Volume 12, Issue 2 (March & April 2021)
BCN 2021, 12(2): 255-268 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Maneshian M, Nasirinezhad F, Mohammadi F, Behzadi M, Asadi-Shekaari M, Shabani M. Minocycline Mitigation of Tremor Syndrome and Defect of Cognitive and Balance Induced by Harmaline. BCN 2021; 12 (2) :255-268
URL: http://bcn.iums.ac.ir/article-1-1581-en.html
URL: http://bcn.iums.ac.ir/article-1-1581-en.html
Marzieh Maneshian1 

 , Farinaz Nasirinezhad1
, Farinaz Nasirinezhad1 

 , Fatemeh Mohammadi2
, Fatemeh Mohammadi2 

 , Mina Behzadi2
, Mina Behzadi2 

 , Majid Asadi-Shekaari2
, Majid Asadi-Shekaari2 

 , Mohammad Shabani *2
, Mohammad Shabani *2 




 , Farinaz Nasirinezhad1
, Farinaz Nasirinezhad1 

 , Fatemeh Mohammadi2
, Fatemeh Mohammadi2 

 , Mina Behzadi2
, Mina Behzadi2 

 , Majid Asadi-Shekaari2
, Majid Asadi-Shekaari2 

 , Mohammad Shabani *2
, Mohammad Shabani *2 


1- Department of Physiology, Physiological Research Center, Iran University of Medical Sciences, Tehran, Iran.
2- Intracellular Recording Lab, Neuroscience Research Center, Neuropharmacology Institute, Kerman University of Medical Sciences, Kerman, Iran.
2- Intracellular Recording Lab, Neuroscience Research Center, Neuropharmacology Institute, Kerman University of Medical Sciences, Kerman, Iran.
Full-Text [PDF 4561 kb]
| Abstract (HTML)
Full-Text:
1. Introduction
Tremor is an unintentional, rhythmic, and sinusoidal movement that repeatedly occurs in one or more body parts (Abdo, van de Warrenburg, Burn, Quinn, & Bloem, 2010). Tremor is a common disabling symptom of many motor-related disorders such as Parkinson Disease (PD), Essential Temor (ET), Multiple Sclerosis (MS), Huntington Disease (HD), and some forms of ataxia, which originates from an involuntary muscle contraction/relaxation cycle (Arjmand et al., 2015; Hughes, Daniel, Blankson, & Lees, 1993). Classic ET is defined by postural kinetic (action) tremor, involving both upper limbs, in at least 95% of patients (Elble, 2000; Whaley, Putzke, Baba, Wszolek, & Uitti, 2007). Patients with ET have other motor problems, which imply cerebellar impairment, such as gait ataxia and eye movement abnormalities (Gitchel, Wetzel, & Baron, 2013; Louis et al., 2013), as well as non-motor conditions, including cognitive, psychiatric, and sensory abnormalities (Benito-Leon, Louis, & Bermejo-Pareja, 2006; Louis, Bromley, Jurewicz, & Watner,, 2002; Ondo, Sutton, Dat Vuong, Lai, & Jankovic, 2003). The exact pathogenesis of ET is uncertain, but neuroimaging studies indicate the role of olivo-cerebellar and cerebello-thalamo-cortical pathways in the creation of the tremors (Bhalsing, Saini, & Pal, 2013). Also, Gamma Amino-Butyric Acid (GABA) abnormalities (Gironell et al., 2012), neurodegeneration associated with gliosis, Purkinje cell loss, and increase in focal axonal swellings of Purkinje cells are associated with these tremors (Louis et al., 2007).
Harmaline can induce tremors similar to tremors in ET with the postural/kinetic exhibition. Studies show that intraperitoneal administration of this substance in animal models of ET increases glutamate transmission of the olivo-cerebellar pathway with an excitotoxic effect of Purkinje cells (Beitz & Saxon, 2004). Besides cytotoxicity, tremor, and motor disorders, harmaline can cause cognitive disturbances and learning and memory deficits (Abbassian et al., 2016; Aghaei et al., 2019; Nasehi, Ketabchi, Khakpai, & Zarrindast, 2015).
Regarding the pathophysiology of ET as a neurodegenerative disorder (Louis, 2009), neuroprotective agents that benefit from immunomodulatory, anti-inflammatory, anti-apoptotic, and anti-oxidant properties may be the most effective drug in the treatment of ET (Kim & Suh, 2009). It has been proposed that minocycline has neuroprotective effects in several animal models of neurological diseases (Garrido-Mesa, Zarzuelo, & Galvez, 2013; Handforth, 2012; O’Hearn & Molliver, 1997). Experimental and clinical studies have been shown its beneficial effects in the treatment of ischemia (Yrjänheikki et al., 1999), traumatic brain injury (Mejia, Ona, Li, & Friedlander, 2001), Amyotrophic Lateral Sclerosis (ALS) (Zhu et al., 2002), PD (Jackson-Lewis et al., 2002), HD (Chen et al., 2000; Wang et al., 2003), MS (Popovic et al., 2002), and Alzheimer's Disease (AD) (Choi et al., 2007). Although the exact mechanism of minocycline neuroprotective action is unclear, several studies suggest an association between this action and the inhibition of cytochrome C involved in cell death, the inhibition of caspase expression, p38 Mitogen-Activated Protein Kinase (p38 MAPK) inhibition, increased expression of 5-lipooxygenase, and the inhibition of Poly (ADP-Ribose) Polymerase1 (PARP1) (Chu et al., 2010; Kim & Suh, 2009; Li & McCullough, 2009; Tang et al., 2007). Minocycline also leads to mitigation of glutamate-induced excitotoxicity (Pi et al., 2004) and mitigation of NMDA-induced (N -methyl-D-aspartate receptor) neurotoxicity by the reduction in NMDA-induced Ca2+ inward current and mitochondria Ca2+ uptake (Garcia-Martinez et al., 2010).
Because of the minocycline ameliorating effect on neurodegenerative diseases, this study aimed to assess the possible neuroprotective effects of minocycline on motor and cognitive deficits induced by harmaline in the experimental rat model of ET.
2. Methods
2.1. Study animals
Male Wistar rats (weighing 100-120 g) were experimented according to the Research and Ethics Committee of Kerman University and internationally accepted principles for laboratory animal use and care. They were kept at a standard laboratory condition (12-12 h light-dark cycle) with ad libitum access to food pellets and water. All tests were performed during the light period.
2.2. Drug preparation and administration
Harmaline HCl and minocycline hydrochloride were purchased from Sigma-Aldrich. Both substances were dissolved in 0.9% sterile, isotonic saline on the day of administration. Before the experiments began, the rats were put into the test room for one hour to get used to the environment. The rats were randomly divided into four groups (10 rats in each group). The first group was received saline. The second group was given minocycline (90 mg/kg, IP). The third group was received a single injection of harmaline (30 mg/kg, IP). Animals in group 4 were given minocycline (90 mg/kg IP) 30 min before harmaline administration (Elewa, Hilali, Hess, Machado, & Fagan, 2006; Plane, Shen, Pleasure, & Deng, 2010). Each group went through five different behavioral studies, which were performed 30 min after harmaline injection with sequentially 15 min rest intervals among each assay in the following order: tremor score assessment, open field test, footprint, rotarod, wire grip, and passive avoidance task (Behavioral procedures timeline; Figure 1).
.jpg)
2.3. Assessment of motor activity
2.3.1. Observation
The scale of tremors was assessed by an observer who was unaware of the treatment protocol. The qualitative grading of tremor severity was based on the Arshaduddin et al. method, which is as follows: no tremor= 0, mild tremor= 1 (only the head and neck affected), moderate intermittent tremor= 2 (whole-body parts affected), moderate persistent tremor= 3 (whole-body parts and tail affected), and pronounced severe tremor= 4 (trembling and unable to mobility) (Abbassian et al., 2016; Dahmardeh, Shabani, Basiri, Kalantaripour, & Asadi-Shekaari, 2019).
2.3.2. Open field test
The locomotion and anxiety-like behaviors of the rats from each group were recorded and analyzed by an automated video tracking software (Ethovision, Noldus Technology, Netherlands) for five minutes. Each animal was individually placed in the middle of the arena of apparatus (90×90×45 [H] cm, divided into 16 small squares). The behavioral parameters consist of total time spent in the center or periphery, Total Distance Moved (TDM), speed, and the number of grooming and rearing were recorded for each rat (Mahmoudvand et al., 2015; Razavinasab et al., 2013).
2.3.3. Footprint
To evaluate the rat’s walking pattern and gait kinematic, we used a footprint-test. The hind limbs of rats were covered with a non-toxic color. The rats were then placed in a Plexiglas narrow corridor (100 cm long×10 cm height×10 cm width) ending in a dark box covered with white absorbent paper to move freely. Three consecutive strides were chosen to assess the footprint so that the distance between the steps of each side (the middle of each foot to the middle of the next foot) was recorded as the length of the step and the distance between the center of one foot with the opposite foot was recorded as the step width, and the mean value of these data was analyzed. The initiating and finishing steps were also excluded from the study. Before testing, the rats were put on the runway to get acquainted with the procedure (Mohammadi, Esfahlani, & Shabani, 2019).
2.3.4. Accelerating rotarod assay: A Motor Performance Test
The rotarod test was used to check the balance and coordination. To do this test, we placed each rat on a rotating rod for 5 minutes for habituation before the test began, and the velocity of the rotation accelerated from 10 revolutions per min (rpm) to 60 rpm during 5 minutes. After habituation and rest, three trials were performed, and the average interval between placing the rat on the rod and falling from the rod was measured and recorded. The total test time for each rat was 300 s, and 5 min rest was between each trial (Nazeri et al., 2015).
2.3.5. Wire Grip Test
Wire grip test was used to measure muscle strength and balance by placing the rat on both forepaws on a steel wire (80 cm long, 7 mm diameter). When the rat grasped the wire, the rat was released. The duration of the time that the rat does not fall and holds the wire was recorded. Three trials were performed with five minutes between trials, and the mean values were recorded as the final result (Haghani, Shabani, & Moazzami, 2013).
2.3.6. Passive Avoidance Test
The passive avoidance test evaluates learning and memory function in rats. The apparatus size was 40 cm long, 20 cm width, and 20 cm height. It comprised two equal size sections (light and dark) with a grid floor, divided by a sliding door. The electrical stimulator was embedded in the dark floor partition (50 Hz, 0.5 mA, 2 ms). This test consists of three trials (adaptation trial, training trial, retention trial). In the learning phase of the test, each animal was first habituated to the test equipment. In the adaptation trial, the rat was placed in the light cavity facing the door, and after 10 seconds, the door was opened, and the time the animal entered the dark chamber was recorded. If, after 120 seconds, the rat was reluctant to cross the dark section, it was deleted from the test.
Two hours later, the training trial was done. The animal was returned to the light compartment, the door opened, and the animal was allowed to move to the dark chamber. Upon arrival, the sliding door was closed, and a foot-shock was given. After 20 seconds, the animal was moved to its home cage. After 5 minutes, this step was repeated, and if the rat entered the dark part, the shock was given again. This step continued until the rat learned the training. If it stayed on for about 300 seconds in the light part, the training was ended. The number of shocks was recorded as a variable. After 24 hours, the memory retention of rats was measured. The rat was placed in the lightbox, and after 10 seconds, the door was opened. The time it took for the animal to enter the dark part for the first time was recorded as Step Through Latency (STL). Cut off point of this trial was 300 seconds (Shabani, Haghani, Sheibani, & Janahmadi, 2009).
2.4. Morphological analysis
Seven days after treatment, five rats from each group were randomly selected for light microscopic study. The animals were guillotined under deep anesthesia (ketamine-xylazine). Then, they were transcardially perfused with normal saline and fixed with 4% paraformaldehyde solution. Next, their cerebellums and brain stems were carefully extracted from the skull and post-fixed in 10% buffered formaldehyde for 48 hours. In short, the tissues were dehydrated, cleared, embedded in paraffin, and finally, 5-μm sections (coronal sections at the vermal level of cerebellum and rostro-caudal of the brainstem) were obtained for histological study. The sections were deparaffinized and were hydrated, followed by staining with Hematoxylin and Eosin (H&E) (Rahimi Shourmasti et al., 2012). For the ultrastructural evaluation, two rats were randomly selected from each group and intracardially perfused with 4% glutaraldehyde in 0.1 M Phosphate Buffer Solution (PBS). Then, their brains were extracted carefully, and target parts were cut into 1×1-mm fragments. The tissues were fixed in 4% glutaraldehyde PBS for 24 hours, followed by post-fixation in 1% osmium tetroxide in PBS for 1 hour. Tissue processing was performed according to the standard method. Briefly, this process consists of washing, dehydrating with ethanol, infiltrating with propylene oxide twice for 30 minutes, infiltrating and embedding with resin (Epon-812). Ultra-cut (50-80 nm) was obtained using Leica ultramicrotome. The sections were placed on copper grids and stained with uranyl acetate and lead citrate. They were analyzed using a transmission electron microscope (Zeiss EM10, Germany) (Asadi-Shekaari et al., 2011).
2.5. Data analysis
We used a 1-way ANOVA test followed by Tukey’s (as a post hoc test) for analyzing the data. The standard error of the mean (Mean±SEM) expresses the results. The significance of the difference between the mean of groups was considered with P<0.05.
3. Results
3.1. Behavioral studies
3.1.1. Effect of minocycline on the scale of tremor
The severity of tremors increased in harmaline groups (F3, 36= 29.7, P<0.001, Figure 2) compared to the saline and minocycline groups.
.jpg)
Minocycline therapy resulted in a significant reduction in the harmaline-induced tremor score (F3, 36=6.3, P<0.05, Figure 2A) compared to the harmaline alone group.
3.1.2. The effect of minocycline on gait disturbance
Footprint assay showed that harmaline markedly increased hind paw stride width, compared with the saline (F3, 36=13.6, P<0.001) and minocycline (P<0.01) groups (Figure 2B). There was a significant step width decrease in the harmaline + minocycline group compared with the harmaline group (F3, 36=9.6, P<0.05, Figure 2B). However, there was no significant difference among harmaline + minocycline and saline groups. The effect of minocycline treatment was not detected in the left (Figure 2C) and right (Figure 2D) step lengths.
3.1.3. Effect of minocycline on locomotors and anxiety-like behaviors
The total distance moved in the harmaline group was significantly lower compared with saline (F3, 36=9.1, P<0.01) and minocycline (P<0.05) groups. Besides, this parameter in the harmaline + minocycline group was also significantly lower compared with the saline group (F3, 36=4.9, P<0.05, Figure 3A).
.jpg)
The velocity of animals in all groups significantly decreased compared with the saline group (F3, 36=16.4, P<0.001). Also, harmaline and harmaline + minocycline groups significantly decreased compared with the minocycline group in the velocity of movement (P<0.05, Figure 3B). Mobility duration in the harmaline (P<0.05) and harmaline + minocycline (F3, 36= 8.6, P<0.01) groups compared with the saline group, significantly decreased. But no significant difference was observed between other groups (Figure 3C). Time spent in the periphery of the box was not significantly different between all groups (Figure 3D).
3.1.4. The effects of minocycline pretreatment on balance function and muscle strength
Our results showed that time of staying on the rod in the rotarod test in the harmaline group compared with the saline group (F3, 36=21.3, P<0.001) and minocycline group (F3, 36=23.8, P<0.001) had a significant decrease. A significant difference was detected between the harmaline + minocycline group compared to the saline group (F3, 36=11.6, P<0.01) as well. No significant difference was observed between other groups (Figure 4A).
.jpg)
In wire grip apparatus, rats of the harmaline group showed a decrease in falling time compared with the saline group (F3, 36=19.3, P<0.001), and minocycline group (F3, 36=7.7, P<0.01). Rats of the minocycline + harmaline group spent a shorter duration on the wire grip compared with the saline group (F3, 36=9.6, P<0.01) and minocycline group (P<0.05, Figure 4B).
3.1.5. Effect of harmaline and minocycline on passive avoidance memory
The number of receiving shocks in the shuttle box test in the harmaline and harmaline + minocycline groups was significantly increased compared with the saline group (F3, 36=4.9, P<0.05). A significant increase was noted compared with the minocycline group (F3, 36=3.5, P<0.05, Figure 5A).
.jpg)
The time of entrance to the dark compartment in the Shuttle Box Test (STL) significantly decreased in the harmaline and harmaline + minocycline compared with the saline group (F3, 36=16.6, P<0.001). Also, a significant decrease was observed in these groups compared with the minocycline group (F3, 36=11.2, P<0.001). In harmaline + minocycline group a significant increase was seen compared with harmaline group (F3, 36=13.8, P<0.001, Figure 5B).
3.2. Histological assessment
3.2.1. Light microscopy findings
The normal morphology of Purkinje cells is shown in Figure 6A.
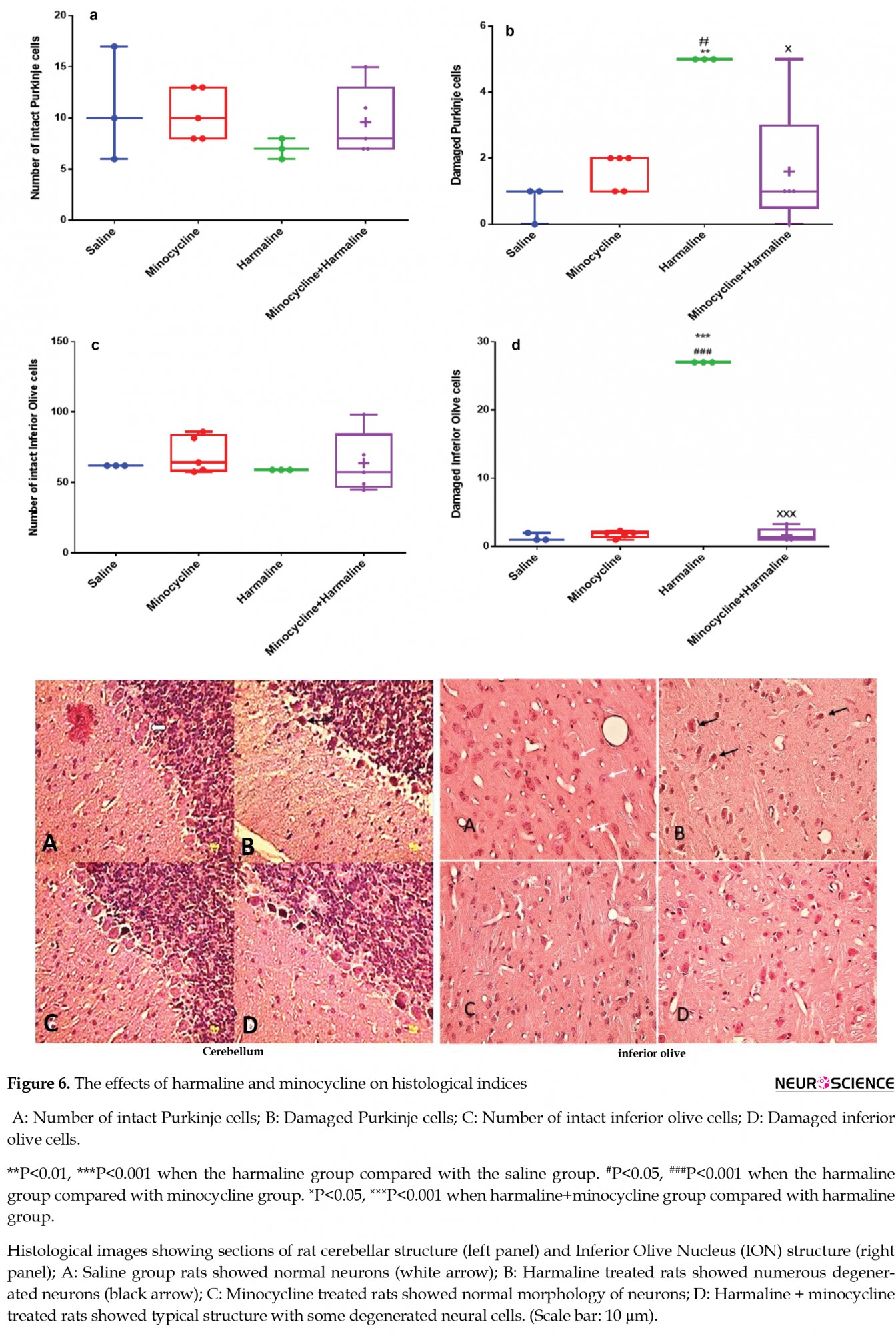
The number of degenerated Purkinje cells significantly increased in the harmaline group compared with the saline (F3, 12=5.7, P<0.01) and the minocycline groups (F3, 12=3.1, P<0.05, Figure 6B). Administration of minocycline 30 minutes before harmaline injection resulted in a significant decrease in the number of degenerated cerebellar Purkinje cells compared with the harmaline group (P<0.05, Figure 6B).
The normal morphology of inferior olive nucleus neurons is shown in Figure 6C. There was a significant increase in the number of degenerated neurons of the Inferior Olive Nucleus (ION) in the harmaline group compared with the saline (F3, 12=10.3, P<0.001) and the minocycline (P<0.001) groups. With the administration of minocycline, a significant decrease was observed in the number of damaged cells in the ION compared with the harmaline group (F3, 12=9.1, P<0.001, Figure 6D).
3.2.2. Electron microscopy findings
In the saline group, cerebellar Purkinje cells and ION neurons had normal ultrastructure, i.e. round nucleus, dispersed chromatin, and intact nucleolar membrane (Figure 7).
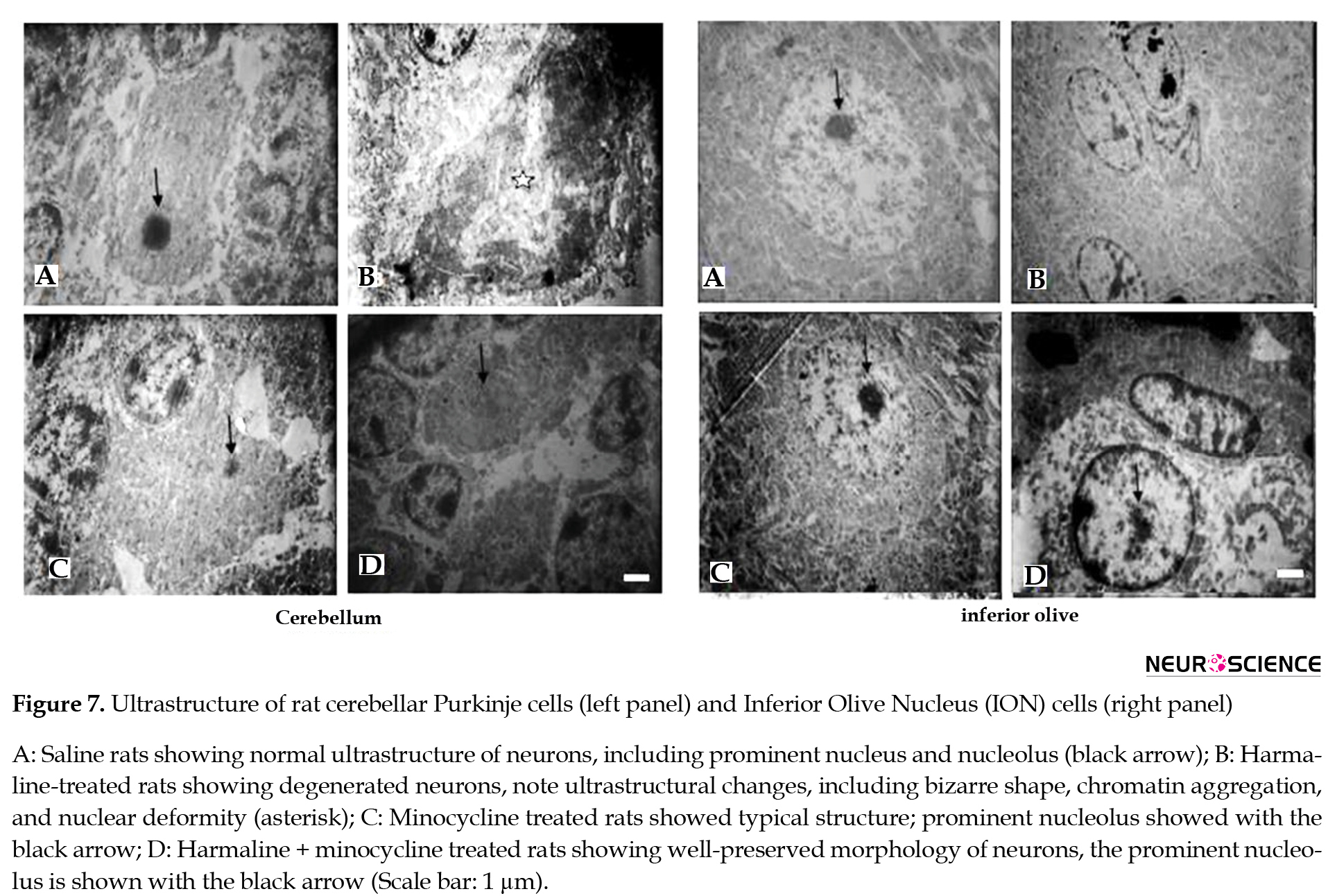
Harmaline administration resulted in ultrastructure changes in the aforementioned cells, including shrinkage and deformity of the nucleus, chromatolysis, nuclear lysis, and increased cytoplasmic density. Pretreatment by minocycline decreased these effects (Figure 7).
4. Discussion
The current study results demonstrated that pretreatment with minocycline improved tremor status and memory deficit in the rat’s model of tremor induced by harmaline, as evidenced by several behavioral tasks. However, there were no significant improvements in balance function and muscle strength.
Tremor is a sign of many neurodegenerative diseases, and several studies have shown the neuroprotective effects of minocycline in several neurodegenerative diseases like PD (Du et al., 2001), HD (Radad, Moldzio, & Rausch, 2010), AD (Fan et al., 2007; Seabrook, Jiang, Maier, & Lemere, 2006), ALS (Zhu et al., 2002), and acute injuries such as spinal cord injury (Kim & Suh, 2009). So, we decided to conduct this study to assess the neuroprotective effect of minocycline on tremors induced by harmaline. Harmaline was chosen to induce the tremor model because its underlying mechanism is excessive stimulation of climbing fiber to the cerebellum and glutamate excitotoxicity in Purkinje cells and ION (Beitz & Saxon, 2004). Many studies have confirmed that harmaline provokes generalized transient action tremors with kinetic and postural components displayed in minutes or hours (Grimaldi & Manto, 2013; Vaziri et al., 2015).
The results of our study showed that harmaline produced moderate tremors, which is consistent with previous behavioral studies (Abbassian et al., 2016; Nasehi et al., 2010). Previous research showed that harmaline performs its tremorogenesis action by raising in glutamate discharge (Llinás & Volkind, 1973), probably acting on NMDA receptors and T-type calcium channels (Handforth, 2012), and excitotoxicity damages of Purkinje cells via hyper-releasing of glutamate from climbing fibers (Choi, 1988). It has been suggested that harmaline increases rebound T-type calcium pulses in inferior olive cells (Miwa & Kondo, 2011). On the other hand, minocycline can reduce neuron excitability by blocking voltage-dependent Na+-channels and attenuates glutamatergic neurotransmission and Ca2+ signaling in the central nervous system as well as the reduction in the cellular glutamate amount related to voltage-dependent Ca2+ channels (Gonzalez et al., 2007). Hence, neuroprotection effects of minocycline on tremor results from its neuronal excitability reduction and glutamate transmission, alleviating and preventing calcium overloading in neurons.
In our study, the results of the wire grip test and rotarod did not show any improvement. This finding is maybe associated with minocycline potential for vestibular toxicity (Li et al., 2013). Minocycline has common ototoxicity side effects with vestibular symptoms such as dizziness, vertigo, ataxia, tinnitus, and other symptoms (Diguet et al., 2004). Also, the result of the open field test showed pretreatment by minocycline could not mitigate anxiety in rats, and previous studies have shown that it has had deleterious effects on motor performance, balance, coordination, and anxiety behaviors on a model of Parkinson study as well (Tikka, Fiebich, Goldsteins, Keinänen, & Koistinaho, 2001; Yang et al., 2003).
The shuttle box test showed that harmaline treatment impaired the learning phase as indicated by the increased number of shocks required before animals met the criterion, and this impairment was not affected by minocycline. When memory impairment was assessed in the shuttle box, harmaline treatment impaired acquisition of passive avoidance as demonstrated by a shorter step-through latency. However, unlike learned inhibition, minocycline partially reversed this harmaline-induced impairment.
Whereas the memory formation and retrieval are mainly associated with the hippocampus and activity of its dopaminergic receptors in the CA1 area (Hartman, Lee, Zipfel, & Wozniak, 2005; Khakpai, Nasehi, Haeri-Rohani, Eidi, & Zarrindast, 2013), so any destruction in this pathway can cause amnesia. Several mechanisms and pathways are suggested for amnesia and cognitive deficiency produced by harmaline. Some studies demonstrated that the interaction between harmaline and dopamine receptors in CA1 are involved in memory impairment (Kim, Hassler, Kurokawa, & Bak, 1970; Nasehi et al., 2015). On the other hand, some studies have reported that minocycline can reduce neurotoxicity in dopaminergic systems in hippocampus and striatum by inhibiting microglia function (Zhang et al., 2006). Therefore, it is likely that the minocycline protection effect on dopaminergic neurons is responsible for improving the memory deficit induced by harmaline administration.
In addition to behavioral assessments, the second objective of the current study was to determine the effect of minocycline administration on the possible morphological and ultrastructure alterations induced by harmaline in a rat model of ET.
Harmaline causes excitotoxicity by increasing the activation and proliferation of microglial and enhancing the release of their toxic substances, such as p38 microtubule-associated protein kinase, interleukine-1b, and nitric oxide, which leads to neuronal cell death (Tikka et al., 2001). Therefore harmaline increases cell loss in inferior olive and Purkinje cells because of glutamate increment, as was illustrated by previous studies (Handforth, 2012). Our histology results showed a positive effect of minocycline on these neurons.
Minocycline exerts its neuroprotective effects through several ways, such as alleviating the underlying neurodegenerative processes, including anti-inflammatory, anti-apoptotic, and anti-oxidant activity, and also applying against excitotoxicity, mainly by inhibition of microglial activity (Li, Yuan, & Schluesener, 2013; Tikka et al., 2001). We assume that preservation of neurons and decrement of neurodegeneration in inferior olive and Purkinje cells in our study was resulted from attenuating the microglial action of minocycline. However, we cannot elucidate if other possible mechanisms are contributed to this process as well or not.
The obtained findings demonstrated that harmaline causes tremor symptoms and impaired cognitive functions in Wistar male rats. Also, some of these impairments were reversible by minocycline. However, minocycline seems to act as a neuroprotective agent to improve tremor severity, gait width disturbance, and memory retrieval impairments induced by harmaline. Of course, more investigations are needed to evaluate the mechanism of its therapeutic effects on motor and cognitive impairments.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee (EC/KNRC/96-59) of the University of Kerman Medical Sciences, Kerman.
Funding
The paper was extracted from the MSc. thesis of the first author at the Kerman University of Medical Sciences Research Deputy and Neuroscience Research Center.
Authors' contributions
Conceptualization, methodology, and writing – original draft: Marzieh Maneshian, Farinaz Nasirinezhad, Fatemeh Mohammadi; Data collection: Mina Behzadi and Majid Asadi –shekaari; Data analysis: Mohammad Shabani. Final approval: All authors.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We appreciate the staff of Kerman University of Medical Sciences for funding the study.
References
Abbassian, H., Esmaeili, P., Tahamtan, M., Aghaei, I., Vaziri, Z., & Sheibani, V., et al. (2016). Cannabinoid receptor agonism suppresses tremor, cognition disturbances, and anxiety-like behaviors in a rat model of essential tremor. Physiology & Behavior, 164, 314-20. [DOI:10.1016/j.physbeh.2016.06.013] [PMID]
Abbassian, H., Whalley, B. J., Sheibani, V., & Shabani, M. (2016). Cannabinoid type 1 receptor antagonism ameliorates harmaline-induced essential tremor in rat. British Journal of Pharmacology, 173(22), 3196-207. [DOI:10.1111/bph.13581] [PMID] [PMCID]
Abdo, W. F., van de Warrenburg, B. P. C., Burn, D. J., Quinn, N. P., & Bloem, B. R. (2010). The clinical approach to movement disorders. Nature Reviews Neurology, 6, 29. [DOI:10.1038/nrneurol.2009.196] [PMID]
Aghaei, I., Hajali, V., Haghani, M., Vaziri, Z., Moosazadeh, M., & Shabani, M. (2019). Peroxisome proliferator-activated receptor-gamma activation attenuates harmaline-induced cognitive impairments in rats. Journal of Clinical Neuroscience, 59, 276-83. [DOI:10.1016/j.jocn.2018.11.004] [PMID]
Arjmand, S., Vaziri, Z., Behzadi, M., Abbassian, H., Stephens, G. J., & Shabani, M. (2015). Cannabinoids and Tremor Induced by Motor-related Disorders: Friend or Foe? Neurotherapeutics, 12(4), 778-87. [DOI:10.1007/s13311-015-0367-5] [PMID] [PMCID]
Asadi-Shekaari, M., Eftekhar Vaghefi, H., Ezzat Abadi Pour, M., Sheibani, V., Shams Ara, A., & Behbahani, P. (2011). Antiapoptotic effects of aspirin on CA1 pyramidal neurons in adult rats. Iranian Journal of Pathology, 6(4), 187-92. http://ijp.iranpath.org/article_8516_1349.html
Beitz, A. J., & Saxon, D. (2004). Harmaline-induced climbing fiber activation causes amino acid and peptide release in the rodent cerebellar cortex and a unique temporal pattern of Fos expression in the olivo-cerebellar pathway. Journal of Neurocytology, 33(1), 49-74. [DOI:10.1023/B:NEUR.0000029648.81071.20] [PMID]
Benito-Leon, J., Louis, E. D., & Bermejo-Pareja, F. (2006). Population-based case-control study of cognitive function in essential tremor. Neurology, 66(1), 69-74. [DOI:10.1212/01.wnl.0000192393.05850.ec] [PMID]
Bhalsing, K. S., Saini, J., & Pal, P. K. (2013). Understanding the pathophysiology of essential tremor through advanced neuroimaging: A review. Journal of the Neurological Sciences, 335(1-2), 9-13. [DOI:10.1016/j.jns.2013.09.003] [PMID]
Chen, M., Ona, V. O., Li, M., Ferrante, R. J., Fink, K. B., & Zhu, S., et al. (2000). Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nature Medicine, 6(7), 797. [DOI:10.1038/77528] [PMID]
Choi, D. W. (1988). Glutamate neurotoxicity and diseases of the nervous system. Neuron, 1(8), 623-34. [DOI:10.1016/0896-6273(88)90162-6]
Choi, Y., Kim, H. S., Shin, K. Y., Kim, E. M., Kim, M., & Kim, H. S., et al. (2007). Minocycline attenuates neuronal cell death and improves cognitive impairment in Alzheimer’s disease models. Neuropsychopharmacology, 32(11), 2393-404. [DOI:10.1038/sj.npp.1301377] [PMID]
Chu, L.-S., Fang, S. H., Zhou, Y., Yin, Y. J., Chen, W.Y., & Li, J. H., et al. (2010). Minocycline inhibits 5-lipoxygenase expression and accelerates functional recovery in chronic phase of focal cerebral ischemia in rats. Life Sciences, 86(5-6), 170-7. [DOI:10.1016/j.lfs.2009.12.001] [PMID]
Dahmardeh, N., Shabani, M., Basiri, M., Kalantaripour, T. P., & Asadi-Shekaari, M. (2019). Functional antagonism of sphingosine-1-phosphate receptor 1 prevents harmaline-induced ultrastructural alterations and caspase-3 mediated apoptosis. Malaysian Journal of Medical Sciences, 26(4), 28-38. [DOI:10.21315/mjms2019.26.4.4] [PMID] [PMCID]
Diguet, E., Fernagut, P.-O., Wei, X., Du, Y., Rouland, R., & Gross, C., et al. (2004). Deleterious effects of minocycline in animal models of Parkinson’s disease and Huntington’s disease. European Journal of Neuroscience, 19(12), 3266-76. [DOI:10.1111/j.0953-816X.2004.03372.x] [PMID]
Du, Y., Ma, Z., Lin, S., Dodel, R. C., Gao, F., & Bales, K. R., et al. (2001). Minocycline prevents nigrostriatal dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Proceedings of the National Academy of Sciences of the United States of America, 98(25), 14669-74. [DOI:10.1073/pnas.251341998] [PMID] [PMCID]
Elble, R. J. (2000). Diagnostic criteria for essential tremor and differential diagnosis. Neurology, 54(suppl 4), 2-6. [PMID]
Elewa, H. F., Hilali, H., Hess, D. C., Machado, L. S., & Fagan, S. C. (2006). Minocycline for short‐term neuroprotection. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 26(4), 515-21. [DOI:10.1592/phco.26.4.515] [PMID] [PMCID]
Fan, R., Xu, F., Previti, M. L., Davis, J., Grande, A. M., & Robinson, J. K., et al. (2007). Minocycline reduces microglial activation and improves behavioral deficits in a transgenic model of cerebral microvascular amyloid. The Journal of Neuroscience, 27(12), 3057-63. [DOI:10.1523/JNEUROSCI.4371-06.2007] [PMID] [PMCID]
Garcia-Martinez, E. M., Sanz-Blasco, S., Karachitos, A., Bandez, M. J., Fernandez-Gomez, F. J., & Perez-Alvarez, S., et al. (2010). Mitochondria and calcium flux as targets of neuroprotection caused by minocycline in cerebellar granule cells. Biochemical Pharmacology, 79(2), 239-50. [DOI:10.1016/j.bcp.2009.07.028] [PMID]
Garrido-Mesa, N., Zarzuelo, A., & Galvez, J. (2013). Minocycline: Far beyond an antibiotic. British Journal of Pharmacology, 169(2), 337-52. [DOI:10.1111/bph.12139] [PMID] [PMCID]
Gironell, A., Figueiras, F. P., Pagonabarraga, J., Herance, J. R., Pascual-Sedano, B., & Trampal, C., et al. (2012). Gaba and serotonin molecular neuroimaging in essential tremor: A clinical correlation study. Parkinsonism Relat Disord, 18(7), 876-80. [DOI:10.1016/j.parkreldis.2012.04.024] [PMID]
Gitchel, G. T., Wetzel, P. A., & Baron, M. S. (2013). Slowed saccades and increased square wave jerks in essential tremor. Tremor and Other Hyperkinetic Movements, 3, tre-03-178-4116-2. [DOI:10.5334/tohm.127] [PMID] [PMID]
Gonzalez, J. C., Egea, J., Del Carmen Godino, M., Fernandez-Gomez, F. J., Sanchez-Prieto, J., & Gandia, L., et al. (2007). Neuroprotectant minocycline depresses glutamatergic neurotransmission and Ca(2+) signalling in hippocampal neurons. European Journal of Neuroscience, 26(9), 2481-95. [DOI:10.1111/j.1460-9568.2007.05873.x] [PMID]
Grimaldi, G., & Manto, M. (2013). Assessment of tremor: Clinical and functional scales. In Grimaldi G., Manto M. (eds), Mechanisms and Emerging Therapies in Tremor Disorders. (pp. 325-340). New York, NY.: Springer. [DOI:10.1007/978-1-4614-4027-7_18]
Haghani, M., Shabani, M., & Moazzami, K. (2013). Maternal mobile phone exposure adversely affects the electrophysiological properties of Purkinje neurons in rat offspring. Neuroscience, 250, 588-98. [DOI:10.1016/j.neuroscience.2013.07.049] [PMID]
Handforth, A. (2012). Harmaline tremor: Underlying mechanisms in a potential animal model of essential tremor. Tremor and Other Hyperkinetic Movements, 2. [DOI:10.5334/tohm.108]
Hartman, R. E., Lee, J. M., Zipfel, G. J., & Wozniak, D. F. (2005). Characterizing learning deficits and hippocampal neuron loss following transient global cerebral ischemia in rats. Brain Research, 1043(1-2), 48-56. [DOI:10.1016/j.brainres.2005.02.030] [PMID]
Hughes, A. J., Daniel, S. E., Blankson, S., & Lees, A. J. (1993). A Clinicopathologic Study of 100 Cases of Parkinson’s Disease. Archives of Neurology, 50(2), 140-8. [DOI:10.1001/archneur.1993.00540020018011] [PMID]
Jackson-Lewis, V., Vila, M., Tieu, K., Teismann, P., Vadseth, C., & Choi, D. K., et al. (2002). Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine mouse model of Parkinson disease. Journal of Neuroscience, 22(5), 1763-71. [DOI:10.1523/JNEUROSCI.22-05-01763.2002] [PMID] [PMCID]
Khakpai, F., Nasehi, M., Haeri-Rohani, A., Eidi, A., & Zarrindast, M. R. (2013). Septo-hippocampo-septal loop and memory formation. Basic and Clinical Neuroscience, 4(1), 5-23. [PMCID] [PMID]
Kim, H. S., & Suh, Y. H. (2009). Minocycline and neurodegenerative diseases. Behavioural Brain Research, 196(2), 168-79. [DOI:10.1016/j.bbr.2008.09.040] [PMID]
Kim, J. S., Hassler, R., Kurokawa, M., & Bak, I. J. (1970). Abnormal movements and rigidity induced by harmaline in relation to striatal acetylcholine, serotonin, and dopamine. Experimental Neurology, 29(2), 189-200. [DOI:10.1016/0014-4886(70)90049-X]
Li, C., Yuan, K., & Schluesener, H. (2013). Impact of minocycline on neurodegenerative diseases in rodents: A meta-analysis. Reviews in the Neurosciences, 24(5), 553-62. [DOI:10.1515/revneuro-2013-0040] [PMID]
Li, J., & McCullough, L. D. (2009). Sex differences in minocycline-induced neuroprotection after experimental stroke. Journal of Cerebral Blood Flow & Metabolism, 29(4), 670-4. [DOI:10.1038/jcbfm.2009.3] [PMID] [PMCID]
Llinás, R., & Volkind, R. A. (1973). The olivo-cerebellar system: Functional properties as revealed by harmaline-induced tremor. Experimental Brain Research, 18(1), 69-87. [DOI:10.1007/BF00236557] [PMID]
Louis, E. D. (2009). Essential tremors: A family of neurodegenerative disorders? JAMA Neurology, 66(10), 1202-8. [DOI:10.1001/archneurol.2009.217]
Louis, E. D., Bromley, S. M., Jurewicz, E. C., & Watner, D. (2002). Olfactory dysfunction in essential tremor: A deficit unrelated to disease duration or severity. Neurology, 59(10), 1631-3. [DOI:10.1212/01.WNL.0000033798.85208.F2] [PMID]
Louis, E. D., Faust, P. L., Vonsattel, J. P., Honig, L. S., Rajput, A., & Robinson, C. A., et al. (2007). Neuropathological changes in essential tremor: 33 cases compared with 21 controls. Brain, 130(Pt 12), 3297-307. [DOI:10.1093/brain/awm266] [PMID]
Louis, E. D., Galecki, M., & Rao, A. K. (2013). Four essential tremor cases with moderately impaired gait: how impaired can gait be in this disease? Tremor and Other Hyperkinetic Movements, 3. [DOI:10.5334/tohm.138]
Mahmoudvand, H., Ziaali, N., Aghaei, I., Sheibani, V., Shojaee, S., & Keshavarz, H., et al. (2015). The possible association between Toxoplasma gondii infection and risk of anxiety and cognitive disorders in BALB/c mice. Pathog Glob Health, 109(8), 369-76. [DOI:10.1080/20477724.2015.1117742] [PMID] [PMCID]
Mejia, R. O. S., Ona, V. O., Li, M., & Friedlander, R. M. (2001). Minocycline reduces traumatic brain injury-mediated caspase-1 activation, tissue damage, and neurological dysfunction. Neurosurgery, 48(6), 1393-401. [DOI:10.1097/00006123-200106000-00051]
Miwa, H., & Kondo, T. (2011). T-type calcium channel as a new therapeutic target for tremor. Cerebellum (London, England), 10(3), 563-9. [DOI:10.1007/s12311-011-0277-y] [PMID]
Mohammadi, F., Esfahlani, M. A., & Shabani, M. (2019). Erythropoietin ameliorates harmaline-induced essential tremor and cognition disturbances. Neuroscience Letters, 704, 153-8. [DOI:10.1016/j.neulet.2019.04.017] [PMID]
Nasehi, M., Ketabchi, M., Khakpai, F., & Zarrindast, M. R. (2015). The effect of CA1 dopaminergic system in harmaline-induced amnesia. Neuroscience, 285, 47-59. [DOI:10.1016/j.neuroscience.2014.11.012] [PMID]
Nasehi, M., Piri, M., Nouri, M., Farzin, D., Nayer-Nouri, T., & Zarrindast, M. R. (2010). Involvement of dopamine D1/D2 receptors on harmane-induced amnesia in the step-down passive avoidance test. European Journal of Pharmacology, 634(1), 77-83. [DOI:10.1016/j.ejphar.2010.02.027] [PMID]
Nazeri, M., Shabani, M., Ghotbi Ravandi, S., Aghaei, I., Nozari, M., & Mazhari, S. (2015). Psychological or physical prenatal stress differentially affects cognition behaviors. Physiology & Behavior, 142, 155-60. [DOI:10.1016/j.physbeh.2015.02.016] [PMID]
O’Hearn, E., & Molliver, M. E. (1997). The olivocerebellar projection mediates ibogaine-induced degeneration of Purkinje cells: a model of indirect, trans-synaptic excitotoxicity. Journal of Neuroscience, 17(22), 8828-41. [DOI:10.1523/JNEUROSCI.17-22-08828.1997] [PMID] [PMCID]
Ondo, W. G., Sutton, L., Dat Vuong, K., Lai, D., & Jankovic, J. (2003). Hearing impairment in essential tremor. Neurology, 61(8), 1093-7. [DOI:10.1212/01.WNL.0000086376.40750.AF] [PMID]
Pi, R., Li, W., Lee, N. T., Chan, H. H., Pu, Y., & Chan, L. N., et al. (2004). Minocycline prevents glutamate‐induced apoptosis of cerebellar granule neurons by differential regulation of p38 and Akt pathways. Journal of Neurochemistry, 91(5), 1219-30. [DOI:10.1111/j.1471-4159.2004.02796.x] [PMID]
Plane, J. M., Shen, Y., Pleasure, D. E., & Deng, W. (2010). Prospects for minocycline neuroprotection. Archives of Neurology, 67(12), 1442-8. [DOI:10.1001/archneurol.2010.191] [PMID] [PMCID]
Popovic, N., Schubart, A., Goetz, B. D., Zhang, S. C., Linington, C., & Duncan, I. D. (2002). Inhibition of autoimmune encephalomyelitis by a tetracycline. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society, 51(2), 215-23. [DOI:10.1002/ana.10092] [PMID]
Radad, K., Moldzio, R., & Rausch, W. D. (2010). Minocycline protects dopaminergic neurons against long-term rotenone toxicity. Canadian Journal of Neurological Sciences, 37(1), 81-5. [DOI:10.1017/S0317167100009690] [PMID]
Rahimi Shourmasti, F., Goudarzi, I., Lashkarbolouki, T., Abrari, K., Elahdadi Salmani, M., & Goudarzi, A. (2012). Effects of riluzole on harmaline induced tremor and ataxia in rats: Biochemical, histological and behavioral studies. European Journal of Pharmacology, 695(1-3), 40-7. [DOI:10.1016/j.ejphar.2012.08.014] [PMID]
Razavinasab, M., Shamsizadeh, A., Shabani, M., Nazeri, M., Allahtavakoli, M., & Asadi‐Shekaari, M., et al. (2013). Pharmacological blockade of TRPV 1 receptors modulates the effects of 6‐OHDA on motor and cognitive functions in a rat model of P arkinson’s disease. Fundamental & Clinical Pharmacology, 27(6), 632-40. [DOI:10.1111/fcp.12015] [PMID]
Seabrook, T. J., Jiang, L., Maier, M., & Lemere, C. A. (2006). Minocycline affects microglia activation, Aβ deposition, and behavior in APP-tg mice. Glia, 53(7), 776-82. [DOI:10.1002/glia.20338] [PMID]
Shabani, M., Haghani, M., Sheibani, V., & Janahmadi, M. (2009). Changes in motor and learning behaviors of rats prenatally exposed to WIN 55212-2, a cannabinoid receptor agonist. Physiology and Pharmacology, 13(2), 120-9. https://www.sid.ir/en/journal/ViewPaper.aspx?id=167206
Tang, X. N., Wang, Q., Koike, M. A., Cheng, D., Goris, M. L., & Blankenberg, F. G., et al. (2007). Monitoring the protective effects of minocycline treatment with radiolabeled annexin V in an experimental model of focal cerebral ischemia. Journal of Nuclear Medicine, 48(11), 1822-8. [DOI:10.2967/jnumed.107.041335] [PMID]
Tikka, T., Fiebich, B. L., Goldsteins, G., Keinänen, R., & Koistinaho, J. (2001). Minocycline, a tetracycline derivative, is neuroprotective against excitotoxicity by inhibiting activation and proliferation of microglia. The Journal of Neuroscience, 21(8), 2580. [DOI:10.1523/JNEUROSCI.21-08-02580.2001] [PMID] [PMCID]
Vaziri, Z., Abbassian, H., Sheibani, V., Haghani, M., Nazeri, M., & Aghaei, I., et al. (2015). The therapeutic potential of Berberine chloride hydrate against harmaline-induced motor impairments in a rat model of tremor. Neuroscience letters, 590, 84-90. [DOI:10.1016/j.neulet.2015.01.078] [PMID]
Wang, X., Zhu, S., Drozda, M., Zhang, W., Stavrovskaya, I. G., & Cattaneo, E., et al. (2003). Minocycline inhibits caspase-independent and-dependent mitochondrial cell death pathways in models of Huntington’s disease. Proceedings of the National Academy of Sciences, 100(18), 10483-7. [DOI:10.1073/pnas.1832501100] [PMID] [PMCID]
Whaley, N. R., Putzke, J. D., Baba, Y., Wszolek, Z. K., & Uitti, R. J. (2007). Essential tremor: Phenotypic expression in a clinical cohort. Parkinsonism Relat Disord, 13(6), 333-9. [DOI:10.1016/j.parkreldis.2006.12.004] [PMID]
Yang, L., Sugama, S., Chirichigno, J. W., Gregorio, J., Lorenzl, S., & Shin, D. H., et al. (2003). Minocycline enhances MPTP toxicity to dopaminergic neurons. Journal of Neuroscience Research, 74(2), 278-85. [DOI:10.1002/jnr.10709] [PMID]
Yrjänheikki, J., Tikka, T., Keinänen, R., Goldsteins, G., Chan, P. H., & Koistinaho, J. (1999). A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window. Proceedings of the National Academy of Sciences of the United States of America, 96(23), 13496-500. [DOI:10.1073/pnas.96.23.13496] [PMID] [PMCID]
Zhang, L., Kitaichi, K., Fujimoto, Y., Nakayama, H., Shimizu, E., & Iyo, M., et al. (2006). Protective effects of minocycline on behavioral changes and neurotoxicity in mice after administration of methamphetamine. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 30(8), 1381-93. [DOI:10.1016/j.pnpbp.2006.05.015] [PMID]
Zhu, S., Stavrovskaya, I. G., Drozda, M., Kim, B. Y., & Ona, V., Li, M., et al. (2002). Minocycline inhibits cytochrome c release and delays progression of amyotrophic lateral sclerosis in mice. Nature, 417(6884), 74-8. [DOI:10.1038/417074a] [PMID]
Tremor is an unintentional, rhythmic, and sinusoidal movement that repeatedly occurs in one or more body parts (Abdo, van de Warrenburg, Burn, Quinn, & Bloem, 2010). Tremor is a common disabling symptom of many motor-related disorders such as Parkinson Disease (PD), Essential Temor (ET), Multiple Sclerosis (MS), Huntington Disease (HD), and some forms of ataxia, which originates from an involuntary muscle contraction/relaxation cycle (Arjmand et al., 2015; Hughes, Daniel, Blankson, & Lees, 1993). Classic ET is defined by postural kinetic (action) tremor, involving both upper limbs, in at least 95% of patients (Elble, 2000; Whaley, Putzke, Baba, Wszolek, & Uitti, 2007). Patients with ET have other motor problems, which imply cerebellar impairment, such as gait ataxia and eye movement abnormalities (Gitchel, Wetzel, & Baron, 2013; Louis et al., 2013), as well as non-motor conditions, including cognitive, psychiatric, and sensory abnormalities (Benito-Leon, Louis, & Bermejo-Pareja, 2006; Louis, Bromley, Jurewicz, & Watner,, 2002; Ondo, Sutton, Dat Vuong, Lai, & Jankovic, 2003). The exact pathogenesis of ET is uncertain, but neuroimaging studies indicate the role of olivo-cerebellar and cerebello-thalamo-cortical pathways in the creation of the tremors (Bhalsing, Saini, & Pal, 2013). Also, Gamma Amino-Butyric Acid (GABA) abnormalities (Gironell et al., 2012), neurodegeneration associated with gliosis, Purkinje cell loss, and increase in focal axonal swellings of Purkinje cells are associated with these tremors (Louis et al., 2007).
Harmaline can induce tremors similar to tremors in ET with the postural/kinetic exhibition. Studies show that intraperitoneal administration of this substance in animal models of ET increases glutamate transmission of the olivo-cerebellar pathway with an excitotoxic effect of Purkinje cells (Beitz & Saxon, 2004). Besides cytotoxicity, tremor, and motor disorders, harmaline can cause cognitive disturbances and learning and memory deficits (Abbassian et al., 2016; Aghaei et al., 2019; Nasehi, Ketabchi, Khakpai, & Zarrindast, 2015).
Regarding the pathophysiology of ET as a neurodegenerative disorder (Louis, 2009), neuroprotective agents that benefit from immunomodulatory, anti-inflammatory, anti-apoptotic, and anti-oxidant properties may be the most effective drug in the treatment of ET (Kim & Suh, 2009). It has been proposed that minocycline has neuroprotective effects in several animal models of neurological diseases (Garrido-Mesa, Zarzuelo, & Galvez, 2013; Handforth, 2012; O’Hearn & Molliver, 1997). Experimental and clinical studies have been shown its beneficial effects in the treatment of ischemia (Yrjänheikki et al., 1999), traumatic brain injury (Mejia, Ona, Li, & Friedlander, 2001), Amyotrophic Lateral Sclerosis (ALS) (Zhu et al., 2002), PD (Jackson-Lewis et al., 2002), HD (Chen et al., 2000; Wang et al., 2003), MS (Popovic et al., 2002), and Alzheimer's Disease (AD) (Choi et al., 2007). Although the exact mechanism of minocycline neuroprotective action is unclear, several studies suggest an association between this action and the inhibition of cytochrome C involved in cell death, the inhibition of caspase expression, p38 Mitogen-Activated Protein Kinase (p38 MAPK) inhibition, increased expression of 5-lipooxygenase, and the inhibition of Poly (ADP-Ribose) Polymerase1 (PARP1) (Chu et al., 2010; Kim & Suh, 2009; Li & McCullough, 2009; Tang et al., 2007). Minocycline also leads to mitigation of glutamate-induced excitotoxicity (Pi et al., 2004) and mitigation of NMDA-induced (N -methyl-D-aspartate receptor) neurotoxicity by the reduction in NMDA-induced Ca2+ inward current and mitochondria Ca2+ uptake (Garcia-Martinez et al., 2010).
Because of the minocycline ameliorating effect on neurodegenerative diseases, this study aimed to assess the possible neuroprotective effects of minocycline on motor and cognitive deficits induced by harmaline in the experimental rat model of ET.
2. Methods
2.1. Study animals
Male Wistar rats (weighing 100-120 g) were experimented according to the Research and Ethics Committee of Kerman University and internationally accepted principles for laboratory animal use and care. They were kept at a standard laboratory condition (12-12 h light-dark cycle) with ad libitum access to food pellets and water. All tests were performed during the light period.
2.2. Drug preparation and administration
Harmaline HCl and minocycline hydrochloride were purchased from Sigma-Aldrich. Both substances were dissolved in 0.9% sterile, isotonic saline on the day of administration. Before the experiments began, the rats were put into the test room for one hour to get used to the environment. The rats were randomly divided into four groups (10 rats in each group). The first group was received saline. The second group was given minocycline (90 mg/kg, IP). The third group was received a single injection of harmaline (30 mg/kg, IP). Animals in group 4 were given minocycline (90 mg/kg IP) 30 min before harmaline administration (Elewa, Hilali, Hess, Machado, & Fagan, 2006; Plane, Shen, Pleasure, & Deng, 2010). Each group went through five different behavioral studies, which were performed 30 min after harmaline injection with sequentially 15 min rest intervals among each assay in the following order: tremor score assessment, open field test, footprint, rotarod, wire grip, and passive avoidance task (Behavioral procedures timeline; Figure 1).
.jpg)
2.3. Assessment of motor activity
2.3.1. Observation
The scale of tremors was assessed by an observer who was unaware of the treatment protocol. The qualitative grading of tremor severity was based on the Arshaduddin et al. method, which is as follows: no tremor= 0, mild tremor= 1 (only the head and neck affected), moderate intermittent tremor= 2 (whole-body parts affected), moderate persistent tremor= 3 (whole-body parts and tail affected), and pronounced severe tremor= 4 (trembling and unable to mobility) (Abbassian et al., 2016; Dahmardeh, Shabani, Basiri, Kalantaripour, & Asadi-Shekaari, 2019).
2.3.2. Open field test
The locomotion and anxiety-like behaviors of the rats from each group were recorded and analyzed by an automated video tracking software (Ethovision, Noldus Technology, Netherlands) for five minutes. Each animal was individually placed in the middle of the arena of apparatus (90×90×45 [H] cm, divided into 16 small squares). The behavioral parameters consist of total time spent in the center or periphery, Total Distance Moved (TDM), speed, and the number of grooming and rearing were recorded for each rat (Mahmoudvand et al., 2015; Razavinasab et al., 2013).
2.3.3. Footprint
To evaluate the rat’s walking pattern and gait kinematic, we used a footprint-test. The hind limbs of rats were covered with a non-toxic color. The rats were then placed in a Plexiglas narrow corridor (100 cm long×10 cm height×10 cm width) ending in a dark box covered with white absorbent paper to move freely. Three consecutive strides were chosen to assess the footprint so that the distance between the steps of each side (the middle of each foot to the middle of the next foot) was recorded as the length of the step and the distance between the center of one foot with the opposite foot was recorded as the step width, and the mean value of these data was analyzed. The initiating and finishing steps were also excluded from the study. Before testing, the rats were put on the runway to get acquainted with the procedure (Mohammadi, Esfahlani, & Shabani, 2019).
2.3.4. Accelerating rotarod assay: A Motor Performance Test
The rotarod test was used to check the balance and coordination. To do this test, we placed each rat on a rotating rod for 5 minutes for habituation before the test began, and the velocity of the rotation accelerated from 10 revolutions per min (rpm) to 60 rpm during 5 minutes. After habituation and rest, three trials were performed, and the average interval between placing the rat on the rod and falling from the rod was measured and recorded. The total test time for each rat was 300 s, and 5 min rest was between each trial (Nazeri et al., 2015).
2.3.5. Wire Grip Test
Wire grip test was used to measure muscle strength and balance by placing the rat on both forepaws on a steel wire (80 cm long, 7 mm diameter). When the rat grasped the wire, the rat was released. The duration of the time that the rat does not fall and holds the wire was recorded. Three trials were performed with five minutes between trials, and the mean values were recorded as the final result (Haghani, Shabani, & Moazzami, 2013).
2.3.6. Passive Avoidance Test
The passive avoidance test evaluates learning and memory function in rats. The apparatus size was 40 cm long, 20 cm width, and 20 cm height. It comprised two equal size sections (light and dark) with a grid floor, divided by a sliding door. The electrical stimulator was embedded in the dark floor partition (50 Hz, 0.5 mA, 2 ms). This test consists of three trials (adaptation trial, training trial, retention trial). In the learning phase of the test, each animal was first habituated to the test equipment. In the adaptation trial, the rat was placed in the light cavity facing the door, and after 10 seconds, the door was opened, and the time the animal entered the dark chamber was recorded. If, after 120 seconds, the rat was reluctant to cross the dark section, it was deleted from the test.
Two hours later, the training trial was done. The animal was returned to the light compartment, the door opened, and the animal was allowed to move to the dark chamber. Upon arrival, the sliding door was closed, and a foot-shock was given. After 20 seconds, the animal was moved to its home cage. After 5 minutes, this step was repeated, and if the rat entered the dark part, the shock was given again. This step continued until the rat learned the training. If it stayed on for about 300 seconds in the light part, the training was ended. The number of shocks was recorded as a variable. After 24 hours, the memory retention of rats was measured. The rat was placed in the lightbox, and after 10 seconds, the door was opened. The time it took for the animal to enter the dark part for the first time was recorded as Step Through Latency (STL). Cut off point of this trial was 300 seconds (Shabani, Haghani, Sheibani, & Janahmadi, 2009).
2.4. Morphological analysis
Seven days after treatment, five rats from each group were randomly selected for light microscopic study. The animals were guillotined under deep anesthesia (ketamine-xylazine). Then, they were transcardially perfused with normal saline and fixed with 4% paraformaldehyde solution. Next, their cerebellums and brain stems were carefully extracted from the skull and post-fixed in 10% buffered formaldehyde for 48 hours. In short, the tissues were dehydrated, cleared, embedded in paraffin, and finally, 5-μm sections (coronal sections at the vermal level of cerebellum and rostro-caudal of the brainstem) were obtained for histological study. The sections were deparaffinized and were hydrated, followed by staining with Hematoxylin and Eosin (H&E) (Rahimi Shourmasti et al., 2012). For the ultrastructural evaluation, two rats were randomly selected from each group and intracardially perfused with 4% glutaraldehyde in 0.1 M Phosphate Buffer Solution (PBS). Then, their brains were extracted carefully, and target parts were cut into 1×1-mm fragments. The tissues were fixed in 4% glutaraldehyde PBS for 24 hours, followed by post-fixation in 1% osmium tetroxide in PBS for 1 hour. Tissue processing was performed according to the standard method. Briefly, this process consists of washing, dehydrating with ethanol, infiltrating with propylene oxide twice for 30 minutes, infiltrating and embedding with resin (Epon-812). Ultra-cut (50-80 nm) was obtained using Leica ultramicrotome. The sections were placed on copper grids and stained with uranyl acetate and lead citrate. They were analyzed using a transmission electron microscope (Zeiss EM10, Germany) (Asadi-Shekaari et al., 2011).
2.5. Data analysis
We used a 1-way ANOVA test followed by Tukey’s (as a post hoc test) for analyzing the data. The standard error of the mean (Mean±SEM) expresses the results. The significance of the difference between the mean of groups was considered with P<0.05.
3. Results
3.1. Behavioral studies
3.1.1. Effect of minocycline on the scale of tremor
The severity of tremors increased in harmaline groups (F3, 36= 29.7, P<0.001, Figure 2) compared to the saline and minocycline groups.
.jpg)
Minocycline therapy resulted in a significant reduction in the harmaline-induced tremor score (F3, 36=6.3, P<0.05, Figure 2A) compared to the harmaline alone group.
3.1.2. The effect of minocycline on gait disturbance
Footprint assay showed that harmaline markedly increased hind paw stride width, compared with the saline (F3, 36=13.6, P<0.001) and minocycline (P<0.01) groups (Figure 2B). There was a significant step width decrease in the harmaline + minocycline group compared with the harmaline group (F3, 36=9.6, P<0.05, Figure 2B). However, there was no significant difference among harmaline + minocycline and saline groups. The effect of minocycline treatment was not detected in the left (Figure 2C) and right (Figure 2D) step lengths.
3.1.3. Effect of minocycline on locomotors and anxiety-like behaviors
The total distance moved in the harmaline group was significantly lower compared with saline (F3, 36=9.1, P<0.01) and minocycline (P<0.05) groups. Besides, this parameter in the harmaline + minocycline group was also significantly lower compared with the saline group (F3, 36=4.9, P<0.05, Figure 3A).
.jpg)
The velocity of animals in all groups significantly decreased compared with the saline group (F3, 36=16.4, P<0.001). Also, harmaline and harmaline + minocycline groups significantly decreased compared with the minocycline group in the velocity of movement (P<0.05, Figure 3B). Mobility duration in the harmaline (P<0.05) and harmaline + minocycline (F3, 36= 8.6, P<0.01) groups compared with the saline group, significantly decreased. But no significant difference was observed between other groups (Figure 3C). Time spent in the periphery of the box was not significantly different between all groups (Figure 3D).
3.1.4. The effects of minocycline pretreatment on balance function and muscle strength
Our results showed that time of staying on the rod in the rotarod test in the harmaline group compared with the saline group (F3, 36=21.3, P<0.001) and minocycline group (F3, 36=23.8, P<0.001) had a significant decrease. A significant difference was detected between the harmaline + minocycline group compared to the saline group (F3, 36=11.6, P<0.01) as well. No significant difference was observed between other groups (Figure 4A).
.jpg)
In wire grip apparatus, rats of the harmaline group showed a decrease in falling time compared with the saline group (F3, 36=19.3, P<0.001), and minocycline group (F3, 36=7.7, P<0.01). Rats of the minocycline + harmaline group spent a shorter duration on the wire grip compared with the saline group (F3, 36=9.6, P<0.01) and minocycline group (P<0.05, Figure 4B).
3.1.5. Effect of harmaline and minocycline on passive avoidance memory
The number of receiving shocks in the shuttle box test in the harmaline and harmaline + minocycline groups was significantly increased compared with the saline group (F3, 36=4.9, P<0.05). A significant increase was noted compared with the minocycline group (F3, 36=3.5, P<0.05, Figure 5A).
.jpg)
The time of entrance to the dark compartment in the Shuttle Box Test (STL) significantly decreased in the harmaline and harmaline + minocycline compared with the saline group (F3, 36=16.6, P<0.001). Also, a significant decrease was observed in these groups compared with the minocycline group (F3, 36=11.2, P<0.001). In harmaline + minocycline group a significant increase was seen compared with harmaline group (F3, 36=13.8, P<0.001, Figure 5B).
3.2. Histological assessment
3.2.1. Light microscopy findings
The normal morphology of Purkinje cells is shown in Figure 6A.

The number of degenerated Purkinje cells significantly increased in the harmaline group compared with the saline (F3, 12=5.7, P<0.01) and the minocycline groups (F3, 12=3.1, P<0.05, Figure 6B). Administration of minocycline 30 minutes before harmaline injection resulted in a significant decrease in the number of degenerated cerebellar Purkinje cells compared with the harmaline group (P<0.05, Figure 6B).
The normal morphology of inferior olive nucleus neurons is shown in Figure 6C. There was a significant increase in the number of degenerated neurons of the Inferior Olive Nucleus (ION) in the harmaline group compared with the saline (F3, 12=10.3, P<0.001) and the minocycline (P<0.001) groups. With the administration of minocycline, a significant decrease was observed in the number of damaged cells in the ION compared with the harmaline group (F3, 12=9.1, P<0.001, Figure 6D).
3.2.2. Electron microscopy findings
In the saline group, cerebellar Purkinje cells and ION neurons had normal ultrastructure, i.e. round nucleus, dispersed chromatin, and intact nucleolar membrane (Figure 7).

Harmaline administration resulted in ultrastructure changes in the aforementioned cells, including shrinkage and deformity of the nucleus, chromatolysis, nuclear lysis, and increased cytoplasmic density. Pretreatment by minocycline decreased these effects (Figure 7).
4. Discussion
The current study results demonstrated that pretreatment with minocycline improved tremor status and memory deficit in the rat’s model of tremor induced by harmaline, as evidenced by several behavioral tasks. However, there were no significant improvements in balance function and muscle strength.
Tremor is a sign of many neurodegenerative diseases, and several studies have shown the neuroprotective effects of minocycline in several neurodegenerative diseases like PD (Du et al., 2001), HD (Radad, Moldzio, & Rausch, 2010), AD (Fan et al., 2007; Seabrook, Jiang, Maier, & Lemere, 2006), ALS (Zhu et al., 2002), and acute injuries such as spinal cord injury (Kim & Suh, 2009). So, we decided to conduct this study to assess the neuroprotective effect of minocycline on tremors induced by harmaline. Harmaline was chosen to induce the tremor model because its underlying mechanism is excessive stimulation of climbing fiber to the cerebellum and glutamate excitotoxicity in Purkinje cells and ION (Beitz & Saxon, 2004). Many studies have confirmed that harmaline provokes generalized transient action tremors with kinetic and postural components displayed in minutes or hours (Grimaldi & Manto, 2013; Vaziri et al., 2015).
The results of our study showed that harmaline produced moderate tremors, which is consistent with previous behavioral studies (Abbassian et al., 2016; Nasehi et al., 2010). Previous research showed that harmaline performs its tremorogenesis action by raising in glutamate discharge (Llinás & Volkind, 1973), probably acting on NMDA receptors and T-type calcium channels (Handforth, 2012), and excitotoxicity damages of Purkinje cells via hyper-releasing of glutamate from climbing fibers (Choi, 1988). It has been suggested that harmaline increases rebound T-type calcium pulses in inferior olive cells (Miwa & Kondo, 2011). On the other hand, minocycline can reduce neuron excitability by blocking voltage-dependent Na+-channels and attenuates glutamatergic neurotransmission and Ca2+ signaling in the central nervous system as well as the reduction in the cellular glutamate amount related to voltage-dependent Ca2+ channels (Gonzalez et al., 2007). Hence, neuroprotection effects of minocycline on tremor results from its neuronal excitability reduction and glutamate transmission, alleviating and preventing calcium overloading in neurons.
In our study, the results of the wire grip test and rotarod did not show any improvement. This finding is maybe associated with minocycline potential for vestibular toxicity (Li et al., 2013). Minocycline has common ototoxicity side effects with vestibular symptoms such as dizziness, vertigo, ataxia, tinnitus, and other symptoms (Diguet et al., 2004). Also, the result of the open field test showed pretreatment by minocycline could not mitigate anxiety in rats, and previous studies have shown that it has had deleterious effects on motor performance, balance, coordination, and anxiety behaviors on a model of Parkinson study as well (Tikka, Fiebich, Goldsteins, Keinänen, & Koistinaho, 2001; Yang et al., 2003).
The shuttle box test showed that harmaline treatment impaired the learning phase as indicated by the increased number of shocks required before animals met the criterion, and this impairment was not affected by minocycline. When memory impairment was assessed in the shuttle box, harmaline treatment impaired acquisition of passive avoidance as demonstrated by a shorter step-through latency. However, unlike learned inhibition, minocycline partially reversed this harmaline-induced impairment.
Whereas the memory formation and retrieval are mainly associated with the hippocampus and activity of its dopaminergic receptors in the CA1 area (Hartman, Lee, Zipfel, & Wozniak, 2005; Khakpai, Nasehi, Haeri-Rohani, Eidi, & Zarrindast, 2013), so any destruction in this pathway can cause amnesia. Several mechanisms and pathways are suggested for amnesia and cognitive deficiency produced by harmaline. Some studies demonstrated that the interaction between harmaline and dopamine receptors in CA1 are involved in memory impairment (Kim, Hassler, Kurokawa, & Bak, 1970; Nasehi et al., 2015). On the other hand, some studies have reported that minocycline can reduce neurotoxicity in dopaminergic systems in hippocampus and striatum by inhibiting microglia function (Zhang et al., 2006). Therefore, it is likely that the minocycline protection effect on dopaminergic neurons is responsible for improving the memory deficit induced by harmaline administration.
In addition to behavioral assessments, the second objective of the current study was to determine the effect of minocycline administration on the possible morphological and ultrastructure alterations induced by harmaline in a rat model of ET.
Harmaline causes excitotoxicity by increasing the activation and proliferation of microglial and enhancing the release of their toxic substances, such as p38 microtubule-associated protein kinase, interleukine-1b, and nitric oxide, which leads to neuronal cell death (Tikka et al., 2001). Therefore harmaline increases cell loss in inferior olive and Purkinje cells because of glutamate increment, as was illustrated by previous studies (Handforth, 2012). Our histology results showed a positive effect of minocycline on these neurons.
Minocycline exerts its neuroprotective effects through several ways, such as alleviating the underlying neurodegenerative processes, including anti-inflammatory, anti-apoptotic, and anti-oxidant activity, and also applying against excitotoxicity, mainly by inhibition of microglial activity (Li, Yuan, & Schluesener, 2013; Tikka et al., 2001). We assume that preservation of neurons and decrement of neurodegeneration in inferior olive and Purkinje cells in our study was resulted from attenuating the microglial action of minocycline. However, we cannot elucidate if other possible mechanisms are contributed to this process as well or not.
The obtained findings demonstrated that harmaline causes tremor symptoms and impaired cognitive functions in Wistar male rats. Also, some of these impairments were reversible by minocycline. However, minocycline seems to act as a neuroprotective agent to improve tremor severity, gait width disturbance, and memory retrieval impairments induced by harmaline. Of course, more investigations are needed to evaluate the mechanism of its therapeutic effects on motor and cognitive impairments.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee (EC/KNRC/96-59) of the University of Kerman Medical Sciences, Kerman.
Funding
The paper was extracted from the MSc. thesis of the first author at the Kerman University of Medical Sciences Research Deputy and Neuroscience Research Center.
Authors' contributions
Conceptualization, methodology, and writing – original draft: Marzieh Maneshian, Farinaz Nasirinezhad, Fatemeh Mohammadi; Data collection: Mina Behzadi and Majid Asadi –shekaari; Data analysis: Mohammad Shabani. Final approval: All authors.
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We appreciate the staff of Kerman University of Medical Sciences for funding the study.
References
Abbassian, H., Esmaeili, P., Tahamtan, M., Aghaei, I., Vaziri, Z., & Sheibani, V., et al. (2016). Cannabinoid receptor agonism suppresses tremor, cognition disturbances, and anxiety-like behaviors in a rat model of essential tremor. Physiology & Behavior, 164, 314-20. [DOI:10.1016/j.physbeh.2016.06.013] [PMID]
Abbassian, H., Whalley, B. J., Sheibani, V., & Shabani, M. (2016). Cannabinoid type 1 receptor antagonism ameliorates harmaline-induced essential tremor in rat. British Journal of Pharmacology, 173(22), 3196-207. [DOI:10.1111/bph.13581] [PMID] [PMCID]
Abdo, W. F., van de Warrenburg, B. P. C., Burn, D. J., Quinn, N. P., & Bloem, B. R. (2010). The clinical approach to movement disorders. Nature Reviews Neurology, 6, 29. [DOI:10.1038/nrneurol.2009.196] [PMID]
Aghaei, I., Hajali, V., Haghani, M., Vaziri, Z., Moosazadeh, M., & Shabani, M. (2019). Peroxisome proliferator-activated receptor-gamma activation attenuates harmaline-induced cognitive impairments in rats. Journal of Clinical Neuroscience, 59, 276-83. [DOI:10.1016/j.jocn.2018.11.004] [PMID]
Arjmand, S., Vaziri, Z., Behzadi, M., Abbassian, H., Stephens, G. J., & Shabani, M. (2015). Cannabinoids and Tremor Induced by Motor-related Disorders: Friend or Foe? Neurotherapeutics, 12(4), 778-87. [DOI:10.1007/s13311-015-0367-5] [PMID] [PMCID]
Asadi-Shekaari, M., Eftekhar Vaghefi, H., Ezzat Abadi Pour, M., Sheibani, V., Shams Ara, A., & Behbahani, P. (2011). Antiapoptotic effects of aspirin on CA1 pyramidal neurons in adult rats. Iranian Journal of Pathology, 6(4), 187-92. http://ijp.iranpath.org/article_8516_1349.html
Beitz, A. J., & Saxon, D. (2004). Harmaline-induced climbing fiber activation causes amino acid and peptide release in the rodent cerebellar cortex and a unique temporal pattern of Fos expression in the olivo-cerebellar pathway. Journal of Neurocytology, 33(1), 49-74. [DOI:10.1023/B:NEUR.0000029648.81071.20] [PMID]
Benito-Leon, J., Louis, E. D., & Bermejo-Pareja, F. (2006). Population-based case-control study of cognitive function in essential tremor. Neurology, 66(1), 69-74. [DOI:10.1212/01.wnl.0000192393.05850.ec] [PMID]
Bhalsing, K. S., Saini, J., & Pal, P. K. (2013). Understanding the pathophysiology of essential tremor through advanced neuroimaging: A review. Journal of the Neurological Sciences, 335(1-2), 9-13. [DOI:10.1016/j.jns.2013.09.003] [PMID]
Chen, M., Ona, V. O., Li, M., Ferrante, R. J., Fink, K. B., & Zhu, S., et al. (2000). Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nature Medicine, 6(7), 797. [DOI:10.1038/77528] [PMID]
Choi, D. W. (1988). Glutamate neurotoxicity and diseases of the nervous system. Neuron, 1(8), 623-34. [DOI:10.1016/0896-6273(88)90162-6]
Choi, Y., Kim, H. S., Shin, K. Y., Kim, E. M., Kim, M., & Kim, H. S., et al. (2007). Minocycline attenuates neuronal cell death and improves cognitive impairment in Alzheimer’s disease models. Neuropsychopharmacology, 32(11), 2393-404. [DOI:10.1038/sj.npp.1301377] [PMID]
Chu, L.-S., Fang, S. H., Zhou, Y., Yin, Y. J., Chen, W.Y., & Li, J. H., et al. (2010). Minocycline inhibits 5-lipoxygenase expression and accelerates functional recovery in chronic phase of focal cerebral ischemia in rats. Life Sciences, 86(5-6), 170-7. [DOI:10.1016/j.lfs.2009.12.001] [PMID]
Dahmardeh, N., Shabani, M., Basiri, M., Kalantaripour, T. P., & Asadi-Shekaari, M. (2019). Functional antagonism of sphingosine-1-phosphate receptor 1 prevents harmaline-induced ultrastructural alterations and caspase-3 mediated apoptosis. Malaysian Journal of Medical Sciences, 26(4), 28-38. [DOI:10.21315/mjms2019.26.4.4] [PMID] [PMCID]
Diguet, E., Fernagut, P.-O., Wei, X., Du, Y., Rouland, R., & Gross, C., et al. (2004). Deleterious effects of minocycline in animal models of Parkinson’s disease and Huntington’s disease. European Journal of Neuroscience, 19(12), 3266-76. [DOI:10.1111/j.0953-816X.2004.03372.x] [PMID]
Du, Y., Ma, Z., Lin, S., Dodel, R. C., Gao, F., & Bales, K. R., et al. (2001). Minocycline prevents nigrostriatal dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Proceedings of the National Academy of Sciences of the United States of America, 98(25), 14669-74. [DOI:10.1073/pnas.251341998] [PMID] [PMCID]
Elble, R. J. (2000). Diagnostic criteria for essential tremor and differential diagnosis. Neurology, 54(suppl 4), 2-6. [PMID]
Elewa, H. F., Hilali, H., Hess, D. C., Machado, L. S., & Fagan, S. C. (2006). Minocycline for short‐term neuroprotection. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 26(4), 515-21. [DOI:10.1592/phco.26.4.515] [PMID] [PMCID]
Fan, R., Xu, F., Previti, M. L., Davis, J., Grande, A. M., & Robinson, J. K., et al. (2007). Minocycline reduces microglial activation and improves behavioral deficits in a transgenic model of cerebral microvascular amyloid. The Journal of Neuroscience, 27(12), 3057-63. [DOI:10.1523/JNEUROSCI.4371-06.2007] [PMID] [PMCID]
Garcia-Martinez, E. M., Sanz-Blasco, S., Karachitos, A., Bandez, M. J., Fernandez-Gomez, F. J., & Perez-Alvarez, S., et al. (2010). Mitochondria and calcium flux as targets of neuroprotection caused by minocycline in cerebellar granule cells. Biochemical Pharmacology, 79(2), 239-50. [DOI:10.1016/j.bcp.2009.07.028] [PMID]
Garrido-Mesa, N., Zarzuelo, A., & Galvez, J. (2013). Minocycline: Far beyond an antibiotic. British Journal of Pharmacology, 169(2), 337-52. [DOI:10.1111/bph.12139] [PMID] [PMCID]
Gironell, A., Figueiras, F. P., Pagonabarraga, J., Herance, J. R., Pascual-Sedano, B., & Trampal, C., et al. (2012). Gaba and serotonin molecular neuroimaging in essential tremor: A clinical correlation study. Parkinsonism Relat Disord, 18(7), 876-80. [DOI:10.1016/j.parkreldis.2012.04.024] [PMID]
Gitchel, G. T., Wetzel, P. A., & Baron, M. S. (2013). Slowed saccades and increased square wave jerks in essential tremor. Tremor and Other Hyperkinetic Movements, 3, tre-03-178-4116-2. [DOI:10.5334/tohm.127] [PMID] [PMID]
Gonzalez, J. C., Egea, J., Del Carmen Godino, M., Fernandez-Gomez, F. J., Sanchez-Prieto, J., & Gandia, L., et al. (2007). Neuroprotectant minocycline depresses glutamatergic neurotransmission and Ca(2+) signalling in hippocampal neurons. European Journal of Neuroscience, 26(9), 2481-95. [DOI:10.1111/j.1460-9568.2007.05873.x] [PMID]
Grimaldi, G., & Manto, M. (2013). Assessment of tremor: Clinical and functional scales. In Grimaldi G., Manto M. (eds), Mechanisms and Emerging Therapies in Tremor Disorders. (pp. 325-340). New York, NY.: Springer. [DOI:10.1007/978-1-4614-4027-7_18]
Haghani, M., Shabani, M., & Moazzami, K. (2013). Maternal mobile phone exposure adversely affects the electrophysiological properties of Purkinje neurons in rat offspring. Neuroscience, 250, 588-98. [DOI:10.1016/j.neuroscience.2013.07.049] [PMID]
Handforth, A. (2012). Harmaline tremor: Underlying mechanisms in a potential animal model of essential tremor. Tremor and Other Hyperkinetic Movements, 2. [DOI:10.5334/tohm.108]
Hartman, R. E., Lee, J. M., Zipfel, G. J., & Wozniak, D. F. (2005). Characterizing learning deficits and hippocampal neuron loss following transient global cerebral ischemia in rats. Brain Research, 1043(1-2), 48-56. [DOI:10.1016/j.brainres.2005.02.030] [PMID]
Hughes, A. J., Daniel, S. E., Blankson, S., & Lees, A. J. (1993). A Clinicopathologic Study of 100 Cases of Parkinson’s Disease. Archives of Neurology, 50(2), 140-8. [DOI:10.1001/archneur.1993.00540020018011] [PMID]
Jackson-Lewis, V., Vila, M., Tieu, K., Teismann, P., Vadseth, C., & Choi, D. K., et al. (2002). Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine mouse model of Parkinson disease. Journal of Neuroscience, 22(5), 1763-71. [DOI:10.1523/JNEUROSCI.22-05-01763.2002] [PMID] [PMCID]
Khakpai, F., Nasehi, M., Haeri-Rohani, A., Eidi, A., & Zarrindast, M. R. (2013). Septo-hippocampo-septal loop and memory formation. Basic and Clinical Neuroscience, 4(1), 5-23. [PMCID] [PMID]
Kim, H. S., & Suh, Y. H. (2009). Minocycline and neurodegenerative diseases. Behavioural Brain Research, 196(2), 168-79. [DOI:10.1016/j.bbr.2008.09.040] [PMID]
Kim, J. S., Hassler, R., Kurokawa, M., & Bak, I. J. (1970). Abnormal movements and rigidity induced by harmaline in relation to striatal acetylcholine, serotonin, and dopamine. Experimental Neurology, 29(2), 189-200. [DOI:10.1016/0014-4886(70)90049-X]
Li, C., Yuan, K., & Schluesener, H. (2013). Impact of minocycline on neurodegenerative diseases in rodents: A meta-analysis. Reviews in the Neurosciences, 24(5), 553-62. [DOI:10.1515/revneuro-2013-0040] [PMID]
Li, J., & McCullough, L. D. (2009). Sex differences in minocycline-induced neuroprotection after experimental stroke. Journal of Cerebral Blood Flow & Metabolism, 29(4), 670-4. [DOI:10.1038/jcbfm.2009.3] [PMID] [PMCID]
Llinás, R., & Volkind, R. A. (1973). The olivo-cerebellar system: Functional properties as revealed by harmaline-induced tremor. Experimental Brain Research, 18(1), 69-87. [DOI:10.1007/BF00236557] [PMID]
Louis, E. D. (2009). Essential tremors: A family of neurodegenerative disorders? JAMA Neurology, 66(10), 1202-8. [DOI:10.1001/archneurol.2009.217]
Louis, E. D., Bromley, S. M., Jurewicz, E. C., & Watner, D. (2002). Olfactory dysfunction in essential tremor: A deficit unrelated to disease duration or severity. Neurology, 59(10), 1631-3. [DOI:10.1212/01.WNL.0000033798.85208.F2] [PMID]
Louis, E. D., Faust, P. L., Vonsattel, J. P., Honig, L. S., Rajput, A., & Robinson, C. A., et al. (2007). Neuropathological changes in essential tremor: 33 cases compared with 21 controls. Brain, 130(Pt 12), 3297-307. [DOI:10.1093/brain/awm266] [PMID]
Louis, E. D., Galecki, M., & Rao, A. K. (2013). Four essential tremor cases with moderately impaired gait: how impaired can gait be in this disease? Tremor and Other Hyperkinetic Movements, 3. [DOI:10.5334/tohm.138]
Mahmoudvand, H., Ziaali, N., Aghaei, I., Sheibani, V., Shojaee, S., & Keshavarz, H., et al. (2015). The possible association between Toxoplasma gondii infection and risk of anxiety and cognitive disorders in BALB/c mice. Pathog Glob Health, 109(8), 369-76. [DOI:10.1080/20477724.2015.1117742] [PMID] [PMCID]
Mejia, R. O. S., Ona, V. O., Li, M., & Friedlander, R. M. (2001). Minocycline reduces traumatic brain injury-mediated caspase-1 activation, tissue damage, and neurological dysfunction. Neurosurgery, 48(6), 1393-401. [DOI:10.1097/00006123-200106000-00051]
Miwa, H., & Kondo, T. (2011). T-type calcium channel as a new therapeutic target for tremor. Cerebellum (London, England), 10(3), 563-9. [DOI:10.1007/s12311-011-0277-y] [PMID]
Mohammadi, F., Esfahlani, M. A., & Shabani, M. (2019). Erythropoietin ameliorates harmaline-induced essential tremor and cognition disturbances. Neuroscience Letters, 704, 153-8. [DOI:10.1016/j.neulet.2019.04.017] [PMID]
Nasehi, M., Ketabchi, M., Khakpai, F., & Zarrindast, M. R. (2015). The effect of CA1 dopaminergic system in harmaline-induced amnesia. Neuroscience, 285, 47-59. [DOI:10.1016/j.neuroscience.2014.11.012] [PMID]
Nasehi, M., Piri, M., Nouri, M., Farzin, D., Nayer-Nouri, T., & Zarrindast, M. R. (2010). Involvement of dopamine D1/D2 receptors on harmane-induced amnesia in the step-down passive avoidance test. European Journal of Pharmacology, 634(1), 77-83. [DOI:10.1016/j.ejphar.2010.02.027] [PMID]
Nazeri, M., Shabani, M., Ghotbi Ravandi, S., Aghaei, I., Nozari, M., & Mazhari, S. (2015). Psychological or physical prenatal stress differentially affects cognition behaviors. Physiology & Behavior, 142, 155-60. [DOI:10.1016/j.physbeh.2015.02.016] [PMID]
O’Hearn, E., & Molliver, M. E. (1997). The olivocerebellar projection mediates ibogaine-induced degeneration of Purkinje cells: a model of indirect, trans-synaptic excitotoxicity. Journal of Neuroscience, 17(22), 8828-41. [DOI:10.1523/JNEUROSCI.17-22-08828.1997] [PMID] [PMCID]
Ondo, W. G., Sutton, L., Dat Vuong, K., Lai, D., & Jankovic, J. (2003). Hearing impairment in essential tremor. Neurology, 61(8), 1093-7. [DOI:10.1212/01.WNL.0000086376.40750.AF] [PMID]
Pi, R., Li, W., Lee, N. T., Chan, H. H., Pu, Y., & Chan, L. N., et al. (2004). Minocycline prevents glutamate‐induced apoptosis of cerebellar granule neurons by differential regulation of p38 and Akt pathways. Journal of Neurochemistry, 91(5), 1219-30. [DOI:10.1111/j.1471-4159.2004.02796.x] [PMID]
Plane, J. M., Shen, Y., Pleasure, D. E., & Deng, W. (2010). Prospects for minocycline neuroprotection. Archives of Neurology, 67(12), 1442-8. [DOI:10.1001/archneurol.2010.191] [PMID] [PMCID]
Popovic, N., Schubart, A., Goetz, B. D., Zhang, S. C., Linington, C., & Duncan, I. D. (2002). Inhibition of autoimmune encephalomyelitis by a tetracycline. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society, 51(2), 215-23. [DOI:10.1002/ana.10092] [PMID]
Radad, K., Moldzio, R., & Rausch, W. D. (2010). Minocycline protects dopaminergic neurons against long-term rotenone toxicity. Canadian Journal of Neurological Sciences, 37(1), 81-5. [DOI:10.1017/S0317167100009690] [PMID]
Rahimi Shourmasti, F., Goudarzi, I., Lashkarbolouki, T., Abrari, K., Elahdadi Salmani, M., & Goudarzi, A. (2012). Effects of riluzole on harmaline induced tremor and ataxia in rats: Biochemical, histological and behavioral studies. European Journal of Pharmacology, 695(1-3), 40-7. [DOI:10.1016/j.ejphar.2012.08.014] [PMID]
Razavinasab, M., Shamsizadeh, A., Shabani, M., Nazeri, M., Allahtavakoli, M., & Asadi‐Shekaari, M., et al. (2013). Pharmacological blockade of TRPV 1 receptors modulates the effects of 6‐OHDA on motor and cognitive functions in a rat model of P arkinson’s disease. Fundamental & Clinical Pharmacology, 27(6), 632-40. [DOI:10.1111/fcp.12015] [PMID]
Seabrook, T. J., Jiang, L., Maier, M., & Lemere, C. A. (2006). Minocycline affects microglia activation, Aβ deposition, and behavior in APP-tg mice. Glia, 53(7), 776-82. [DOI:10.1002/glia.20338] [PMID]
Shabani, M., Haghani, M., Sheibani, V., & Janahmadi, M. (2009). Changes in motor and learning behaviors of rats prenatally exposed to WIN 55212-2, a cannabinoid receptor agonist. Physiology and Pharmacology, 13(2), 120-9. https://www.sid.ir/en/journal/ViewPaper.aspx?id=167206
Tang, X. N., Wang, Q., Koike, M. A., Cheng, D., Goris, M. L., & Blankenberg, F. G., et al. (2007). Monitoring the protective effects of minocycline treatment with radiolabeled annexin V in an experimental model of focal cerebral ischemia. Journal of Nuclear Medicine, 48(11), 1822-8. [DOI:10.2967/jnumed.107.041335] [PMID]
Tikka, T., Fiebich, B. L., Goldsteins, G., Keinänen, R., & Koistinaho, J. (2001). Minocycline, a tetracycline derivative, is neuroprotective against excitotoxicity by inhibiting activation and proliferation of microglia. The Journal of Neuroscience, 21(8), 2580. [DOI:10.1523/JNEUROSCI.21-08-02580.2001] [PMID] [PMCID]
Vaziri, Z., Abbassian, H., Sheibani, V., Haghani, M., Nazeri, M., & Aghaei, I., et al. (2015). The therapeutic potential of Berberine chloride hydrate against harmaline-induced motor impairments in a rat model of tremor. Neuroscience letters, 590, 84-90. [DOI:10.1016/j.neulet.2015.01.078] [PMID]
Wang, X., Zhu, S., Drozda, M., Zhang, W., Stavrovskaya, I. G., & Cattaneo, E., et al. (2003). Minocycline inhibits caspase-independent and-dependent mitochondrial cell death pathways in models of Huntington’s disease. Proceedings of the National Academy of Sciences, 100(18), 10483-7. [DOI:10.1073/pnas.1832501100] [PMID] [PMCID]
Whaley, N. R., Putzke, J. D., Baba, Y., Wszolek, Z. K., & Uitti, R. J. (2007). Essential tremor: Phenotypic expression in a clinical cohort. Parkinsonism Relat Disord, 13(6), 333-9. [DOI:10.1016/j.parkreldis.2006.12.004] [PMID]
Yang, L., Sugama, S., Chirichigno, J. W., Gregorio, J., Lorenzl, S., & Shin, D. H., et al. (2003). Minocycline enhances MPTP toxicity to dopaminergic neurons. Journal of Neuroscience Research, 74(2), 278-85. [DOI:10.1002/jnr.10709] [PMID]
Yrjänheikki, J., Tikka, T., Keinänen, R., Goldsteins, G., Chan, P. H., & Koistinaho, J. (1999). A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window. Proceedings of the National Academy of Sciences of the United States of America, 96(23), 13496-500. [DOI:10.1073/pnas.96.23.13496] [PMID] [PMCID]
Zhang, L., Kitaichi, K., Fujimoto, Y., Nakayama, H., Shimizu, E., & Iyo, M., et al. (2006). Protective effects of minocycline on behavioral changes and neurotoxicity in mice after administration of methamphetamine. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 30(8), 1381-93. [DOI:10.1016/j.pnpbp.2006.05.015] [PMID]
Zhu, S., Stavrovskaya, I. G., Drozda, M., Kim, B. Y., & Ona, V., Li, M., et al. (2002). Minocycline inhibits cytochrome c release and delays progression of amyotrophic lateral sclerosis in mice. Nature, 417(6884), 74-8. [DOI:10.1038/417074a] [PMID]
Type of Study: Original |
Subject:
Behavioral Neuroscience
Received: 2020/08/10 | Accepted: 2020/10/5 | Published: 2021/03/1
Received: 2020/08/10 | Accepted: 2020/10/5 | Published: 2021/03/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





