Volume 13, Issue 6 (November & December 2022)
BCN 2022, 13(6): 883-892 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Rostami M, Faridi F, Khosrowabadi R. Brain Functional Correlates of Intelligence Score in ADHD Based on EEG. BCN 2022; 13 (6) :883-892
URL: http://bcn.iums.ac.ir/article-1-1726-en.html
URL: http://bcn.iums.ac.ir/article-1-1726-en.html
1- Department of Psychology, Faculty of Psychology and Educational Sciences, Allameh Tabatabaie University, Tehran, Iran.
2- Institut for Cognitive and Brain Sciences, Shahid Beheshti University, Tehran, Iran.
3- Institute for Cognitive and Brain Sciences, Shahid Beheshti University, Tehran, Iran.
2- Institut for Cognitive and Brain Sciences, Shahid Beheshti University, Tehran, Iran.
3- Institute for Cognitive and Brain Sciences, Shahid Beheshti University, Tehran, Iran.
Full-Text [PDF 805 kb]
| Abstract (HTML)
Full-Text:
1. Introduction
Intelligence is a general mental ability, which includes planning, reasoning, comprehension, abstraction, and learning (Gottfredson, 1997). Therefore, intelligence scores may be used to predict one’s performance (Haier & Jung, 2008). This cognitive ability is produced by brain structure and functions, which could be traced by neuroimaging techniques. In this regard, neuroimaging studies have mainly presented a region-specific pattern of anatomy as well as activities associated with the intelligence score. In terms of structure, with the advent of magnetic resonance imaging (MRI) studies, researchers have found a strong association between intelligence and grey matter density (Narr et al., 2006). For example, an association between intelligence and grey matter density has been reported in the basal ganglia (Dietrich, 2004), hippocampus (Deary et al., 2010), fusiform (Deary et al., 2010), posterior region (Heilman et al., 2003), Parietal and frontal (Thatcher et al., 2008; Thatcher et al., 2007) and dorsolateral prefrontal cortex (DLPFC) (Cabeza & Nyberg, 2000). Other studies associated global properties of the structural brain network with general intelligence (Fischer et al., 2014). Similar to structural studies, functional studies also suggest a network for intelligence (Jung & Haier, 2007). In this context, about the relationship between intelligence and the function of the brain, a variety of psychophysiological measurement methods ranging from positron emission tomography (PET) (Kane & Engle, 2002; Gray et al., 2003), functional magnetic resonance imaging (fMRI) (Geake & Hansen, 2005; Burgess et al., 2011; Tang et al., 2010) and electroencephalography (EEG) (Neubauer et al., 2005; Anokhin et al., 1999) has been employed. Of the all above-mentioned methods, EEG is one of the most popular used method because it is cheap, portable, and easy to wear, and has a high temporal resolution (Reis et al., 2014) and it is proper to use in the applied studies. EEG presented a correlation with intelligence (Mundy‐Castle, 1958). Recent studies have demonstrated that highly intelligent individuals allocate fewer resources and recruit minimum task-relevant brain areas when performing an easy task, while they allocate more resources and recruit several brain areas when the task demand is high. In contrast, low intelligent individuals do not use different resource allocation strategies during easy and hard tasks (Kang & Lee, 2015).
In general, the association of IQ scores and EEG properties have been studied using the power or amplitude (Klimesch, 1999b; Giannitrapani & Liberson, 1985) (Martín‐Loeches et al., 2001), and the functional connectivity networks extracted with coherence and phase delays (Thatcher et al., 2005; Thatcher et al., 2007). Moreover, the brain works in a frequency-specific manner; therefore, the association between IQ scores and EEG parameters has also been investigated in the conventional frequency bands as well. The conventional frequency bands in EEG include delta (slowest), theta, alpha, beta, and gamma (fastest) bands. These frequency bands have a critical role for performing the cognitive tasks (Klimesch et al., 1993; Moretti, 2015). In addition, cognitive disabilities have also been related to changes in the neural oscillatory pattern (frequency-specific activities). For instance, an increase in task demand increases the theta power and causes more synchronization in this band. In contrast, an increase in a task demand decreases the alpha power and causes desynchronization in this band (Schacter, 1977) (Webster, 1978) (Marosi et al., 1999) (Fogel et al., 2007). EEG waves are caused by large numbers of neurons firing in synchrony. Synchronization behavior of EEG signals is important for decoding information processing in human brain.
In terms of the IQ index of EEG, it has been shown that alpha power is the most closely correlated with IQ score (Stankova & Myshkin, 2016). In addition, high-IQ individuals present higher modularity in their brain functional connectivity network (FCN) and have different patterns of activations between short-range and long-range connections in the FCN (Hearn et al., 2016) (Song et al., 2008). The above-mentioned studies have mainly focused on normal individuals.
Neurodevelopmental disorders, such as attention deficit and hyperactive disorder (ADHD) could influence the pattern of brain activities. For instance, studies have reported greater frontocentral theta activity, lower beta activity, and higher theta/beta ratio in ADHD subjects compared to healthy controls (Clarke et al., 2001b; Loo et al., 2010) (Bresnahan & Barry, 2002) (Arns et al., 2013) (Loo et al., 2010). However, there is inconsistency in the previous studies on the effect of IQ on EEG power in ADHD. For instance, Clark et al. reported that IQ does not affect the EEG power in ADHD individuals compared to their matched control subjects (Clarke et al., 2006) While Chabot and Serfontein (1996) showed that there is a different oscillatory pattern in EEG of ADHD children with high and low IQ. They suggested that children with low IQs show lower alpha and beta bands (Chabot & Serfontein, 1996). Other studies have also highlighted the importance of IQ on any potential group’s differences in ADHD with high and low IQ (Kitsune et al., 2015). Therefore, it seems that studies should try to control the effects of IQ on their data. In this study, we hypothesized that neural correlates of intelligence should not have a fixed pattern rather it must follow a dynamic pattern to compensate for the deficits caused by the disorder.
Therefore, in this study, EEG was recorded from three ADHD subtypes and a control group in eye closed condition. Then, the correlation of EEG spectral bands using estimates of absolute and ratio with IQ and significant differences within groups were computed at a specific brain region. We also estimated the whole pattern of association at each absolute frequency band. Details of the experimental design and data analysis are explained in the next section.
2. Materials and Methods
A schematic of the experimental design is presented in Figure 1.

Subjects
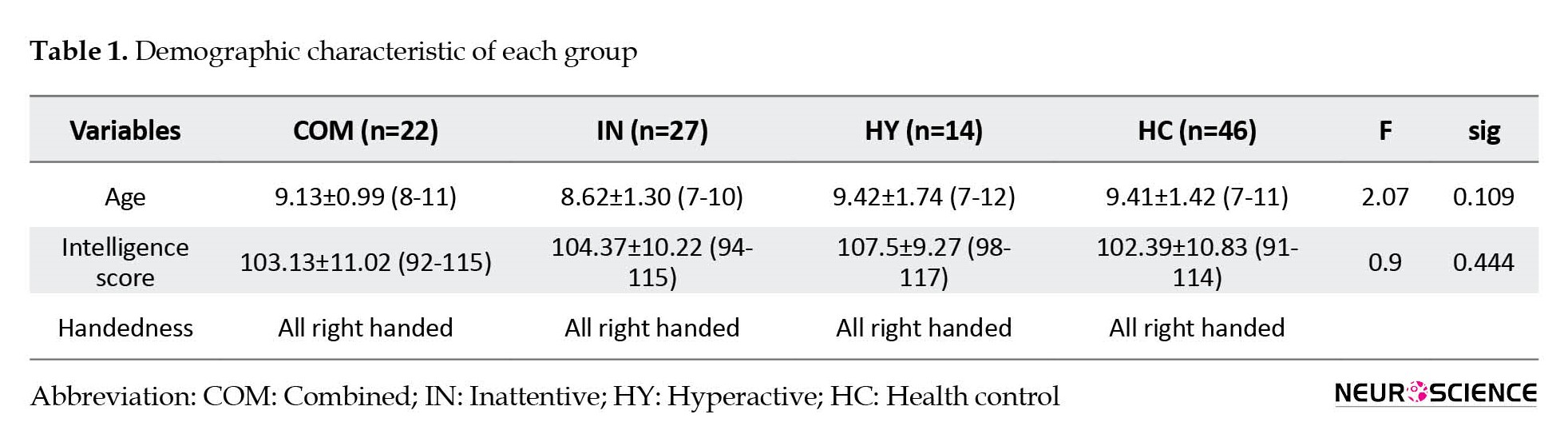
One hundred nine boy participants comprising 22 combined (age=9.13±0.99, IQ=103.13±11.02), 27 inattentive (age=8.62±1.30, IQ=104.37±10.22), 14 hyperactive (age=9.42±1.74, IQ=107.5±9.27), and 46 healthy controls (age=9.41±1.42, IQ= 102.39±10.83) were recruited in this study. All subjects were right-handed and there were no significant differences in age and IQ between the groups (Table 1). The diagnostic assessment was performed by a psychiatrist and a senior clinical psychologist using the Persian version of the Structural Clinical Interview for DSM-V (SCID). Average DSM-V symptom scores of inattentive, hyperactive, and combined were 7.0±1.3, 8.2±2.9, and 6.3±1.3, respectively. Children with brain damage, neurological disorder, and epilepsy as well as children who consume stimulants or were under neurotherapy were excluded. The research protocol was approved by the Iran University of Medical Science Ethics Committee and all parents signed informed consent.
IQ scores
All participants completed Ravan’s standard progressive matrices (SPM) test. This multiple-choice test is used to assess abstract reasoning and nonverbal ability (Raven, 2000b). It has also been considered to be the best measure of spearman’s g factor (Reynolds & Brown, 1984) and intelligence (Raven, 2000a).
EEG data recording and analysis
EEG data were recorded during an eyes-closed resting state for 4 minutes while children were seated on a comfortable chair. All subjects were asked to sit relax and try not to blink. The EEG data were registered using a 19-electrode Mitsar amplifier (www.mitsar-medical.com) with a sampling rate of 250 Hz. Electrodes were placed on the scalp using a standard 10-20 montage (Fp1, Fp2, Fz, F3, F4, F7, F8, Cz, C3, C4, T3, T4, T5, T6, Pz, P3, P4, O1, and O2) with the average of ear channels used as reference and FPz as the ground electrode. Electrode impedances were kept below 5 kΩ.
After recording, a self-written program using Matlab (https://www.mathworks.com) and EEGlab functions (https://sccn.ucsd.edu/eeglab/) was used to process the data. Standard pre-processing included band-pass filtering (1-40 Hz), segmenting the data into epochs of 1-second duration, automatic rejection of disturbing channels using probability (artifact rejection was carried out using both to single electrodes and the collection of all electrodes), spectrum (thresholds are expressed in terms of amplitude changes relative to baseline in dB), and kurtosis (technically called the four first cumulates of the distribution) of the signal interpolation of rejected channels by averaging its spherical neighbors, removing unreliable epochs, and referencing to the average. Subsequently, the pre-processed EEG data were transformed to the frequency domain using Fast Fourier Transform (FFT), and the absolute power of the data was then calculated in Delta (1-4 Hz), Theta (4-8 Hz), Alpha (8-13 Hz), Alpha1 (8-10), Alpha2 (10-12), and Beta (13-30Hz) frequency bands. After that, the power ratio was estimated for the theta/beta and theta/alpha frequency bands.
Statistical analysis
Pearson’s correlation was calculated using the score of SPM and the spectra of each frequency band. Spectral power was calculated in absolute and ratio style from the cleaned data. Then p- the values of each correlation score were considered and significant associations were recognized by putting a threshold of 0.05 for the p-values. This analysis was performed using the statistical analysis toolbox of MATLAB 2015 (www.mathworks.com). Then, the group comparison of correlation values was performed using the cocor package (Diedenhofen & Musch, 2015).
3. Results
Detailed information on significant results (P<0.05) for absolute and ratio power are presented in Tables 2-7 and Figures 2-6, and the whole pattern analysis is shown in Table 8 and Figure 7.
Correlation maps were scaled between -1 and 1. Only significant correlations (P<0.05) were plotted. The hot color indicates a positive and cold color shows a negative correlation.

Association between EEG power spectrum and IQ score
Absolute power
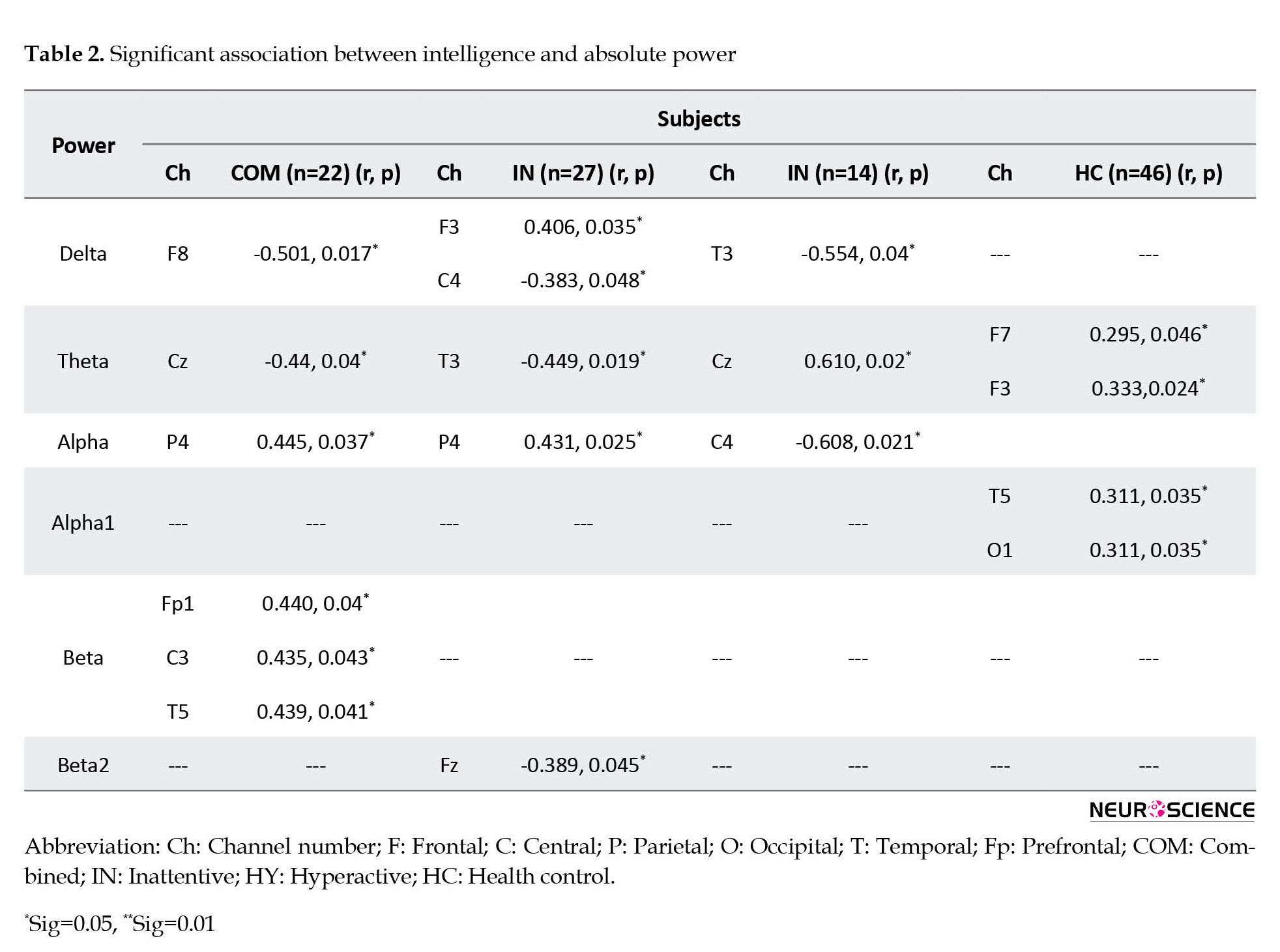
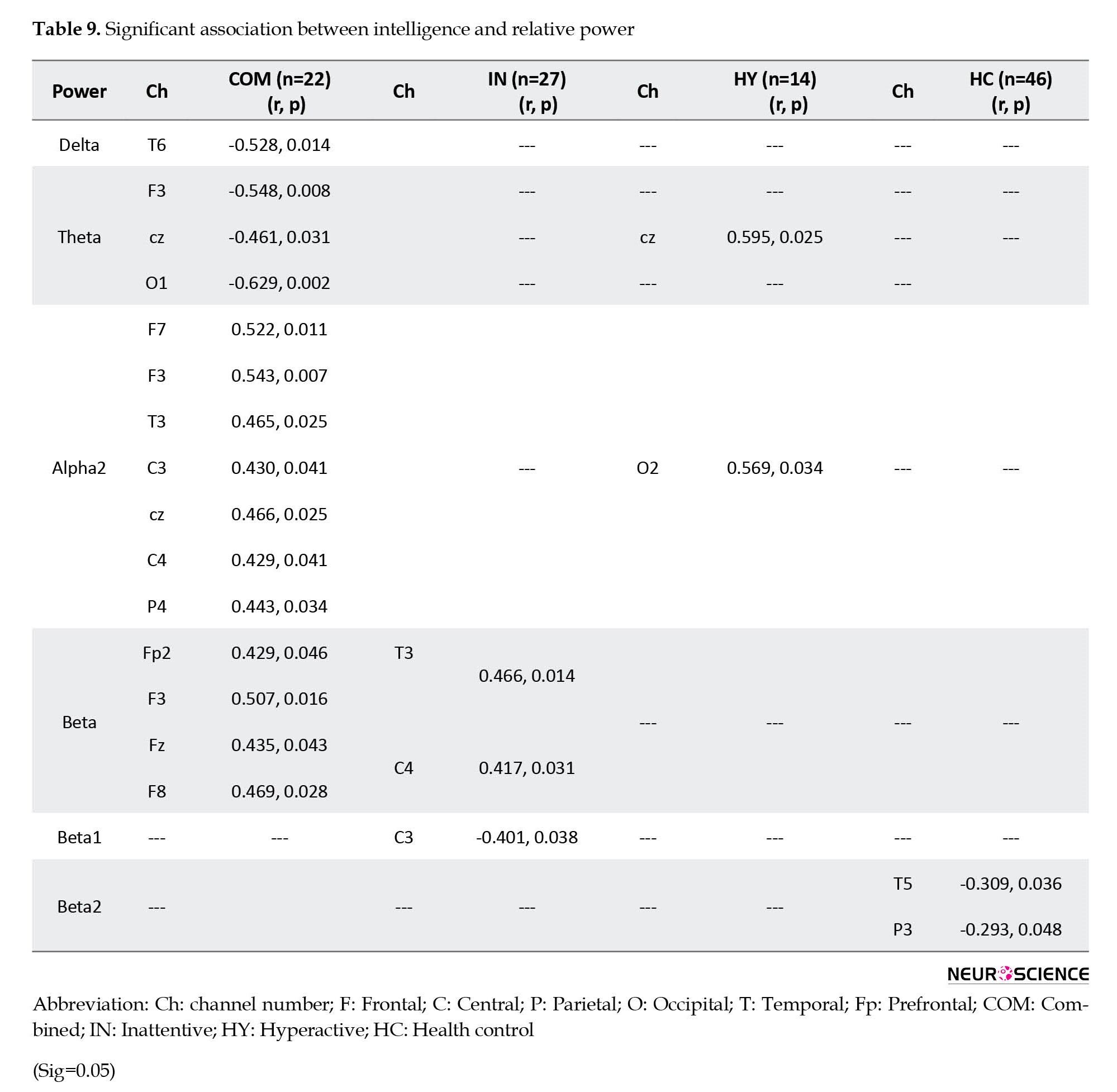
Figure 2 and Table 2 present the significant correlation between absolute power and intelligence in ADHD subtypes and healthy control groups. the combined ADHD group showed significant positive correlations between IQ scores mainly at P4 in the alpha band, and at Fp1, C3, and T5 in the beta band. Negative correlations in this group were also found between IQ score and delta power at F8, and theta power at Cz. The inattentive ADHD group showed a significant positive correlation between IQ and delta power at F3 and alpha power at P4. A negative correlation in this group was also observed between IQ score and delta power at C4, and theta power at T3. ADHD-hyperactive/impulsive group showed a positive correlation between IQ score and theta power at Cz and a negative correlation between IQ score and delta power at T3, and alpha power at C4. The healthy control group did not show any negative correlation between IQ and EEG bands, while positive correlations were observed between IQ score and theta power at F3 and F7, and apha1 power at T5 and O1.
The association between ratio power (theta/alpha, theta/beta) and intelligence score is presented in Table 3 and Figure 3.
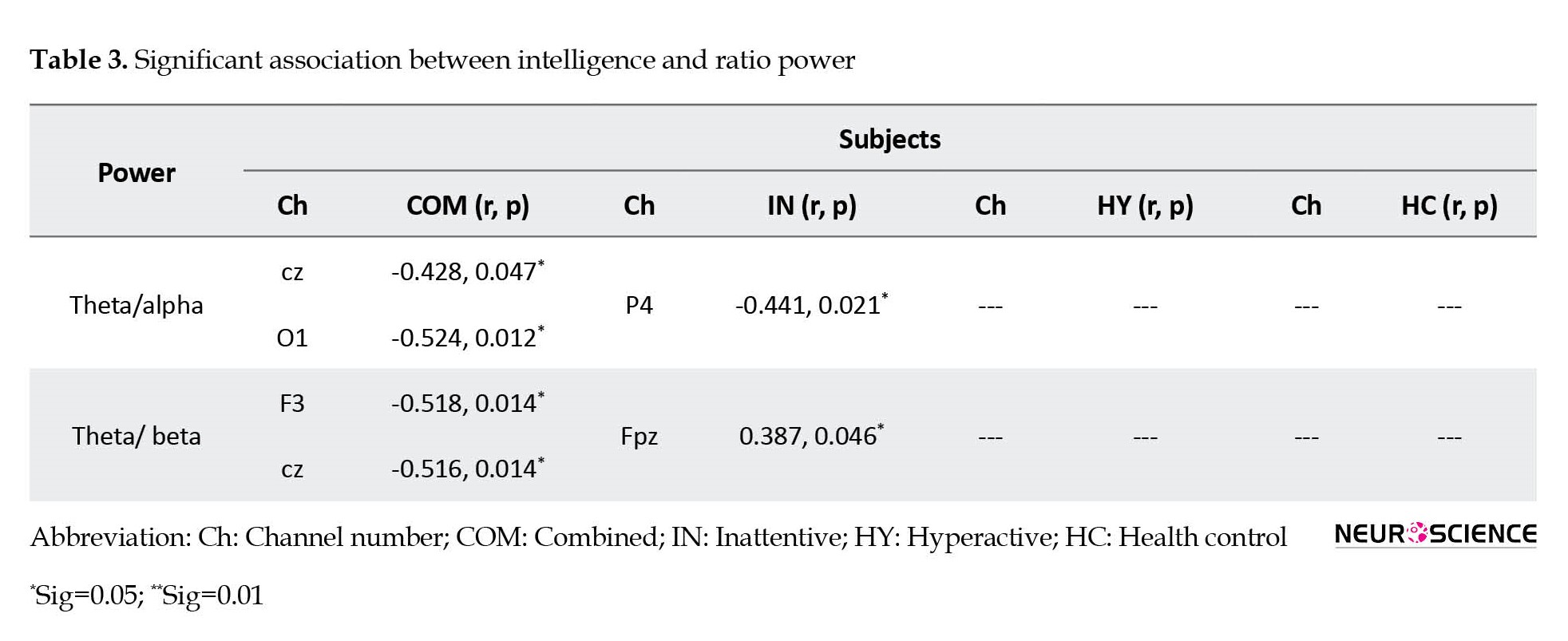
The ADHD-combined group showed negative correlations between IQ score and theta/alpha at Cz, and O2 as well as theta/beta at F3, and Cz areas. The inattentive ADHD group revealed a positive correlation between IQ score and theta/beta at Fp2, and a negative correlation between IQ score and theta/alpha at P4. The hyperactive ADHD group and the healthy control group did not show any significant correlation between IQ score and the above-mentioned power ratios.

Differential patterns of the association between IQ scores and EEG power spectrum in ADHD subtypes and healthy control groups
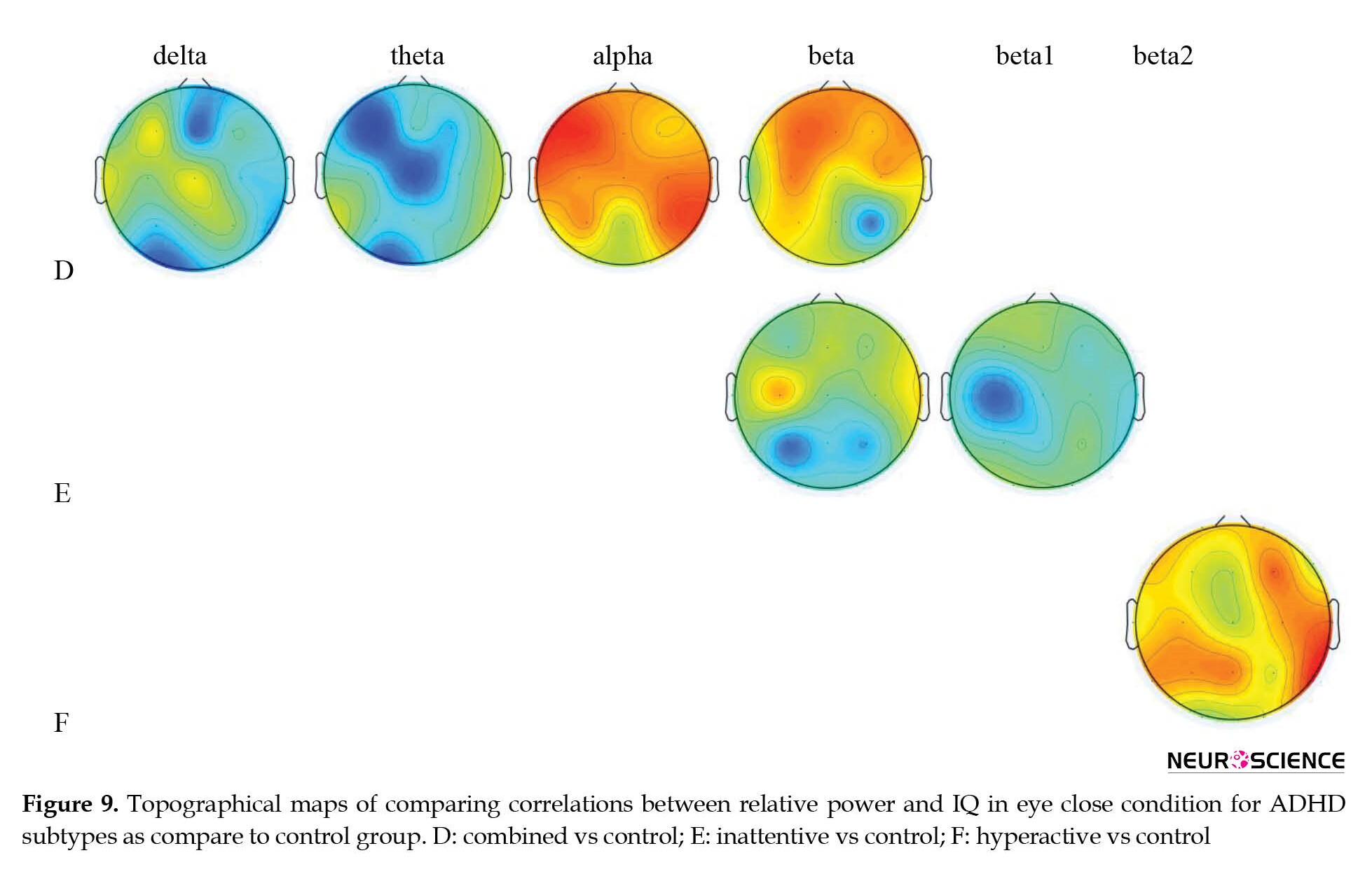
Topographical differences in the association between IQ score and absolute power in various frequency bands in ADHD subtypes were compared with the healthy control group (Table 4 and Figure 4).

Compared to the healthy control group, ADHD-combined cases showed increased alpha power at P3 and decreased delta at F8 and Fz, and theta at F3 and Cz, and alpha1 power at F3, C4, T4 locations. ADHD-inattentive cases revealed increased theta power at F8 and T3, increased alpha power at P4, and decreased beta power at P3. In addition, ADHD-hyperactive cases also showed decreased theta power at F8 and Fz, decreased alpha power at T5, and decreased power of the lower alpha band at F4 and C4 areas.

Subsequently, a comparison between the patterns of ADHD subtypes was also performed and the results are shown in Table 5 and Figure 5.
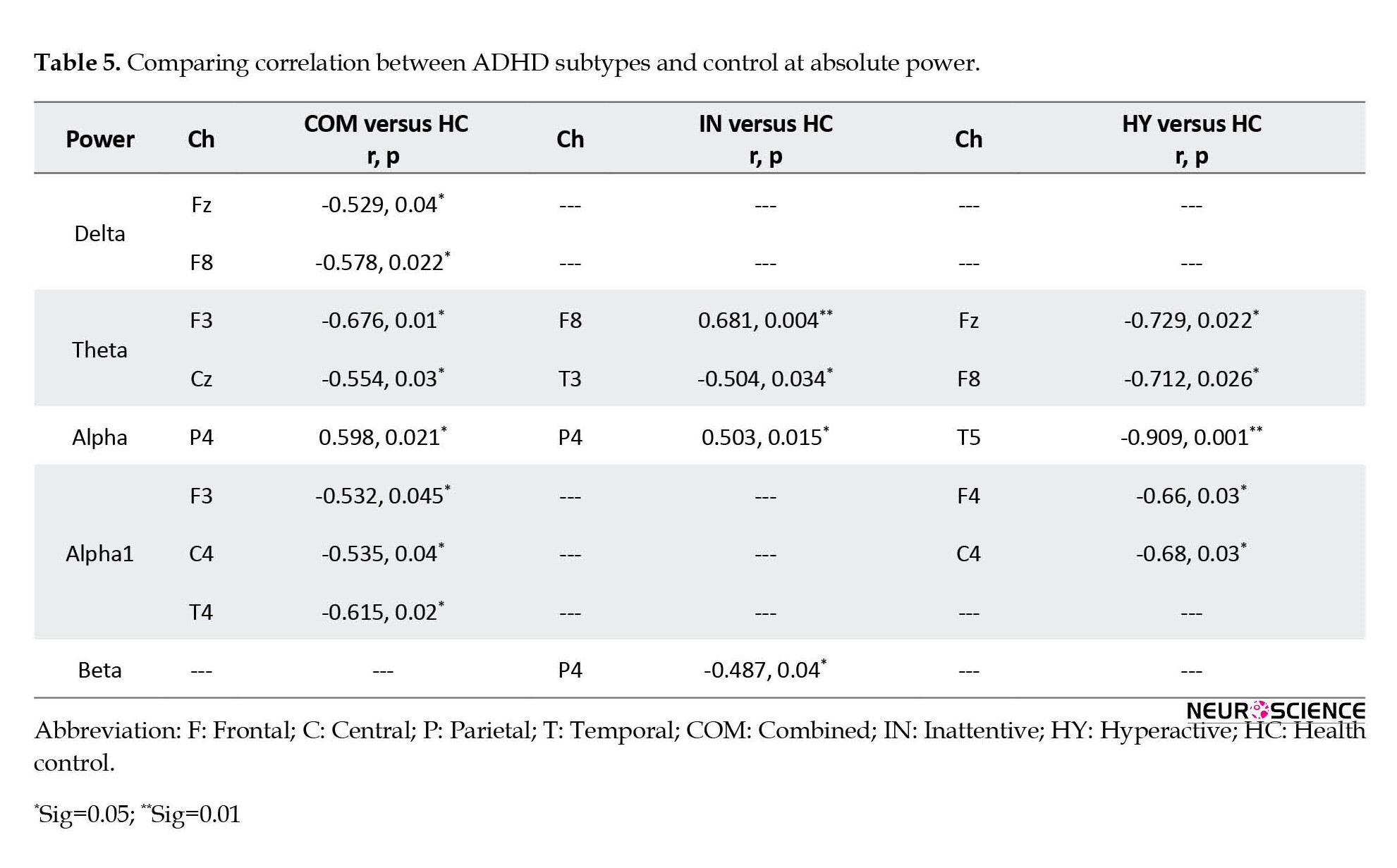
Elevated alpha 2 power at F7 and Cz, decreased delta power at F8 and T6, and decreased theta power at O1 were observed in ADHD-combined cases as compared to the ADHD-inattentive group. Moreover, the ADHD-combined group showed increased alpha power at C4 and T5, increased beta power at Fp2, F3, and O1, and decreased theta power at Cz compared to the ADHD-hyperactive group. In addition, the ADHD-inattentive group was also compared to the ADHD-hyperactive group and presented increased theta power at F7 and alpha power at T5, and lower alpha power at F4 and beta power at O1.

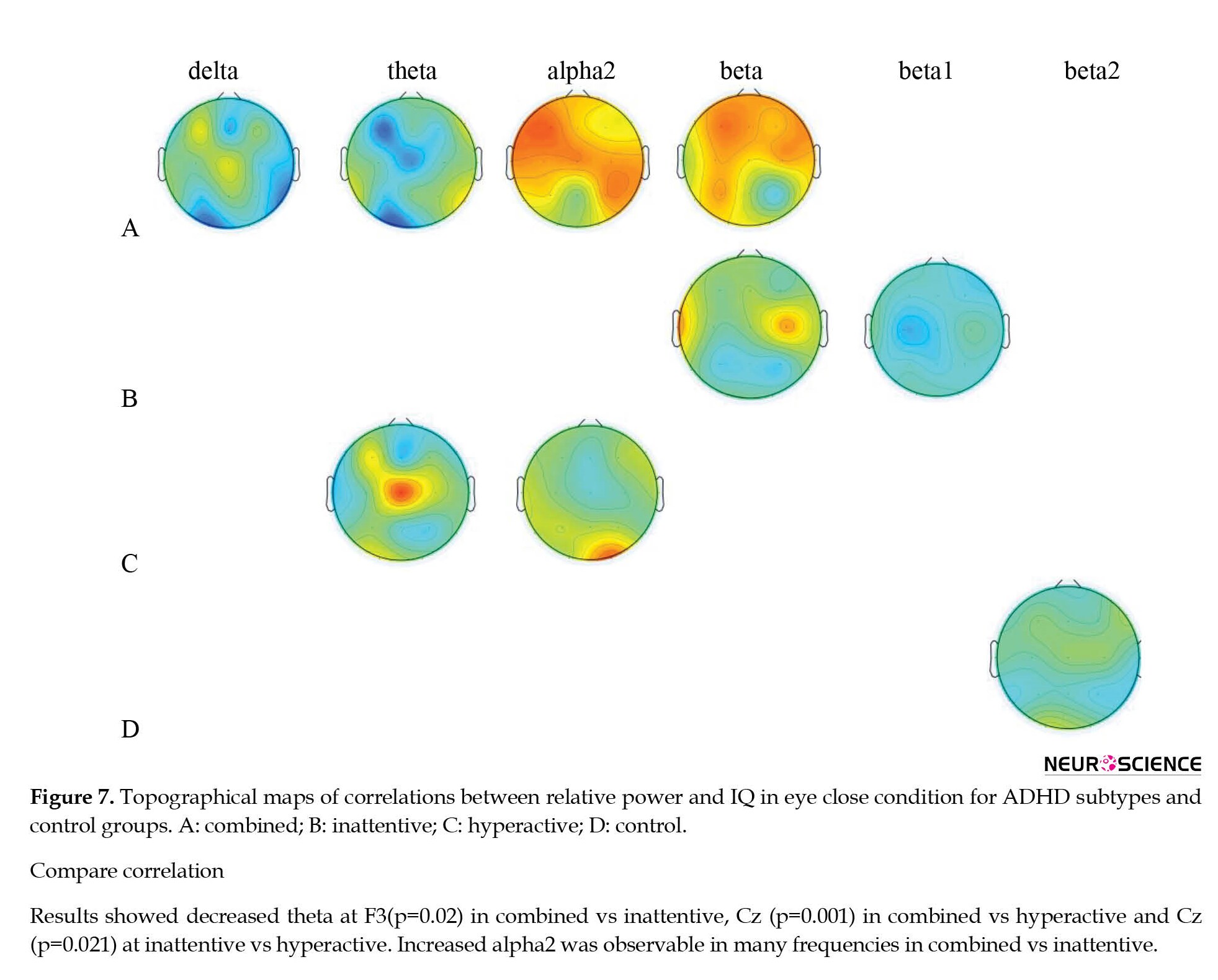
Topographical differences in the association between intelligence and patterns of ratio power for ADHD subtypes as well as control group are presented in Tables 6 and 7 and Figure 6.

In comparison with the healthy control group, the ADHD-combined group showed decreased theta/alpha at f3, cz, o1, and o2, and decreased theta/beta at f3, c3, and cz compared to the control group. The ADHD-inattentive group also showed decreased theta/alpha at f3, and the ADHD-hyperactive group presented decreased theta/beta at f3 (Table 6 and Figure 6).

In addition, the ADHD-combined group compared to the ADHD-inattentive present ed decreased theta/alpha at o1 and theta/beta at cz. A decrease in theta/beta at cz was also observed in the ADHD-combined group compared to the ADHD-hyperactive group (Table 7 and Figure 6).

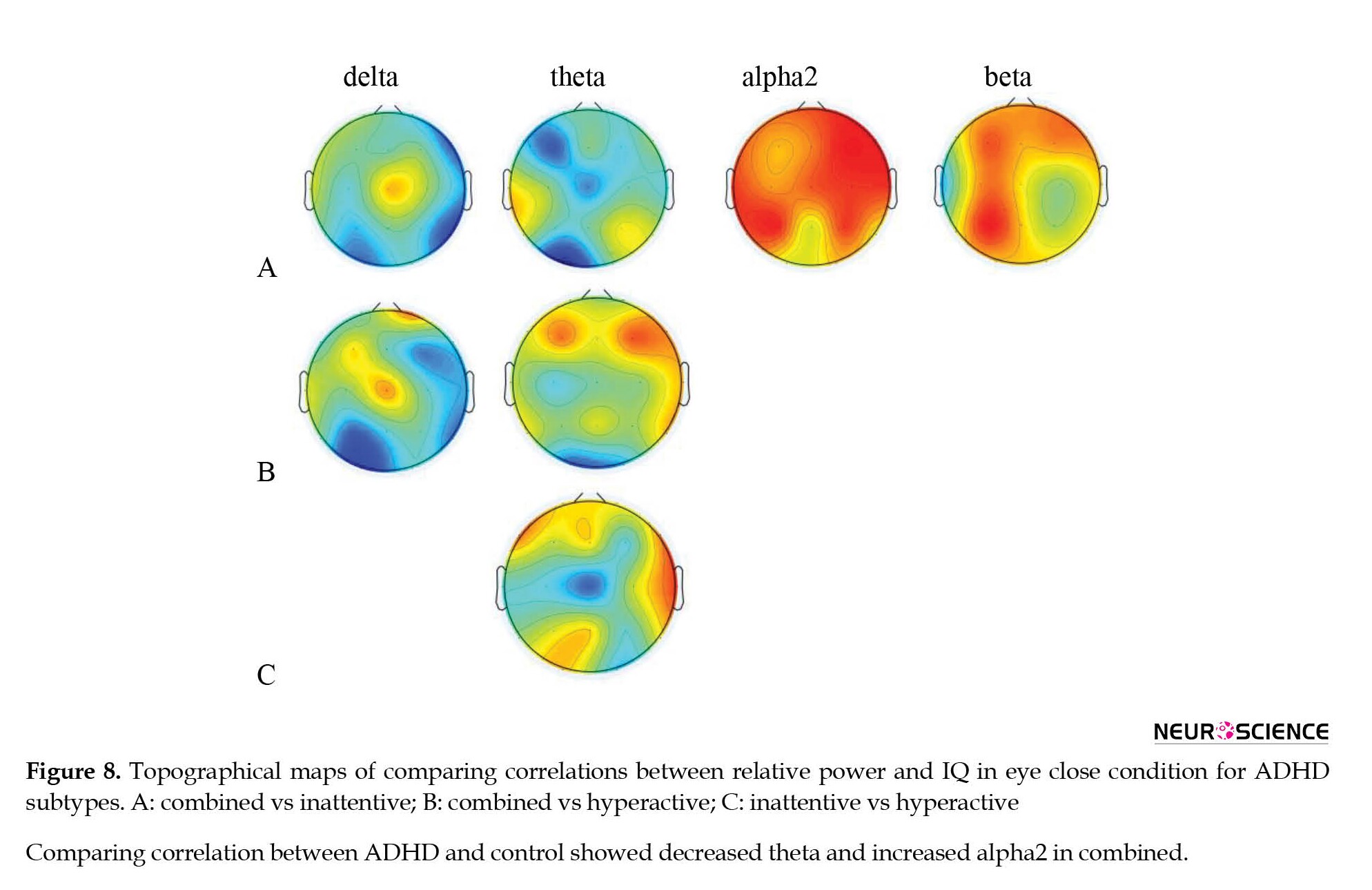
In the second phase of analysis, comparisons of whole-brain patterns of the association between IQ scores and EEG power were performed to discriminate patterns of EEG power related to IQ scores in ADHD subtypes from healthy controls (Table 8 and Figure 7).
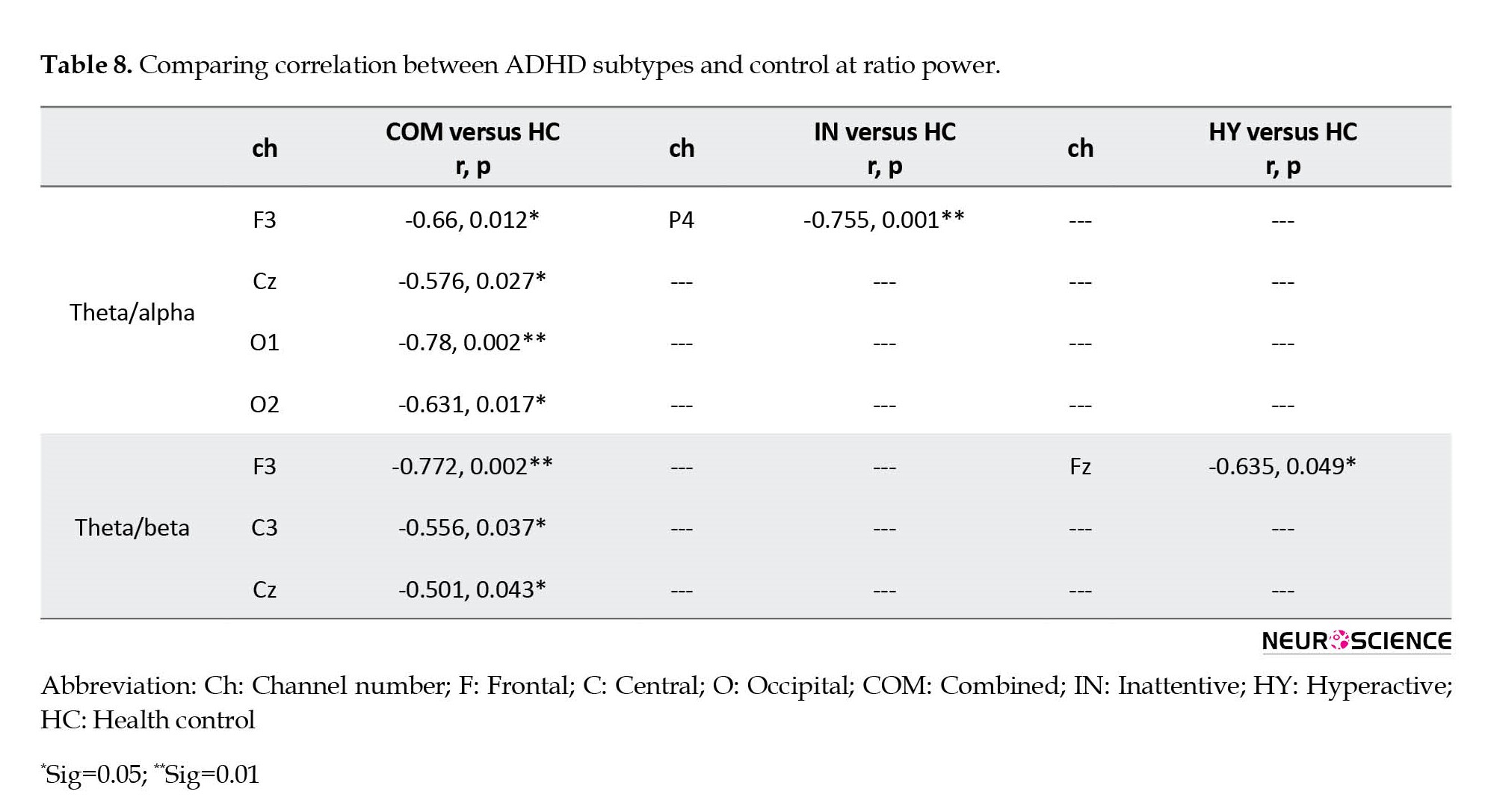
The ADHD-combined group revealed the increased correlation between IQ score and whole-brain pattern of beta power, and the decreased correlation between IQ score and whole-brain pattern of power at theta and alpha1 bands. The inattentive ADHD group showed no significant differences, and the hyperactive/impulsive ADHD group showed a decreased correlation between IQ score and whole-brain pattern of power at theta, alpha1, and beta bands compared to the healthy control group.
In addition, comparisons of ADHD subtypes also showed that the combined ADHD group compared to the ADHD-inattentive group presented an increased correlation between IQ score and whole-brain pattern of power at alpha2 and beta bands, while the correlation decreased at alpha1. The combined ADHD group compared to the hyperactive ADHD group showed an increased correlation at alpha and beta bands. The inattentive ADHD groupcompared to the hyperactive ADHD group showed an increased correlation between alpha and alpha1 and IQ scores.
4. Discussion
The present study primarily investigated the association between brain functions based on absolute, and ratio power of EEG, and IQ scores in various individuals within a normal range of IQ. The association pattern was then compared between three ADHD subtypes and a healthy control group. The absolute and ratio powers were calculated in the conventional EEG frequency bands, including delta, theta, and alpha with sub-bands alpha1 and alpha2, and beta. Then, the association between IQ score and EEG powers was compared within the groups. Calculation of the absolute power index provides a reliable method to differentiate clinical and normal groups (Dumermuth & Molinari, 1987). This study also represented theta/alpha and theta/beta power ratios because these ratio powers can differentiate ADHD individuals from healthy controls (Clarke, et al., 2002).
The association between IQ score and EEG power has shown cognitive implication, which is discussed here. In our healthy control group, the association between IQ score and EEG bands showed a pattern of lateralization, more significant in the left hemisphere (frontal, temporal and occipital regions). Several studies have associated the left hemisphere with intelligence and working memory (Basso, et al., 1982) (Paulraj, et al., 2018) (Basso, et al., 1981). Our findings in the healthy control group also showed a significant association between IQ score and EEG power at theta and alpha bands, which is nicely fit with the previous studies (Klimesch, 1999a), (Yılmaz, et al., 2014). The association was mainly observed in the frontal and temporal regions. The frontal region is involved in many functions, such as cognition, emotional behavior, memory, and language (Mundy-Castle & Nelson, 1960) and the temporal region has been shown to associate with the vocabulary score of the Wechsler test (Dobbins & Russell, 1990) and arithmetic score as well (Inouye, et al., 1993).
Nevertheless, the pattern of association between the IQ score of the Ravan test and EEG power was completely different in ADHD individuals compared to the healthy control group. Our ADHD group showed a decreased association between EEG bands and IQ scores acquired by the Ravan test compared to the healthy control group. The Ravan test mainly estimates novel problem-solving, which is defined as solving problems that cannot be answered directly by referring to long-term knowledge but instead requires analytic or fluid reasoning (Prabhakaran et al., 1997). The network of brain regions involved in fluid reasoning is comprised of the frontopolar, middle and inferior frontal gyri, and parietal region (Rapport et al., 2008). These regions, more specifically the frontal, are also associated with working memory, which is weak in ADHD cases (Rapport et al., 2008). Therefore, decreased association between EEG bands and IQ score at frontal regions in our ADHD groups nicely fit with the deficiency of working memory and fluid reasoning acquired by the Raven test in past studies (Tamm & Juranek, 2012). In addition, our finding has implications in terms of the default mode network (DMN). This network is comprised of the medial prefrontal, precuneus, and angular gyrus (Raichle et al., 2001). DMN, which is typically activated during resting state conditions and deactivated during task performance (Broyd et al., 2009), was reported to be an underlying cause of inadequate performance in ADHD (Sonuga-Barke & Castellanos, 2007). Decreased correlation between IQ and EEG bands in our ADHD individuals compared to the healthy control group in the anterior region, plays an important role in working memory and attention (Castellanos et al., 2008), which may imply deficits in the anterior part of default mode network (DMN) in ADHD patients as reported in previous studies (Castellanos et al., 2008). In this study, the patterns of correlation between IQ score and EEG power in ADHD-combined and ADHD-inattentive were similar. This finding is corroborated by former studies, suggesting similar EEG abnormality in combined and inattentive subtypes (Clarke et al., 2001a). However, the pattern for our hyperactive ADHD cases is different from two other subtypes, which can be due to the small sample size of this group in the current study.
On the other hand, the ADHD individuals revealed a decreased association between IQ score and power of EEG at the theta band. While the association between IQ score and alpha power increased compared to the healthy control group. This finding shows the existence of a compensatory mechanism such that decreased theta can be compensated by increased alpha in the ADHD group to keep adequate intellectual function. The combined ADHD group also showed a positive association at the beta band. It has been shown that active attention and psychomotor (Polunina & Davydov, 2006) have been related to brain activities at the beta band. Therefore, a positive association between EEG beta power and intelligence, in our findings, seems to be a logical pattern. IQ and beta power were highly associated compared to that of alpha in our combined ADHD group. We think this finding of the combined group is in the same line with Webster’s theory who suggested that in the condition of serious mental activity beta replaced alpha’s place (Webster, 1978).
Moreover, unlike the healthy control group, no specific lateralization pattern was found in ADHD subtypes. The recent finding may provide additional support for the presence of a compensatory mechanism such that deficiencies in the right hemisphere are compensated by the left hemisphere to have adequate intellectual performance.
Our findings of the different association between EEG bands and IQ between the healthy controls and the ADHD groups were not limited to some specific brain regions and significant differences were also observed in the global pattern extracted from the whole brain regions. Decreased theta in all subtypes of ADHD has been shown in all brain regions. The differential patterns of associations in local and global measures potentially imply that a dynamic change in the brain’s regional functions towards increasing the alpha (or beta) band activities and decreasing the laterality effect could be a compensatory mechanism to maintain a normal intelligence score in ADHD individuals.
At ratio power, our results showed a decreased theta/alpha ratio in the ADHD group (all subtypes) compared to the healthy control group. Assuming that the theta/alpha power ratio has been introduced as a criterion for intelligence (Markowitsch & Pritzel, 1985), it is expected that the theta/alpha ratio should not be different in IQ-matched individuals. While our findings put a question mark on the validity of the above-mentioned criteria for intelligence in ADHD individuals.
5. Conclusion
Significant changes observed in the score of association between IQ and EEG power in the ADHD individuals compared to the IQ-matched healthy control group suggests the existence of a compensatory mechanism to hold a suitable cognitive performance (IQ) in the ADHD subjects. We conclude that the dynamic of the brain activities in addition to the specific brain structure plays a great role in intelligence. The discrepancy of theta/alpha ratio in groups with a similar IQ score may suggest a weakness of this marker for intelligence. We hope that these findings could provide additional information to pave the way to a better understanding of the relationship between brain functions and intelligence. Extra information have been provided in tables (9-11) and figures (6-9).


Limitation
The results of this study had a number of limitations. First, it was limited to a few numbers of hyperactivity/impulsivity ADHD subjects compared to other subtypes of the ADHD and control group. Second, the study focused on the age range of 8-12 years. Because EEG activity shifts from less to high-frequency bands during maturation, a more detailed developmental analysis may provide a better insight into the relationship between intelligence and EEG oscillations. Furthermore, the study was performed in the eyes-close resting state; therefore, further investigation on the task-based EEG will be helpful to strengthen the results. Although the findings are inspiring, more studies are required to test the reliability and validity of the results.
Ethical Considerations
Compliance with ethical guidelines
The subjects were assessed after obtaining the in -formed consent signed by the parents, and the research protocol was approved by the Ethics Committee of Iran University of Medical Sciences.
Funding
This research was supported by grants from the Cog-nitive Sciences and Technologies Council of Iran (Grant No.: 1948).
Authors' contributions
All authors contributed in designing, running, and writing all parts of the research.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
We would like to thank Reza Rostami, -Masoud Nosrat-abadi, and their colleagues for their generous assistance in the data collection. We also would like to thank the Iranian Council of Cognitive Science and Technologies for financial support on this study.
References
Anokhin, A. P., Lutzenberger, W., & Birbaumer, N. (1999).Spatiotemporal organization of brain dynamics and intelligence: an EEG study in adolescents. International Journal of Psychophysiology, 33(3), 259-273. [PMID]
Arns, M., Conners, C. K., & Kraemer, H. C. (2013). A decade of EEG theta/beta ratio research in ADHD: A meta-analysis. Journal of Attention Disorders, 17(5), 374-383. [DOI:10.1177/1087054712460087] [PMID]
Basso, A., Capitani, E., Luzzatti, C., & Spinnler, H. (1981). Intelligence and left hemisphere disease. The role of aphasia, apraxia and size of lesion. Brain: A Journal of Neurology, 104(Pt 4), 721-734. [DOI:10.1093/brain/104.4.721] [PMID]
Basso, A., Spinnler, H., Vallar, G., & Zanobio, M. E. (1982). Left hemisphere damage and selective impairment of auditory verbal short-term memory. A case study. Neuropsychologia, 20(3), 263-274. [DOI:10.1016/0028-3932(82)90101-4] [PMID]
Bloemsma, J. M., Boer, F., Arnold, R., Banaschewski, T., Faraone, S. V., & Buitelaar, J. K., et al. (2013). Comorbid anxiety and neurocognitive dysfunctions in children with ADHD. European Child & Adolescent Psychiatry, 22(4), 225–234. [DOI:10.1007/s00787-012-0339-9] [PMID]
Bresnahan, S. M., & Barry, R. J. (2002). Specificity of quantitative EEG analysis in adults with attention deficit hyperactivity disorder. Psychiatry Research, 112(2), 133-144. [DOI:10.1016/S0165-1781(02)00190-7] [PMID]
Broyd, S. J., Demanuele, C., Debener, S., Helps, S. K., James, C. J., & Sonuga-Barke, E. J. (2009). Default-mode brain dysfunction in mental disorders: A systematic review. Neuroscience and Biobehavioral Reviews, 33(3), 279–296. [DOI:10.1016/j.neubiorev.2008.09.002] [PMID]
Burgess, G. C., Gray, J. R., Conway, A. R. A., & Braver, T. S. (2011). Neural mechanisms of interference control underlie the relationship between fluid intelligence and working memory span. Journal of Experimental Psychology. General, 140(4), 674–692. [PMID]
Cabeza, R., & Nyberg, L. (2000). Imaging cognition II: An empirical review of 275 PET and fMRI studies. Journal of cognitive neuroscience, 12(1), 1-47. [DOI:10.1162/08989290051137585]
Castellanos, F. X., Margulies, D. S., Kelly, C., Uddin, L. Q., Ghaffari, M., & Kirsch, A., et al. (2008). Cingulate-precuneus interactions: A new locus of dysfunction in adult attention-deficit/hyperactivity disorder. Biological Psychiatry, 63(3), 332-337. [PMID] [PMCID]
Chabot, R. J., & Serfontein, G. (1996). Quantitative electroencephalographic profiles of children with attention deficit disorder. Biological Psychiatry, 40(10), 951-963. [DOI:10.1016/0006-3223(95)00576-5] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2001). Age and sex effects in the EEG: Differences in two subtypes of attention-deficit/hyperactivity disorder. Clinical Neurophysiology, 112(5), 815-826. [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2001). Electroencephalogram differences in two subtypes of attention-deficit/hyperactivity disorder. Psychophysiology, 38(2), 212-221. [DOI:10.1111/1469-8986.3820212] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2002).EEG analysis of children with attention-deficit/hyperactivity disorder and comorbid reading disabilities. Journal of Learning Disabilities, 35(3), 276-285. [DOI:10.1177/002221940203500309] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., Selikowitz, M., Magee, C. A., & Johnstone, S. J., et al. (2006). Quantitative EEG in low-IQ children with attention-deficit/hyperactivity disorder. Clinical Neurophysiology, 117(8), 1708-1714. [PMID]
Deary, I. J., Penke, L., & Johnson, W. (2010). The neuroscience of human intelligence differences. Nature Reviews Neuroscience, 11(3), 201–211. [DOI:10.1038/nrn2793] [PMID]
Diedenhofen, B., & Musch, J. (2015). Cocor: A comprehensive solution for the statistical comparison of correlations. Plos One, 10(3), e0121945. [PMID] [PMCID]
Dietrich, A. (2004). The cognitive neuroscience of creativity. Psychonomic Bulletin & Review, 11(6), 1011-1026. [DOI:10.3758/BF03196731] [PMID]
Dobbins, C., & Russell, E. W. (1990). Left temporal lobe brain damage pattern on the Wechsler Adult Intelligence Scale. Journal of Clinical Psychology, 46(6), 863-868. [PMID]
Dumermuth, G., & Molinari, L. (1987). Spectral analysis of the EEG. Some fundamentals revisited and some open problems. Neuropsychobiology, 17(1-2), 85–99. [PMID]
Fischer, F. U., Wolf, D., Scheurich, A., & Fellgiebel, A. (2014). Association of structural global brain network properties with intelligence in normal aging. PloS One, 9(1), e86258. [PMID] [PMCID]
Fogel, S. M., Nader, R., Cote, K. A., & Smith, C. T. (2007). Sleep spindles and learning potential. Behavioral Neuroscience, 121(1), 1-10. [DOI:10.1037/0735-7044.121.1.1] [PMID]
Geake, J. G., & Hansen, P. C. (2005). Neural correlates of intelligence as revealed by fMRI of fluid analogies. Neuroimage, 26(2), 555-564. [PMID]
Giannitrapani, D., & Liberson, W. (1985). The electrophysiology of intellectual functions. Basel: Karger. [Link]
Gottfredson, L. S. (1997). Mainstream science on intelligence: An editorial with 52 signatories, history, and bibliography. Intelligence, 24(I), 13-23. [Link]
Gray, J. R., Chabris, C. F., & Braver, T. S. (2003). Neural mechanisms of general fluid intelligence. Nature Neuroscience, 6(3), 316-322. [DOI:10.1038/nn1014] [PMID]
Haier, R. J., & Jung, R. E. (2008). Brain imaging studies of intelligence and creativity: What is the picture for education? Roeper Review, 30(3), 171-180. [DOI:10.1080/02783190802199347]
Hearne, L. J., Mattingley, J. B., & Cocchi, L. (2016). Functional brain networks related to individual differences in human intelligence at rest. Scientific Reports, 6, 32328. [PMID] [PMCID]
Heilman, K. M., Nadeau, S. E., & Beversdorf, D. O. (2003). Creative innovation: Possible brain mechanisms. Neurocase, 9(5), 369-379. [DOI:10.1076/neur.9.5.369.16553] [PMID]
Inouye, T., Shinosaki, K., Iyama, A., & Matsumoto, Y. (1993).Localization of activated areas and directional EEG patterns during mental arithmetic. Electroencephalography and Clinical Neurophysiology, 86(4), 224-230. [PMID]
Jung, R. E., & Haier, R. J. (2007). The Parieto-Frontal Integration Theory (P-FIT) of intelligence: Converging neuroimaging evidence. Behavioral and Brain Sciences, 30(2), 135-154. [PMID]
Kane, M. J., & Engle, R. W. (2002). The role of prefrontal cortex in working-memory capacity, executive attention, and general fluid intelligence: An individual-differences perspective. Psychonomic Bulletin & Review, 9(4), 637-671. [DOI:10.3758/BF03196323] [PMID]
Kang, J. S., & Lee, M. (2015). Characteristics of EEG signals during tough and easy linguistic problem solving. Paper presented at: The Brain-Computer Interface (BCI), Gangwon, Korea, 12-14 January 2015. [DOI:10.1109/IWW-BCI.2015.7073026]
Kitsune, G. L., Cheung, C. H., Brandeis, D., Banaschewski, T., Asherson, P., & McLoughlin, G., et al. (2015). A matter of time: The influence of recording context on EEG spectral power in adolescents and young adults with ADHD. Brain Topography, 28(4), 580-590. [PMID] [PMCID]
Klimesch, W. (1999a). EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Research Reviews, 29(2-3), 169–195. [DOI:10.1016/S0165-0173(98)00056-3] [PMID]
Klimesch, W. (1999). EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Research Reviews, 29(2-3), 169-195. [DOI:10.1016/S0165-0173(98)00056-3] [PMID]
Klimesch, W., Schimke, H., & Pfurtscheller, G. (1993). Alpha frequency, cognitive load and memory performance. Brain Topography, 5(3), 241-251. [DOI:10.1007/BF01128991] [PMID]
Loo, S. K., Hale, S. T., Hanada, G., Macion, J., Shrestha, A., & McGough, J. J., et al. (2010). Familial clustering and DRD4 effects on electroencephalogram measures in multiplex families with attention deficit/hyperactivity disorder. Journal of the American Academy of Child & Adolescent Psychiatry, 49(4), 368-377. [PMID]
Markowitsch, H. J., & Pritzel, M. (1985). The neuropathology of amnesia. Progress in Neurobiology, 25(3), 189-287. [DOI:10.1016/0301-0082(85)90016-4] [PMID]
Marosi, E., Rodríguez, H., Harmony, T., Yañez, G., Rodrìguez, M., & Bernal, J., et al. (1999). Broad band spectral EEG parameters correlated with different IQ measurements. The International Journal of Neuroscience, 97(1-2), 17–27. [PMID]
Martín-Loeches, M., Muñoz-Ruata, J., Martínez-Lebrusant, L., & Gómez-Jarabo, G. (2001). Electrophysiology and intelligence: The electrophysiology of intellectual functions in intellectual disability. Journal of Intellectual Disability Research : JIDR, 45(Pt 1), 63–75. [DOI:10.1111/j.1365-2788.2001.00292.x] [PMID]
Moretti, D. V. (2015). Theta and alpha EEG frequency interplay in subjects with mild cognitive impairment: Evidence from EEG, MRI, and SPECT brain modifications. Frontiers in Aging Neuroscience, 7, 31. [PMID] [PMCID]
Mundy-Castle, A., & Nelson, G. (1960). Intelligence, personality and brain rhythms in a socially isolated community. Nature, 185(4711), 484-485. [DOI:10.1038/185484a0] [PMID]
Mundy‐Castle, A. (1958). Electrophysiological correlates of intelligence. Journal of Personality, 26(2), 184-199. [DOI:10.1111/j.1467-6494.1958.tb01579.x] [PMID]
Narr, K. L., Woods, R. P., Thompson, P. M., Szeszko, P., Robinson, D., & Dimtcheva, T., et al. (2006). Relationships between IQ and regional cortical gray matter thickness in healthy adults. Cerebral cortex (New York, N.Y. : 1991), 17(9), 2163–2171. [PMID]
Neubauer, A. C., Grabner, R. H., Fink, A., & Neuper, C. (2005).Intelligence and neural efficiency: Further evidence of the influence of task content and sex on the brain-IQ relationship. Cognitive Brain Research, 25(1), 217-225. [DOI:10.1016/j.cogbrainres.2005.05.011] [PMID]
Paulraj, S. R., Schendel, K., Curran, B., Dronkers, N. F., & Baldo, J. V. (2018). Role of the left hemisphere in visuospatial working memory. Journal of Neurolinguistics, 48, 133–141. [PMID] [PMCID]
Polunina, A. G., & Davydov, D. M. (2006). EEG correlates of Wechsler Adult Intelligence Scale. The International Journal of Neuroscience, 116(10), 1231–1248. [PMID]
Prabhakaran, V., Smith, J. A., Desmond, J. E., Glover, G. H., & Gabrieli, J. D. (1997). Neural substrates of fluid reasoning: An fMRI study of neocortical activation during performance of the Raven’s Progressive Matrices Test. Cognitive Psychology, 33(1), 43-63. [DOI:10.1006/cogp.1997.0659] [PMID]
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences, 98(2), 676-682. [PMID] [PMCID]
Rapport, M. D., Alderson, R. M., Kofler, M. J., Sarver, D. E., Bolden, J., & Sims, V. (2008). Working memory deficits in boys with attention-deficit/hyperactivity disorder (ADHD): The contribution of central executive and subsystem processes. Journal of Abnormal Child Psychology, 36(6), 825-837. [PMID]
Raven, J. C. (2000). Manual for Raven’s progressive matrices and vocabulary scales. San Antonio: Harcourt Assessment. [Link]
Raven, J. (2000). The Raven’s progressive matrices: Change and stability over culture and time. Cognitive Psychology, 41(1), 1-48. [DOI:10.1006/cogp.1999.0735] [PMID]
Reis, P. M., Hebenstreit, F., Gabsteiger, F., von Tscharner, V., & Lochmann, M. (2014). Methodological aspects of EEG and body dynamics measurements during motion. Frontiers in Human Neuroscience, 8, 156. [PMID] [PMCID]
Reynolds, C. R., & Brown, R. T. (1984). Bias in mental testing. In C. R. Reynolds, & R. T. Brown (eds.), Perspectives on bias in mental testing (pp. 1-39). Boston: Springer. [DOI:10.1007/978-1-4684-4658-6_1]
Schacter, D. L. (1977). EEG theta waves and psychological phenomena: A review and analysis. Biological Psychology, 5(1), 47-82. [DOI:10.1016/0301-0511(77)90028-X] [PMID]
Song, M., Zhou, Y., Li, J., Liu, Y., Tian, L., & Yu, C., et al. (2008). Brain spontaneous functional connectivity and intelligence. Neuroimage, 41(3), 1168-1176. [PMID]
Sonuga-Barke, E. J., & Castellanos, F. X. (2007). Spontaneous attentional fluctuations in impaired states and pathological conditions: A neurobiological hypothesis. Neuroscience & Biobehavioral Reviews, 31(7), 977-986. [DOI:10.1016/j.neubiorev.2007.02.005] [PMID]
Stankova, E. P., & Myshkin, I. Y. (2016). Association between individual EEG characteristics and the level of intelligence. Moscow University Biological Sciences Bulletin, 71(4), 256-261. [DOI:10.3103/S0096392516040118]
Tamm, L., & Juranek, J. (2012). Fluid reasoning deficits in children with ADHD: Evidence from fMRI. Brain Research, 1465, 48-56. [PMID] [PMCID]
Tang, C. Y., Eaves, E. L., Ng, J. C., Carpenter, D. M., Mai, X., & Schroeder, D. H., et al. (2010). Brain networks for working memory and factors of intelligence assessed in males and females with fMRI and DTI. Intelligence, 38(3), 293-303. [DOI:10.1016/j.intell.2010.03.003]
Thatcher, R. W., North, D., & Biver, C. (2007). Intelligence and EEG current density using low‐resolution electromagnetic tomography (LORETA). Human Brain Mapping, 28(2), 118-133. [PMID]
Thatcher, R. W., North, D., & Biver, C. (2005). EEG and intelligence: Relations between EEG coherence, EEG phase delay and power. Clinical Neurophysiology, 116(9), 2129-2141. [PMID]
Thatcher, R. W., North, D. M., & Biver, C. J. (2008). Intelligence and EEG phase reset: A two compartmental model of phase shift and lock. Neuroimage, 42(4), 1639-1653. [DOI:10.1016/j.neuroimage.2008.06.009] [PMID]
Webster, J. G., & Clark, J. W. (2010). Medical instrumentation: Application and design. New Jersey: John Wiley & Sons. [Link]
Webster, J. G. (1978). Medical instrumentation-application and design. Journal of Clinical Engineering, 3(3), 306. [DOI:10.1097/00004669-197807000-00017]
Yılmaz, B., Korkmaz, S., Arslan, D. B., Güngör, E., & Asyalı, M. H. (2014). Like/dislike analysis using EEG: Determination of most discriminative channels and frequencies. Computer Methods and Programs in Biomedicine, 113(2), 705-713. [DOI:10.1016/j.cmpb.2013.11.010] [PMID]
Intelligence is a general mental ability, which includes planning, reasoning, comprehension, abstraction, and learning (Gottfredson, 1997). Therefore, intelligence scores may be used to predict one’s performance (Haier & Jung, 2008). This cognitive ability is produced by brain structure and functions, which could be traced by neuroimaging techniques. In this regard, neuroimaging studies have mainly presented a region-specific pattern of anatomy as well as activities associated with the intelligence score. In terms of structure, with the advent of magnetic resonance imaging (MRI) studies, researchers have found a strong association between intelligence and grey matter density (Narr et al., 2006). For example, an association between intelligence and grey matter density has been reported in the basal ganglia (Dietrich, 2004), hippocampus (Deary et al., 2010), fusiform (Deary et al., 2010), posterior region (Heilman et al., 2003), Parietal and frontal (Thatcher et al., 2008; Thatcher et al., 2007) and dorsolateral prefrontal cortex (DLPFC) (Cabeza & Nyberg, 2000). Other studies associated global properties of the structural brain network with general intelligence (Fischer et al., 2014). Similar to structural studies, functional studies also suggest a network for intelligence (Jung & Haier, 2007). In this context, about the relationship between intelligence and the function of the brain, a variety of psychophysiological measurement methods ranging from positron emission tomography (PET) (Kane & Engle, 2002; Gray et al., 2003), functional magnetic resonance imaging (fMRI) (Geake & Hansen, 2005; Burgess et al., 2011; Tang et al., 2010) and electroencephalography (EEG) (Neubauer et al., 2005; Anokhin et al., 1999) has been employed. Of the all above-mentioned methods, EEG is one of the most popular used method because it is cheap, portable, and easy to wear, and has a high temporal resolution (Reis et al., 2014) and it is proper to use in the applied studies. EEG presented a correlation with intelligence (Mundy‐Castle, 1958). Recent studies have demonstrated that highly intelligent individuals allocate fewer resources and recruit minimum task-relevant brain areas when performing an easy task, while they allocate more resources and recruit several brain areas when the task demand is high. In contrast, low intelligent individuals do not use different resource allocation strategies during easy and hard tasks (Kang & Lee, 2015).
In general, the association of IQ scores and EEG properties have been studied using the power or amplitude (Klimesch, 1999b; Giannitrapani & Liberson, 1985) (Martín‐Loeches et al., 2001), and the functional connectivity networks extracted with coherence and phase delays (Thatcher et al., 2005; Thatcher et al., 2007). Moreover, the brain works in a frequency-specific manner; therefore, the association between IQ scores and EEG parameters has also been investigated in the conventional frequency bands as well. The conventional frequency bands in EEG include delta (slowest), theta, alpha, beta, and gamma (fastest) bands. These frequency bands have a critical role for performing the cognitive tasks (Klimesch et al., 1993; Moretti, 2015). In addition, cognitive disabilities have also been related to changes in the neural oscillatory pattern (frequency-specific activities). For instance, an increase in task demand increases the theta power and causes more synchronization in this band. In contrast, an increase in a task demand decreases the alpha power and causes desynchronization in this band (Schacter, 1977) (Webster, 1978) (Marosi et al., 1999) (Fogel et al., 2007). EEG waves are caused by large numbers of neurons firing in synchrony. Synchronization behavior of EEG signals is important for decoding information processing in human brain.
In terms of the IQ index of EEG, it has been shown that alpha power is the most closely correlated with IQ score (Stankova & Myshkin, 2016). In addition, high-IQ individuals present higher modularity in their brain functional connectivity network (FCN) and have different patterns of activations between short-range and long-range connections in the FCN (Hearn et al., 2016) (Song et al., 2008). The above-mentioned studies have mainly focused on normal individuals.
Neurodevelopmental disorders, such as attention deficit and hyperactive disorder (ADHD) could influence the pattern of brain activities. For instance, studies have reported greater frontocentral theta activity, lower beta activity, and higher theta/beta ratio in ADHD subjects compared to healthy controls (Clarke et al., 2001b; Loo et al., 2010) (Bresnahan & Barry, 2002) (Arns et al., 2013) (Loo et al., 2010). However, there is inconsistency in the previous studies on the effect of IQ on EEG power in ADHD. For instance, Clark et al. reported that IQ does not affect the EEG power in ADHD individuals compared to their matched control subjects (Clarke et al., 2006) While Chabot and Serfontein (1996) showed that there is a different oscillatory pattern in EEG of ADHD children with high and low IQ. They suggested that children with low IQs show lower alpha and beta bands (Chabot & Serfontein, 1996). Other studies have also highlighted the importance of IQ on any potential group’s differences in ADHD with high and low IQ (Kitsune et al., 2015). Therefore, it seems that studies should try to control the effects of IQ on their data. In this study, we hypothesized that neural correlates of intelligence should not have a fixed pattern rather it must follow a dynamic pattern to compensate for the deficits caused by the disorder.
Therefore, in this study, EEG was recorded from three ADHD subtypes and a control group in eye closed condition. Then, the correlation of EEG spectral bands using estimates of absolute and ratio with IQ and significant differences within groups were computed at a specific brain region. We also estimated the whole pattern of association at each absolute frequency band. Details of the experimental design and data analysis are explained in the next section.
2. Materials and Methods
A schematic of the experimental design is presented in Figure 1.

Subjects
One hundred nine boy participants comprising 22 combined (age=9.13±0.99, IQ=103.13±11.02), 27 inattentive (age=8.62±1.30, IQ=104.37±10.22), 14 hyperactive (age=9.42±1.74, IQ=107.5±9.27), and 46 healthy controls (age=9.41±1.42, IQ= 102.39±10.83) were recruited in this study. All subjects were right-handed and there were no significant differences in age and IQ between the groups (Table 1). The diagnostic assessment was performed by a psychiatrist and a senior clinical psychologist using the Persian version of the Structural Clinical Interview for DSM-V (SCID). Average DSM-V symptom scores of inattentive, hyperactive, and combined were 7.0±1.3, 8.2±2.9, and 6.3±1.3, respectively. Children with brain damage, neurological disorder, and epilepsy as well as children who consume stimulants or were under neurotherapy were excluded. The research protocol was approved by the Iran University of Medical Science Ethics Committee and all parents signed informed consent.

IQ scores
All participants completed Ravan’s standard progressive matrices (SPM) test. This multiple-choice test is used to assess abstract reasoning and nonverbal ability (Raven, 2000b). It has also been considered to be the best measure of spearman’s g factor (Reynolds & Brown, 1984) and intelligence (Raven, 2000a).
EEG data recording and analysis
EEG data were recorded during an eyes-closed resting state for 4 minutes while children were seated on a comfortable chair. All subjects were asked to sit relax and try not to blink. The EEG data were registered using a 19-electrode Mitsar amplifier (www.mitsar-medical.com) with a sampling rate of 250 Hz. Electrodes were placed on the scalp using a standard 10-20 montage (Fp1, Fp2, Fz, F3, F4, F7, F8, Cz, C3, C4, T3, T4, T5, T6, Pz, P3, P4, O1, and O2) with the average of ear channels used as reference and FPz as the ground electrode. Electrode impedances were kept below 5 kΩ.
After recording, a self-written program using Matlab (https://www.mathworks.com) and EEGlab functions (https://sccn.ucsd.edu/eeglab/) was used to process the data. Standard pre-processing included band-pass filtering (1-40 Hz), segmenting the data into epochs of 1-second duration, automatic rejection of disturbing channels using probability (artifact rejection was carried out using both to single electrodes and the collection of all electrodes), spectrum (thresholds are expressed in terms of amplitude changes relative to baseline in dB), and kurtosis (technically called the four first cumulates of the distribution) of the signal interpolation of rejected channels by averaging its spherical neighbors, removing unreliable epochs, and referencing to the average. Subsequently, the pre-processed EEG data were transformed to the frequency domain using Fast Fourier Transform (FFT), and the absolute power of the data was then calculated in Delta (1-4 Hz), Theta (4-8 Hz), Alpha (8-13 Hz), Alpha1 (8-10), Alpha2 (10-12), and Beta (13-30Hz) frequency bands. After that, the power ratio was estimated for the theta/beta and theta/alpha frequency bands.
Statistical analysis
Pearson’s correlation was calculated using the score of SPM and the spectra of each frequency band. Spectral power was calculated in absolute and ratio style from the cleaned data. Then p- the values of each correlation score were considered and significant associations were recognized by putting a threshold of 0.05 for the p-values. This analysis was performed using the statistical analysis toolbox of MATLAB 2015 (www.mathworks.com). Then, the group comparison of correlation values was performed using the cocor package (Diedenhofen & Musch, 2015).
3. Results
Detailed information on significant results (P<0.05) for absolute and ratio power are presented in Tables 2-7 and Figures 2-6, and the whole pattern analysis is shown in Table 8 and Figure 7.

Correlation maps were scaled between -1 and 1. Only significant correlations (P<0.05) were plotted. The hot color indicates a positive and cold color shows a negative correlation.

Association between EEG power spectrum and IQ score
Absolute power
Figure 2 and Table 2 present the significant correlation between absolute power and intelligence in ADHD subtypes and healthy control groups. the combined ADHD group showed significant positive correlations between IQ scores mainly at P4 in the alpha band, and at Fp1, C3, and T5 in the beta band. Negative correlations in this group were also found between IQ score and delta power at F8, and theta power at Cz. The inattentive ADHD group showed a significant positive correlation between IQ and delta power at F3 and alpha power at P4. A negative correlation in this group was also observed between IQ score and delta power at C4, and theta power at T3. ADHD-hyperactive/impulsive group showed a positive correlation between IQ score and theta power at Cz and a negative correlation between IQ score and delta power at T3, and alpha power at C4. The healthy control group did not show any negative correlation between IQ and EEG bands, while positive correlations were observed between IQ score and theta power at F3 and F7, and apha1 power at T5 and O1.
The association between ratio power (theta/alpha, theta/beta) and intelligence score is presented in Table 3 and Figure 3.

The ADHD-combined group showed negative correlations between IQ score and theta/alpha at Cz, and O2 as well as theta/beta at F3, and Cz areas. The inattentive ADHD group revealed a positive correlation between IQ score and theta/beta at Fp2, and a negative correlation between IQ score and theta/alpha at P4. The hyperactive ADHD group and the healthy control group did not show any significant correlation between IQ score and the above-mentioned power ratios.

Differential patterns of the association between IQ scores and EEG power spectrum in ADHD subtypes and healthy control groups
Topographical differences in the association between IQ score and absolute power in various frequency bands in ADHD subtypes were compared with the healthy control group (Table 4 and Figure 4).

Compared to the healthy control group, ADHD-combined cases showed increased alpha power at P3 and decreased delta at F8 and Fz, and theta at F3 and Cz, and alpha1 power at F3, C4, T4 locations. ADHD-inattentive cases revealed increased theta power at F8 and T3, increased alpha power at P4, and decreased beta power at P3. In addition, ADHD-hyperactive cases also showed decreased theta power at F8 and Fz, decreased alpha power at T5, and decreased power of the lower alpha band at F4 and C4 areas.

Subsequently, a comparison between the patterns of ADHD subtypes was also performed and the results are shown in Table 5 and Figure 5.

Elevated alpha 2 power at F7 and Cz, decreased delta power at F8 and T6, and decreased theta power at O1 were observed in ADHD-combined cases as compared to the ADHD-inattentive group. Moreover, the ADHD-combined group showed increased alpha power at C4 and T5, increased beta power at Fp2, F3, and O1, and decreased theta power at Cz compared to the ADHD-hyperactive group. In addition, the ADHD-inattentive group was also compared to the ADHD-hyperactive group and presented increased theta power at F7 and alpha power at T5, and lower alpha power at F4 and beta power at O1.

Topographical differences in the association between intelligence and patterns of ratio power for ADHD subtypes as well as control group are presented in Tables 6 and 7 and Figure 6.

In comparison with the healthy control group, the ADHD-combined group showed decreased theta/alpha at f3, cz, o1, and o2, and decreased theta/beta at f3, c3, and cz compared to the control group. The ADHD-inattentive group also showed decreased theta/alpha at f3, and the ADHD-hyperactive group presented decreased theta/beta at f3 (Table 6 and Figure 6).

In addition, the ADHD-combined group compared to the ADHD-inattentive present ed decreased theta/alpha at o1 and theta/beta at cz. A decrease in theta/beta at cz was also observed in the ADHD-combined group compared to the ADHD-hyperactive group (Table 7 and Figure 6).

In the second phase of analysis, comparisons of whole-brain patterns of the association between IQ scores and EEG power were performed to discriminate patterns of EEG power related to IQ scores in ADHD subtypes from healthy controls (Table 8 and Figure 7).


The ADHD-combined group revealed the increased correlation between IQ score and whole-brain pattern of beta power, and the decreased correlation between IQ score and whole-brain pattern of power at theta and alpha1 bands. The inattentive ADHD group showed no significant differences, and the hyperactive/impulsive ADHD group showed a decreased correlation between IQ score and whole-brain pattern of power at theta, alpha1, and beta bands compared to the healthy control group.
In addition, comparisons of ADHD subtypes also showed that the combined ADHD group compared to the ADHD-inattentive group presented an increased correlation between IQ score and whole-brain pattern of power at alpha2 and beta bands, while the correlation decreased at alpha1. The combined ADHD group compared to the hyperactive ADHD group showed an increased correlation at alpha and beta bands. The inattentive ADHD groupcompared to the hyperactive ADHD group showed an increased correlation between alpha and alpha1 and IQ scores.
4. Discussion
The present study primarily investigated the association between brain functions based on absolute, and ratio power of EEG, and IQ scores in various individuals within a normal range of IQ. The association pattern was then compared between three ADHD subtypes and a healthy control group. The absolute and ratio powers were calculated in the conventional EEG frequency bands, including delta, theta, and alpha with sub-bands alpha1 and alpha2, and beta. Then, the association between IQ score and EEG powers was compared within the groups. Calculation of the absolute power index provides a reliable method to differentiate clinical and normal groups (Dumermuth & Molinari, 1987). This study also represented theta/alpha and theta/beta power ratios because these ratio powers can differentiate ADHD individuals from healthy controls (Clarke, et al., 2002).
The association between IQ score and EEG power has shown cognitive implication, which is discussed here. In our healthy control group, the association between IQ score and EEG bands showed a pattern of lateralization, more significant in the left hemisphere (frontal, temporal and occipital regions). Several studies have associated the left hemisphere with intelligence and working memory (Basso, et al., 1982) (Paulraj, et al., 2018) (Basso, et al., 1981). Our findings in the healthy control group also showed a significant association between IQ score and EEG power at theta and alpha bands, which is nicely fit with the previous studies (Klimesch, 1999a), (Yılmaz, et al., 2014). The association was mainly observed in the frontal and temporal regions. The frontal region is involved in many functions, such as cognition, emotional behavior, memory, and language (Mundy-Castle & Nelson, 1960) and the temporal region has been shown to associate with the vocabulary score of the Wechsler test (Dobbins & Russell, 1990) and arithmetic score as well (Inouye, et al., 1993).
Nevertheless, the pattern of association between the IQ score of the Ravan test and EEG power was completely different in ADHD individuals compared to the healthy control group. Our ADHD group showed a decreased association between EEG bands and IQ scores acquired by the Ravan test compared to the healthy control group. The Ravan test mainly estimates novel problem-solving, which is defined as solving problems that cannot be answered directly by referring to long-term knowledge but instead requires analytic or fluid reasoning (Prabhakaran et al., 1997). The network of brain regions involved in fluid reasoning is comprised of the frontopolar, middle and inferior frontal gyri, and parietal region (Rapport et al., 2008). These regions, more specifically the frontal, are also associated with working memory, which is weak in ADHD cases (Rapport et al., 2008). Therefore, decreased association between EEG bands and IQ score at frontal regions in our ADHD groups nicely fit with the deficiency of working memory and fluid reasoning acquired by the Raven test in past studies (Tamm & Juranek, 2012). In addition, our finding has implications in terms of the default mode network (DMN). This network is comprised of the medial prefrontal, precuneus, and angular gyrus (Raichle et al., 2001). DMN, which is typically activated during resting state conditions and deactivated during task performance (Broyd et al., 2009), was reported to be an underlying cause of inadequate performance in ADHD (Sonuga-Barke & Castellanos, 2007). Decreased correlation between IQ and EEG bands in our ADHD individuals compared to the healthy control group in the anterior region, plays an important role in working memory and attention (Castellanos et al., 2008), which may imply deficits in the anterior part of default mode network (DMN) in ADHD patients as reported in previous studies (Castellanos et al., 2008). In this study, the patterns of correlation between IQ score and EEG power in ADHD-combined and ADHD-inattentive were similar. This finding is corroborated by former studies, suggesting similar EEG abnormality in combined and inattentive subtypes (Clarke et al., 2001a). However, the pattern for our hyperactive ADHD cases is different from two other subtypes, which can be due to the small sample size of this group in the current study.
On the other hand, the ADHD individuals revealed a decreased association between IQ score and power of EEG at the theta band. While the association between IQ score and alpha power increased compared to the healthy control group. This finding shows the existence of a compensatory mechanism such that decreased theta can be compensated by increased alpha in the ADHD group to keep adequate intellectual function. The combined ADHD group also showed a positive association at the beta band. It has been shown that active attention and psychomotor (Polunina & Davydov, 2006) have been related to brain activities at the beta band. Therefore, a positive association between EEG beta power and intelligence, in our findings, seems to be a logical pattern. IQ and beta power were highly associated compared to that of alpha in our combined ADHD group. We think this finding of the combined group is in the same line with Webster’s theory who suggested that in the condition of serious mental activity beta replaced alpha’s place (Webster, 1978).
Moreover, unlike the healthy control group, no specific lateralization pattern was found in ADHD subtypes. The recent finding may provide additional support for the presence of a compensatory mechanism such that deficiencies in the right hemisphere are compensated by the left hemisphere to have adequate intellectual performance.
Our findings of the different association between EEG bands and IQ between the healthy controls and the ADHD groups were not limited to some specific brain regions and significant differences were also observed in the global pattern extracted from the whole brain regions. Decreased theta in all subtypes of ADHD has been shown in all brain regions. The differential patterns of associations in local and global measures potentially imply that a dynamic change in the brain’s regional functions towards increasing the alpha (or beta) band activities and decreasing the laterality effect could be a compensatory mechanism to maintain a normal intelligence score in ADHD individuals.
At ratio power, our results showed a decreased theta/alpha ratio in the ADHD group (all subtypes) compared to the healthy control group. Assuming that the theta/alpha power ratio has been introduced as a criterion for intelligence (Markowitsch & Pritzel, 1985), it is expected that the theta/alpha ratio should not be different in IQ-matched individuals. While our findings put a question mark on the validity of the above-mentioned criteria for intelligence in ADHD individuals.
5. Conclusion
Significant changes observed in the score of association between IQ and EEG power in the ADHD individuals compared to the IQ-matched healthy control group suggests the existence of a compensatory mechanism to hold a suitable cognitive performance (IQ) in the ADHD subjects. We conclude that the dynamic of the brain activities in addition to the specific brain structure plays a great role in intelligence. The discrepancy of theta/alpha ratio in groups with a similar IQ score may suggest a weakness of this marker for intelligence. We hope that these findings could provide additional information to pave the way to a better understanding of the relationship between brain functions and intelligence. Extra information have been provided in tables (9-11) and figures (6-9).





Limitation
The results of this study had a number of limitations. First, it was limited to a few numbers of hyperactivity/impulsivity ADHD subjects compared to other subtypes of the ADHD and control group. Second, the study focused on the age range of 8-12 years. Because EEG activity shifts from less to high-frequency bands during maturation, a more detailed developmental analysis may provide a better insight into the relationship between intelligence and EEG oscillations. Furthermore, the study was performed in the eyes-close resting state; therefore, further investigation on the task-based EEG will be helpful to strengthen the results. Although the findings are inspiring, more studies are required to test the reliability and validity of the results.
Ethical Considerations
Compliance with ethical guidelines
The subjects were assessed after obtaining the in -formed consent signed by the parents, and the research protocol was approved by the Ethics Committee of Iran University of Medical Sciences.
Funding
This research was supported by grants from the Cog-nitive Sciences and Technologies Council of Iran (Grant No.: 1948).
Authors' contributions
All authors contributed in designing, running, and writing all parts of the research.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
We would like to thank Reza Rostami, -Masoud Nosrat-abadi, and their colleagues for their generous assistance in the data collection. We also would like to thank the Iranian Council of Cognitive Science and Technologies for financial support on this study.
References
Anokhin, A. P., Lutzenberger, W., & Birbaumer, N. (1999).Spatiotemporal organization of brain dynamics and intelligence: an EEG study in adolescents. International Journal of Psychophysiology, 33(3), 259-273. [PMID]
Arns, M., Conners, C. K., & Kraemer, H. C. (2013). A decade of EEG theta/beta ratio research in ADHD: A meta-analysis. Journal of Attention Disorders, 17(5), 374-383. [DOI:10.1177/1087054712460087] [PMID]
Basso, A., Capitani, E., Luzzatti, C., & Spinnler, H. (1981). Intelligence and left hemisphere disease. The role of aphasia, apraxia and size of lesion. Brain: A Journal of Neurology, 104(Pt 4), 721-734. [DOI:10.1093/brain/104.4.721] [PMID]
Basso, A., Spinnler, H., Vallar, G., & Zanobio, M. E. (1982). Left hemisphere damage and selective impairment of auditory verbal short-term memory. A case study. Neuropsychologia, 20(3), 263-274. [DOI:10.1016/0028-3932(82)90101-4] [PMID]
Bloemsma, J. M., Boer, F., Arnold, R., Banaschewski, T., Faraone, S. V., & Buitelaar, J. K., et al. (2013). Comorbid anxiety and neurocognitive dysfunctions in children with ADHD. European Child & Adolescent Psychiatry, 22(4), 225–234. [DOI:10.1007/s00787-012-0339-9] [PMID]
Bresnahan, S. M., & Barry, R. J. (2002). Specificity of quantitative EEG analysis in adults with attention deficit hyperactivity disorder. Psychiatry Research, 112(2), 133-144. [DOI:10.1016/S0165-1781(02)00190-7] [PMID]
Broyd, S. J., Demanuele, C., Debener, S., Helps, S. K., James, C. J., & Sonuga-Barke, E. J. (2009). Default-mode brain dysfunction in mental disorders: A systematic review. Neuroscience and Biobehavioral Reviews, 33(3), 279–296. [DOI:10.1016/j.neubiorev.2008.09.002] [PMID]
Burgess, G. C., Gray, J. R., Conway, A. R. A., & Braver, T. S. (2011). Neural mechanisms of interference control underlie the relationship between fluid intelligence and working memory span. Journal of Experimental Psychology. General, 140(4), 674–692. [PMID]
Cabeza, R., & Nyberg, L. (2000). Imaging cognition II: An empirical review of 275 PET and fMRI studies. Journal of cognitive neuroscience, 12(1), 1-47. [DOI:10.1162/08989290051137585]
Castellanos, F. X., Margulies, D. S., Kelly, C., Uddin, L. Q., Ghaffari, M., & Kirsch, A., et al. (2008). Cingulate-precuneus interactions: A new locus of dysfunction in adult attention-deficit/hyperactivity disorder. Biological Psychiatry, 63(3), 332-337. [PMID] [PMCID]
Chabot, R. J., & Serfontein, G. (1996). Quantitative electroencephalographic profiles of children with attention deficit disorder. Biological Psychiatry, 40(10), 951-963. [DOI:10.1016/0006-3223(95)00576-5] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2001). Age and sex effects in the EEG: Differences in two subtypes of attention-deficit/hyperactivity disorder. Clinical Neurophysiology, 112(5), 815-826. [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2001). Electroencephalogram differences in two subtypes of attention-deficit/hyperactivity disorder. Psychophysiology, 38(2), 212-221. [DOI:10.1111/1469-8986.3820212] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., & Selikowitz, M. (2002).EEG analysis of children with attention-deficit/hyperactivity disorder and comorbid reading disabilities. Journal of Learning Disabilities, 35(3), 276-285. [DOI:10.1177/002221940203500309] [PMID]
Clarke, A. R., Barry, R. J., McCarthy, R., Selikowitz, M., Magee, C. A., & Johnstone, S. J., et al. (2006). Quantitative EEG in low-IQ children with attention-deficit/hyperactivity disorder. Clinical Neurophysiology, 117(8), 1708-1714. [PMID]
Deary, I. J., Penke, L., & Johnson, W. (2010). The neuroscience of human intelligence differences. Nature Reviews Neuroscience, 11(3), 201–211. [DOI:10.1038/nrn2793] [PMID]
Diedenhofen, B., & Musch, J. (2015). Cocor: A comprehensive solution for the statistical comparison of correlations. Plos One, 10(3), e0121945. [PMID] [PMCID]
Dietrich, A. (2004). The cognitive neuroscience of creativity. Psychonomic Bulletin & Review, 11(6), 1011-1026. [DOI:10.3758/BF03196731] [PMID]
Dobbins, C., & Russell, E. W. (1990). Left temporal lobe brain damage pattern on the Wechsler Adult Intelligence Scale. Journal of Clinical Psychology, 46(6), 863-868. [PMID]
Dumermuth, G., & Molinari, L. (1987). Spectral analysis of the EEG. Some fundamentals revisited and some open problems. Neuropsychobiology, 17(1-2), 85–99. [PMID]
Fischer, F. U., Wolf, D., Scheurich, A., & Fellgiebel, A. (2014). Association of structural global brain network properties with intelligence in normal aging. PloS One, 9(1), e86258. [PMID] [PMCID]
Fogel, S. M., Nader, R., Cote, K. A., & Smith, C. T. (2007). Sleep spindles and learning potential. Behavioral Neuroscience, 121(1), 1-10. [DOI:10.1037/0735-7044.121.1.1] [PMID]
Geake, J. G., & Hansen, P. C. (2005). Neural correlates of intelligence as revealed by fMRI of fluid analogies. Neuroimage, 26(2), 555-564. [PMID]
Giannitrapani, D., & Liberson, W. (1985). The electrophysiology of intellectual functions. Basel: Karger. [Link]
Gottfredson, L. S. (1997). Mainstream science on intelligence: An editorial with 52 signatories, history, and bibliography. Intelligence, 24(I), 13-23. [Link]
Gray, J. R., Chabris, C. F., & Braver, T. S. (2003). Neural mechanisms of general fluid intelligence. Nature Neuroscience, 6(3), 316-322. [DOI:10.1038/nn1014] [PMID]
Haier, R. J., & Jung, R. E. (2008). Brain imaging studies of intelligence and creativity: What is the picture for education? Roeper Review, 30(3), 171-180. [DOI:10.1080/02783190802199347]
Hearne, L. J., Mattingley, J. B., & Cocchi, L. (2016). Functional brain networks related to individual differences in human intelligence at rest. Scientific Reports, 6, 32328. [PMID] [PMCID]
Heilman, K. M., Nadeau, S. E., & Beversdorf, D. O. (2003). Creative innovation: Possible brain mechanisms. Neurocase, 9(5), 369-379. [DOI:10.1076/neur.9.5.369.16553] [PMID]
Inouye, T., Shinosaki, K., Iyama, A., & Matsumoto, Y. (1993).Localization of activated areas and directional EEG patterns during mental arithmetic. Electroencephalography and Clinical Neurophysiology, 86(4), 224-230. [PMID]
Jung, R. E., & Haier, R. J. (2007). The Parieto-Frontal Integration Theory (P-FIT) of intelligence: Converging neuroimaging evidence. Behavioral and Brain Sciences, 30(2), 135-154. [PMID]
Kane, M. J., & Engle, R. W. (2002). The role of prefrontal cortex in working-memory capacity, executive attention, and general fluid intelligence: An individual-differences perspective. Psychonomic Bulletin & Review, 9(4), 637-671. [DOI:10.3758/BF03196323] [PMID]
Kang, J. S., & Lee, M. (2015). Characteristics of EEG signals during tough and easy linguistic problem solving. Paper presented at: The Brain-Computer Interface (BCI), Gangwon, Korea, 12-14 January 2015. [DOI:10.1109/IWW-BCI.2015.7073026]
Kitsune, G. L., Cheung, C. H., Brandeis, D., Banaschewski, T., Asherson, P., & McLoughlin, G., et al. (2015). A matter of time: The influence of recording context on EEG spectral power in adolescents and young adults with ADHD. Brain Topography, 28(4), 580-590. [PMID] [PMCID]
Klimesch, W. (1999a). EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Research Reviews, 29(2-3), 169–195. [DOI:10.1016/S0165-0173(98)00056-3] [PMID]
Klimesch, W. (1999). EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Research Reviews, 29(2-3), 169-195. [DOI:10.1016/S0165-0173(98)00056-3] [PMID]
Klimesch, W., Schimke, H., & Pfurtscheller, G. (1993). Alpha frequency, cognitive load and memory performance. Brain Topography, 5(3), 241-251. [DOI:10.1007/BF01128991] [PMID]
Loo, S. K., Hale, S. T., Hanada, G., Macion, J., Shrestha, A., & McGough, J. J., et al. (2010). Familial clustering and DRD4 effects on electroencephalogram measures in multiplex families with attention deficit/hyperactivity disorder. Journal of the American Academy of Child & Adolescent Psychiatry, 49(4), 368-377. [PMID]
Markowitsch, H. J., & Pritzel, M. (1985). The neuropathology of amnesia. Progress in Neurobiology, 25(3), 189-287. [DOI:10.1016/0301-0082(85)90016-4] [PMID]
Marosi, E., Rodríguez, H., Harmony, T., Yañez, G., Rodrìguez, M., & Bernal, J., et al. (1999). Broad band spectral EEG parameters correlated with different IQ measurements. The International Journal of Neuroscience, 97(1-2), 17–27. [PMID]
Martín-Loeches, M., Muñoz-Ruata, J., Martínez-Lebrusant, L., & Gómez-Jarabo, G. (2001). Electrophysiology and intelligence: The electrophysiology of intellectual functions in intellectual disability. Journal of Intellectual Disability Research : JIDR, 45(Pt 1), 63–75. [DOI:10.1111/j.1365-2788.2001.00292.x] [PMID]
Moretti, D. V. (2015). Theta and alpha EEG frequency interplay in subjects with mild cognitive impairment: Evidence from EEG, MRI, and SPECT brain modifications. Frontiers in Aging Neuroscience, 7, 31. [PMID] [PMCID]
Mundy-Castle, A., & Nelson, G. (1960). Intelligence, personality and brain rhythms in a socially isolated community. Nature, 185(4711), 484-485. [DOI:10.1038/185484a0] [PMID]
Mundy‐Castle, A. (1958). Electrophysiological correlates of intelligence. Journal of Personality, 26(2), 184-199. [DOI:10.1111/j.1467-6494.1958.tb01579.x] [PMID]
Narr, K. L., Woods, R. P., Thompson, P. M., Szeszko, P., Robinson, D., & Dimtcheva, T., et al. (2006). Relationships between IQ and regional cortical gray matter thickness in healthy adults. Cerebral cortex (New York, N.Y. : 1991), 17(9), 2163–2171. [PMID]
Neubauer, A. C., Grabner, R. H., Fink, A., & Neuper, C. (2005).Intelligence and neural efficiency: Further evidence of the influence of task content and sex on the brain-IQ relationship. Cognitive Brain Research, 25(1), 217-225. [DOI:10.1016/j.cogbrainres.2005.05.011] [PMID]
Paulraj, S. R., Schendel, K., Curran, B., Dronkers, N. F., & Baldo, J. V. (2018). Role of the left hemisphere in visuospatial working memory. Journal of Neurolinguistics, 48, 133–141. [PMID] [PMCID]
Polunina, A. G., & Davydov, D. M. (2006). EEG correlates of Wechsler Adult Intelligence Scale. The International Journal of Neuroscience, 116(10), 1231–1248. [PMID]
Prabhakaran, V., Smith, J. A., Desmond, J. E., Glover, G. H., & Gabrieli, J. D. (1997). Neural substrates of fluid reasoning: An fMRI study of neocortical activation during performance of the Raven’s Progressive Matrices Test. Cognitive Psychology, 33(1), 43-63. [DOI:10.1006/cogp.1997.0659] [PMID]
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences, 98(2), 676-682. [PMID] [PMCID]
Rapport, M. D., Alderson, R. M., Kofler, M. J., Sarver, D. E., Bolden, J., & Sims, V. (2008). Working memory deficits in boys with attention-deficit/hyperactivity disorder (ADHD): The contribution of central executive and subsystem processes. Journal of Abnormal Child Psychology, 36(6), 825-837. [PMID]
Raven, J. C. (2000). Manual for Raven’s progressive matrices and vocabulary scales. San Antonio: Harcourt Assessment. [Link]
Raven, J. (2000). The Raven’s progressive matrices: Change and stability over culture and time. Cognitive Psychology, 41(1), 1-48. [DOI:10.1006/cogp.1999.0735] [PMID]
Reis, P. M., Hebenstreit, F., Gabsteiger, F., von Tscharner, V., & Lochmann, M. (2014). Methodological aspects of EEG and body dynamics measurements during motion. Frontiers in Human Neuroscience, 8, 156. [PMID] [PMCID]
Reynolds, C. R., & Brown, R. T. (1984). Bias in mental testing. In C. R. Reynolds, & R. T. Brown (eds.), Perspectives on bias in mental testing (pp. 1-39). Boston: Springer. [DOI:10.1007/978-1-4684-4658-6_1]
Schacter, D. L. (1977). EEG theta waves and psychological phenomena: A review and analysis. Biological Psychology, 5(1), 47-82. [DOI:10.1016/0301-0511(77)90028-X] [PMID]
Song, M., Zhou, Y., Li, J., Liu, Y., Tian, L., & Yu, C., et al. (2008). Brain spontaneous functional connectivity and intelligence. Neuroimage, 41(3), 1168-1176. [PMID]
Sonuga-Barke, E. J., & Castellanos, F. X. (2007). Spontaneous attentional fluctuations in impaired states and pathological conditions: A neurobiological hypothesis. Neuroscience & Biobehavioral Reviews, 31(7), 977-986. [DOI:10.1016/j.neubiorev.2007.02.005] [PMID]
Stankova, E. P., & Myshkin, I. Y. (2016). Association between individual EEG characteristics and the level of intelligence. Moscow University Biological Sciences Bulletin, 71(4), 256-261. [DOI:10.3103/S0096392516040118]
Tamm, L., & Juranek, J. (2012). Fluid reasoning deficits in children with ADHD: Evidence from fMRI. Brain Research, 1465, 48-56. [PMID] [PMCID]
Tang, C. Y., Eaves, E. L., Ng, J. C., Carpenter, D. M., Mai, X., & Schroeder, D. H., et al. (2010). Brain networks for working memory and factors of intelligence assessed in males and females with fMRI and DTI. Intelligence, 38(3), 293-303. [DOI:10.1016/j.intell.2010.03.003]
Thatcher, R. W., North, D., & Biver, C. (2007). Intelligence and EEG current density using low‐resolution electromagnetic tomography (LORETA). Human Brain Mapping, 28(2), 118-133. [PMID]
Thatcher, R. W., North, D., & Biver, C. (2005). EEG and intelligence: Relations between EEG coherence, EEG phase delay and power. Clinical Neurophysiology, 116(9), 2129-2141. [PMID]
Thatcher, R. W., North, D. M., & Biver, C. J. (2008). Intelligence and EEG phase reset: A two compartmental model of phase shift and lock. Neuroimage, 42(4), 1639-1653. [DOI:10.1016/j.neuroimage.2008.06.009] [PMID]
Webster, J. G., & Clark, J. W. (2010). Medical instrumentation: Application and design. New Jersey: John Wiley & Sons. [Link]
Webster, J. G. (1978). Medical instrumentation-application and design. Journal of Clinical Engineering, 3(3), 306. [DOI:10.1097/00004669-197807000-00017]
Yılmaz, B., Korkmaz, S., Arslan, D. B., Güngör, E., & Asyalı, M. H. (2014). Like/dislike analysis using EEG: Determination of most discriminative channels and frequencies. Computer Methods and Programs in Biomedicine, 113(2), 705-713. [DOI:10.1016/j.cmpb.2013.11.010] [PMID]
Type of Study: Original |
Subject:
Cognitive Neuroscience
Received: 2020/03/5 | Accepted: 2020/09/28 | Published: 2022/11/1
Received: 2020/03/5 | Accepted: 2020/09/28 | Published: 2022/11/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |








