 increased amplitude seen in our study (Hadjikhani et al., 2015).
The PL of N170 was not significant in any channels. It seems that bumetanide had no significant effect on the speed of neural response while watching facial emotions. The behavioral responses mentioned above are consistent with neural responses reflected in the ERPS's PL.
EPN components
Based on the result, the PA of EPN was significantly increased in Cp1 after treatment. This finding shows that bumetanide increased neural brain activity while recognizing emotions, increasing accuracy in detecting facial expressions.
The PL of this component was significantly decreased in the Cp6 channel just in neutral faces.
EPN is the emotion-sensitive early posterior negativity that arises over temporo-occipital electrodes about 150 to 300 ms after the stimulus onset. It is related to the visual cortex’s increased perceptual encoding of emotional stimuli (Rellecke et al., 2012). Studies on children with autism indicated a weak and delayed appearance of this component in these patients (Faja et al., 2016; Monteiro et al., 2017; Rellecke et al., 2013). Based on the current study, bumetanide can increase neural activity and cognitive effort in the temporo-occipital region while approaching emotions.
N250 component
The results showed that the PA of N250 significantly increased in the P3 channel while recognizing happy faces. It means that bumetanide can increase neural responses in individuals with ASD while detecting positive emotions, which leads to increased accuracy in detecting emotions.
Based on this study's results, bumetanide treatment significantly decreased the latency of N250 in P3, Pz, and Fc2 electrodes. Moreover, significant decreases were shown for happy and sad faces in the Fc2 channel based on the significant interaction between emotion and treatment in repeated measure analysis. This finding demonstrated that using bumetanide can increase the speed of neural response in youth with ASD while viewing happy and sad faces, which causes more rapid recognition of these emotions.
The N250 marks higher-order face processing, such as affect decoding. This ERP component reflects the earliest evaluation of emotion processing involved in facial emotion perception (Lerner et al., 2013). Other research studies showed that it is associated with decoding emotional concepts and is evoked by observing an emotionally expressive face (Balconi & Pozzoli, 2008; Carretié et al., 2001; Wynn et al., 2008). In the present study, the amplitude of this component increased in P3, especially in happy faces, while its latency significantly decreased for sad and happy faces in P3, Pz, and Fc2 after the intervention. It means that bumetanide can increase neural involvement and rapid response in reaction to emotional faces and recognizing the content of facial expressions. Based on previous studies, it has been found a decrease in amplitude and an increase in latency of this component in individuals with ASD (Jeste & Nelson, 2009; Lerner et al., 2013). Therefore, it seems that bumetanide can increase neural activity and its response speed in individuals with ASD while evaluating emotions.
5. Conclusion
To the best of our knowledge, this study was the first one to evaluate the effect of bumetanide on facial emotion recognition and its neural correlates in youth with ASD using ERP. The findings showed that using bumetanide improved the clinical and behavioral aspects of ASD according to the total score of the CARS. Furthermore, the results demonstrated an increase in the amplitude and a decrease in the latency of ERP components, including N170, EPN, and N250, in some channels while viewing happy and sad faces. Thus, it seems that using bumetanide can increase the ability of emotion recognition and alter the neural activity of involved brain areas. These results confirmed previous findings regarding the improvements in symptoms and brain function of individuals with ASD after bumetanide treatment.
The results of this study should be considered in light of some limitations, including the small sample size and the absence of a placebo control group. A multi-center, double-blind, placebo-controlled study with a larger sample size is recommended.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of the Iran University of Medical Sciences, Tehran, Iran (Code: IR.IUMS.REC.1395.4537), and a consent form was obtained from the participants’ parents before initiating the study.
Funding
The Cognitive Sciences and Technologies Council, Tehran, Iran, supported this study. The funder played no role in study design, data collection and analysis, decision for publication, or manuscript preparation.
Authors' contributions
All authors contributed equally to the conception and design of the study, data collection and analysis, interception of the results and drafting of the manuscript. Each author approved the final version of the manuscript for submission.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the Cognitive Sciences and Technologies Council, Tehran, Iran, for funding this research project.
References
Adolphs, R., Gosselin, F., Buchanan, T. W., Tranel, D., Schyns, P., & Damasio, A. R. (2005). A mechanism for impaired fear recognition after amygdala damage. Nature, 433(7021), 68–72. [DOI:10.1038/nature03086] [PMID]
Akechi, H., Senju, A., Kikuchi, Y., Tojo, Y., Osanai, H., & Hasegawa, T. (2010). The effect of gaze direction on the processing of facial expressions in children with autism spectrum disorder: An ERP study. Neuropsychologia, 48(10), 2841–2851. [DOI:10.1016/j.neuropsychologia.2010.05.026] [PMID]
American Psychiatric Association (APA). (2013). Diagnostic and statistical manual of mental disorders: DSM-5TM, 5th ed. Arlington: American Psychiatric Publishing. [DOI:10.1176/appi.books.9780890425596]
Apicella, F., Sicca, F., Federico, R. R., Campatelli, G., & Muratori, F. (2013). Fusiform gyrus responses to neutral and emotional faces in children with autism spectrum disorders: A high density ERP study. Behavioural Brain Research, 251, 155–162. [DOI:10.1016/j.bbr.2012.10.040] [PMID]
Ashwin, C., Chapman, E., Colle, L., & Baron-Cohen, S. (2006). Impaired recognition of negative basic emotions in autism: A test of the amygdala theory. Social Neuroscience, 1(3-4), 349–363. [DOI:10.1080/17470910601040772] [PMID]
Baio, J. (2012). Prevalence of autism spectrum disorders—autism and developmental disabilities monitoring network, 14 sites, United States, 2008. Morbidity and Mortality Weekly Report (MMWR), 61(3), 1-19. [Link]
Baker, K. F., Montgomery, A. A., & Abramson, R. (2010). Brief report: perception and lateralization of spoken emotion by youths with high-functioning forms of autism. Journal of Autism and Developmental Disorders, 40(1), 123–129. [DOI:10.1007/s10803-009-0841-1] [PMID]
Balconi, M., & Pozzoli, U. (2008). Event-related oscillations (ERO) and event-related potentials (ERP) in emotional face recognition. The International Journal of Neuroscience, 118(10), 1412–1424. [DOI:10.1080/00207450601047119] [PMID]
Baron-Cohen, S., Cox, A., Baird, G., Swettenham, J., Nightingale, N., & Morgan, K., et al. (1996). Psychological markers in the detection of autism in infancy in a large population. The British Journal of Psychiatry, 168(2), 158–163. [DOI:10.1192/bjp.168.2.158] [PMID]
Baron-Cohen, S., Wheelwright, S., Hill, J., Raste, Y., & Plumb, I. (2001). The “reading the mind in the eyes” test revised version: A study with normal adults, and adults with Asperger syndrome or high-functioning autism. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 42(2), 241-251. [DOI:10.1111/1469-7610.00715]
Batty, M., Meaux, E., Wittemeyer, K., Rogé, B., & Taylor, M. J. (2011). Early processing of emotional faces in children with autism: An event-related potential study. Journal of Experimental Child Psychology, 109(4), 430–444. [DOI:10.1016/j.jecp.2011.02.001] [PMID]
Behrmann, M., Thomas, C., & Humphreys, K. (2006). Seeing it differently: Visual processing in autism. Trends in Cognitive Sciences, 10(6), 258–264. [DOI:10.1016/j.tics.2006.05.001] [PMID]
Ben-Ari Y. (2015). Is birth a critical period in the pathogenesis of autism spectrum disorders? Nature Reviews. Neuroscience, 16(8), 498–505. [DOI:10.1038/nrn3956] [PMID]
Blaesse, P., Airaksinen, M. S., Rivera, C., & Kaila, K. (2009). Cation-chloride cotransporters and neuronal function. Neuron, 61(6), 820–838. [DOI:10.1016/j.neuron.2009.03.003] [PMID]
Carretié, L., Martín-Loeches, M., Hinojosa, J. A., & Mercado, F. (2001). Emotion and attention interaction studied through event-related potentials. Journal of Cognitive Neuroscience, 13(8), 1109–1128. [DOI:10.1162/089892901753294400] [PMID]
Constantino, J. N., Lajonchere, C., Lutz, M., Gray, T., Abbacchi, A., & McKenna, K., et al. (2006). Autistic social impairment in the siblings of children with pervasive developmental disorders. The American Journal of Psychiatry, 163(2), 294–296. [DOI:10.1176/appi.ajp.163.2.294] [PMID]
Corden, B., Chilvers, R., & Skuse, D. (2008). Avoidance of emotionally arousing stimuli predicts social-perceptual impairment in Asperger's syndrome. Neuropsychologia, 46(1), 137–147. [DOI:10.1016/j.neuropsychologia.2007.08.005] [PMID]
Eftekhari, S., Shahrokhi, A., Tsintsadze, V., Nardou, R., Brouchoud, C., & Conesa, M., et al. (2014). Response to comment on "oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science, 346(6206), 176. [DOI:10.1126/science.1256009] [PMID]
Eimer M. (2000). Effects of face inversion on the structural encoding and recognition of faces. Evidence from event-related brain potentials. Brain research. Cognitive Brain Research, 10(1-2), 145–158. [DOI:10.1016/S0926-6410(00)00038-0] [PMID]
Eimer, M., & Holmes, A. (2007). Event-related brain potential correlates of emotional face processing. Neuropsychologia, 45(1), 15–31. [DOI:10.1016/j.neuropsychologia.2006.04.022] [PMID]
Faja, S., Dawson, G., Aylward, E., Wijsman, E. M., & Webb, S. J. (2016). Early event-related potentials to emotional faces differ for adults with autism spectrum disorder and by serotonin transporter genotype. Clinical Neurophysiology, 127(6), 2436–2447. [DOI:10.1016/j.clinph.2016.02.022] [PMID]
Feng, C., Wang, L., Wang, N., Gu, R., & Luo, Y. J. (2012). The time course of implicit processing of erotic pictures: An event-related potential study. Brain Research, 1489, 48–55. [DOI:10.1016/j.brainres.2012.10.019] [PMID]
Fung, L. K., Mahajan, R., Nozzolillo, A., Bernal, P., Krasner, A., Jo, B., & Coury, D., et al. (2016). Pharmacologic treatment of severe irritability and problem behaviors in autism: A systematic review and meta-analysis. Pediatrics, 137(Suppl 2), S124–S135. [DOI:10.1542/peds.2015-2851K] [PMID]
Hadjikhani, N., Åsberg Johnels, J., Lassalle, A., Zürcher, N. R., Hippolyte, L., & Gillberg, C., et al. (2018). Bumetanide for autism: More eye contact, less amygdala activation. Scientific Reports, 8(1), 3602. [DOI:10.1038/s41598-018-21958-x] [PMID]
Hadjikhani, N., Zürcher, N. R., Rogier, O., Ruest, T., Hippolyte, L., & Ben-Ari, Y., et al. (2015). Improving emotional face perception in autism with diuretic bumetanide: A proof-of-concept behavioral and functional brain imaging pilot study. Autism, 19(2), 149–157. [DOI:10.1177/1362361313514141] [PMID]
Harms, M. B., Martin, A., & Wallace, G. L. (2010). Facial emotion recognition in autism spectrum disorders: A review of behavioral and neuroimaging studies. Neuropsychology Review, 20(3), 290–322. [DOI:10.1007/s11065-010-9138-6] [PMID]
Haviland, J. M., & Lelwica, M. (1987). The induced affect response: 10-week-old infants' responses to three emotion expressions. Developmental Psychology, 23(1), 97–104. [DOI:/10.1037/0012-1649.23.1.97]
Herba, C., & Phillips, M. (2004). Annotation: Development of facial expression recognition from childhood to adolescence: Behavioural and neurological perspectives. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 45(7), 1185–1198. [DOI:10.1111/j.1469-7610.2004.00316.x] [PMID]
Jeste, S. S., & Nelson, C. A., 3rd (2009). Event related potentials in the understanding of autism spectrum disorders: An analytical review. Journal of Autism and Developmental Disorders, 39(3), 495–510. [DOI:10.1007/s10803-008-0652-9] [PMID]
Junghöfer, M., Bradley, M. M., Elbert, T. R., & Lang, P. J. (2001). Fleeting images: A new look at early emotion discrimination. Psychophysiology, 38(2), 175-178. [DOI:10.1111/1469-8986.3820175]
Kaland, N., Smith, L., & Mortensen, E. L. (2007). Response times of children and adolescents with Asperger syndrome on an 'advanced' test of theory of mind. Journal of Autism and Developmental Disorders, 37(2), 197–209. [DOI:10.1007/s10803-006-0152-8] [PMID]
Kaufman, J., Birmaher, B., Brent, D., Rao, U., Flynn, C., & Moreci, P., et al. (1997). Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): Initial reliability and validity data. Journal of the American Academy of Child and Adolescent Psychiatry, 36(7), 980–988. [DOI:10.1097/00004583-199707000-00021] [PMID]
Lang, P., Bradley, M.M., & Cuthbert, B.N. (1997). International affective picture system (IAPS): Technical manual and affective ratings. International Affective Picture System, 1-5. [Link]
Lemonnier, E., Degrez, C., Phelep, M., Tyzio, R., Josse, F., & Grandgeorge, M., et al. (2012). A randomised controlled trial of bumetanide in the treatment of autism in children. Translational Psychiatry, 2(12), e202. [DOI:10.1038/tp.2012.124] [PMID]
Lemonnier, E., Villeneuve, N., Sonie, S., Serret, S., Rosier, A., & Roue, M., et al. (2017). Effects of bumetanide on neurobehavioral function in children and adolescents with autism spectrum disorders. Translational Psychiatry, 7(3), e1056. [DOI:10.1038/tp.2017.10] [PMID]
Lemonnier, E., & Ben-Ari, Y. (2010). The diuretic bumetanide decreases autistic behaviour in five infants treated during 3 months with no side effects. Acta paediatrica, 99(12), 1885–1888. [DOI:10.1111/j.1651-2227.2010.01933.x] [PMID]
Lerner, M. D., McPartland, J. C., & Morris, J. P. (2013). Multimodal emotion processing in autism spectrum disorders: An event-related potential study. Developmental Cognitive Neuroscience, 3, 11–21. [DOI:10.1016/j.dcn.2012.08.005] [PMID]
Luo, W., Feng, W., He, W., Wang, N. Y., & Luo, Y. J. (2010). Three stages of facial expression processing: ERP study with rapid serial visual presentation. NeuroImage, 49(2), 1857–1867. [DOI:10.1016/j.neuroimage.2009.09.018] [PMID]
Makeig, S., & Enghof, S. (2000). Moving-window ica decomposition of eeg data reveals event-related changes in oscillatory brain activity. Paper presented in: Proceedings of the 2nd International Workshop on ICA and Blind Signal Separation, Helsinki, Finland, 22 June 2000. [Link]
McCracken, J. T., McGough, J., Shah, B., Cronin, P., Hong, D., & Aman, M. G., et al (2002). Risperidone in children with autism and serious behavioral problems. The New England Journal of Medicine, 347(5), 314–321. [DOI:10.1056/NEJMoa013171] [PMID]
Mollajani, R., Joghataei, M. T., & Tehrani-Doost, M. (2019). Bumetanide therapeutic effect in children and adolescents with autism spectrum disorder: A review study. Basic and Clinical Neuroscience, 10(5), 433–441. [DOI:10.32598/bcn.9.10.380] [PMID]
Monteiro, R., Simões, M., Andrade, J., & Castelo Branco, M. (2017). Processing of facial expressions in autism: A systematic review of EEG/ERP evidence. Review Journal of Autism and Developmental Disorders, 4, 255-276. [DOI:10.1007/s40489-017-0112-6]
Nardou, R., Yamamoto, S., Chazal, G., Bhar, A., Ferrand, N., & Dulac, O., et al. (2011). Neuronal chloride accumulation and excitatory GABA underlie aggravation of neonatal epileptiform activities by phenobarbital. Brain, 134(Pt 4), 987–1002. [DOI:10.1093/brain/awr041] [PMID]
O'Connor, K., Hamm, J. P., & Kirk, I. J. (2005). The neurophysiological correlates of face processing in adults and children with Asperger's syndrome. Brain and Cognition, 59(1), 82–95. [DOI:10.1016/j.bandc.2005.05.004] [PMID]
Philip, R. C., Whalley, H. C., Stanfield, A. C., Sprengelmeyer, R., Santos, I. M., & Young, A. W., et al. (2010). Deficits in facial, body movement and vocal emotional processing in autism spectrum disorders. Psychological Medicine, 40(11), 1919–1929. [DOI:10.1017/S0033291709992364] [PMID]
Rellecke, J., Sommer, W., & Schacht, A. (2012). Does processing of emotional facial expressions depend on intention? Time-resolved evidence from event-related brain potentials. Biological Psychology, 90(1), 23–32. [DOI:10.1016/j.biopsycho.2012.02.002] [PMID]
Rellecke, J., Sommer, W., & Schacht, A. (2013). Emotion effects on the n170: A question of reference? Brain Topography, 26(1), 62–71. [DOI:10.1007/s10548-012-0261-y] [PMID]
Rump, K. M., Giovannelli, J. L., Minshew, N. J., & Strauss, M. S. (2009). The development of emotion recognition in individuals with autism. Child Development, 80(5), 1434–1447. [DOI:10.1111/j.1467-8624.2009.01343.x] [PMID]
Schopler, E., Reichler, R. J., DeVellis, R. F., & Daly, K. (1980). Toward objective classification of childhood autism: Childhood Autism Rating Scale (CARS). Journal of Autism and Developmental Disorders, 10(1), 91–103. [DOI:10.1007/BF02408436] [PMID]
Schupp, H. T., Ohman, A., Junghöfer, M., Weike, A. I., Stockburger, J., & Hamm, A. O. (2004). The facilitated processing of threatening faces: An ERP analysis. Emotion, 4(2), 189–200. [DOI:10.1037/1528-3542.4.2.189] [PMID]
Shahrivar, Z., Kousha, M., Moallemi, S., Tehrani-Doost, M., & Alaghband-Rad, J. (2010). The reliability and validity of kiddie-schedule for affective disorders and schizophrenia-present and life-time version-Persian version. Child and adolescent Mental Health, 15(2), 97–102. [DOI:10.1111/j.1475-3588.2008.00518.x] [PMID]
Shahym, S. (1991). [The standardization of the wechsler intelligence scale for children (Persian)]. Journal of Social Sciences and Humanities, Shiraz University, 7-13-14. [Link]
Tehrani-Doost, M., Shahrivar, Z., Torabi, N., Ansari, S., Haji-Esmaeelzadeh, M., & Saeed-Ahmadi, S. (2020). Cross-cultural validation and normative data of the social responsiveness scale in a group of iranian general child population. Journal of Autism and Developmental Disorders, 50(7), 2389–2396. [DOI:10.1007/s10803-018-3773-9] [PMID]
Tyzio, R., Nardou, R., Ferrari, D. C., Tsintsadze, T., Shahrokhi, A., & Eftekhari, S., et al. (2014). Oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science, 343(6171), 675–679. [DOI:10.1126/science.1247190] [PMID]
Uljarevic, M., & Hamilton, A. (2013). Recognition of emotions in autism: a formal meta-analysis. Journal of Autism and Developmental Disorders, 43(7), 1517–1526. [DOI:10.1007/s10803-012-1695-5] [PMID]
Vlaskamp, C., Poil, S. S., Jansen, F., Linkenkaer-Hansen, K., Durston, S., & Oranje, B., et al. (2017). Bumetanide as a candidate treatment for behavioral problems in tuberous sclerosis complex. Frontiers in Neurology, 8, 469. [DOI:10.3389/fneur.2017.00469] [PMID]
Wallace, S., Coleman, M., & Bailey, A. (2008). An investigation of basic facial expression recognition in autism spectrum disorders. Cognition and Emotion, 22(7), 1353-1380. [DOI:10.1080/02699930701782153]
Wechsler, D. (1949). Wechsler intelligence scale for children. wechsler intelligence scale for children. San Antonio: Psychological Corporation. [Link]
Weiss, M. J., & Harris, S. L. (2001). Teaching social skills to people with autism. Behavior Modification, 25(5), 785–802. [DOI:10.1177/0145445501255007] [PMID]
Wink, L. K., Erickson, C. A., & McDougle, C. J. (2010). Pharmacologic treatment of behavioral symptoms associated with autism and other pervasive developmental disorders. Current treatment Options in Neurology, 12(6), 529–538. [DOI:10.1007/s11940-010-0091-8] [PMID]
Wong, T. K., Fung, P. C., Chua, S. E., & McAlonan, G. M. (2008). Abnormal spatiotemporal processing of emotional facial expressions in childhood autism: Dipole source analysis of event-related potentials. The European Journal of Neuroscience, 28(2), 407–416. [DOI:10.1111/j.1460-9568.2008.06328.x] [PMID]
Wynn, J. K., Lee, J., Horan, W. P., & Green, M. F. (2008). Using event related potentials to explore stages of facial affect recognition deficits in schizophrenia. Schizophrenia Bulletin, 34(4), 679–687. [DOI:10.1093/schbul/sbn047] [PMID]
Yirmiya, N., Sigman, M. D., Kasari, C., & Mundy, P. (1992). Empathy and cognition in high-functioning children with autism. Child Development, 63(1), 150–160. [DOI:10.2307/1130909] [PMID]
Type of Study: Original |
Subject:
Clinical Neuroscience
Received: 2022/02/6 | Accepted: 2023/05/22 | Published: 2025/01/1
Received: 2022/02/6 | Accepted: 2023/05/22 | Published: 2025/01/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |