Volume 13, Issue 4 (July & August: Special Issue on Cognitive Sciences 2022)
BCN 2022, 13(4): 551-572 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Faridi F, Alvand A, Khosrowabadi R. Brain Structural Correlates of Intelligence in Attention Deficit Hyperactivity Disorder (ADHD) Individuals. BCN 2022; 13 (4) :551-572
URL: http://bcn.iums.ac.ir/article-1-1560-en.html
URL: http://bcn.iums.ac.ir/article-1-1560-en.html
1- Institute for Cognitive and Brain Sciences, Shahid Beheshti University, Tehran, Iran.
Keywords: Attention deficit hyperactivity disorder (ADHD), Magnetic Resonance Imaging, Intelligence, Grey matter density
Full-Text [PDF 5229 kb]
| Abstract (HTML)
Full-Text:
1. Introduction
Intelligence is a capacity involving planning, reasoning, comprehension, abstraction, and learning (Gottfredson, 1997) that can predict critical life outcomes (Deary, Johnson, & Houlihan, 2009). Therefore, understanding the biological systems taking apart intelligence can be necessary for psychological science (Deary et al., 2009). The neural basis of intelligence has been investigated by a range of neuroimaging studies, including magnetic resonance imaging (MRI) (McDaniel, 2005), electroencephalogram (EEG) (Thatcher, North & Biver, 2005), positron emission tomography (PET) (Tang et al., 2014), task-based functional magnetic resonance imaging (fMRI) (Jung & Haier, 2007) and resting-state fMRI (Li & Tian, 2014). Most structural studies associate intelligence quotient (IQ) with frontal and parietal lobes (Anderson et al., 2004; Basten, Hilger & Fiebach, 2015; Vakhtin, Ryman, Flores, & Jung, 2014; Jung & Haier, 2007). For instance, a relationship between grey matter (GM) volume in the posterior cingulate cortex (PCC) and intelligence has been reported (Deary, Penke, & Johnson, 2010). Moreover, the involvement of subcortical regions, such as the right striatum (Burgaleta et al., 2014), basal ganglia (McNab & Klingberg, 2008), hippocampus (Burgess, Maguire, & O’Keefe, 2002), and caudate (G: has been observed. In addition, the relationship between (GM) volume and cognitive deficits have been investigated in lesion studies (Karussis, Leker, & Abramsky, 2000), (Pinter, Eliez, Schmitt, Capone, & Reiss, 2001). Despite these findings, studies addressing the relationship between brain structures with intelligence in the attention deficit hyperactivity disorder (ADHD) population are rare (Vilgis, Sun, Chen, Silk, & Vance, 2016; McAlonan et al., 2007).
In this study, we hypothesized that the structural basis of intelligence should not be different in normal and ADHD individuals. Therefore, 56 ADHD individuals, including 30 individuals from the combined and 26 individuals from the inattentive subcategory, and 30 healthy individuals in the control group were scanned by MRI. Then, GM densities of anatomically separated brain regions were estimated from the brain images. Subsequently, Pearson’s correlation analysis was employed to detect the association between the GM densities of the brain regions and IQ. The whole pattern of association is also estimated. Then, analysis was performed at the group level and the correlation of IQ and GM densities in ADHD groups were compared with each other as well as the healthy control group. Furthermore, the differential pattern of GM density was observed in three groups. The main aim of the study was to find out whether the relationship between GM density and IQ in ADHD is similar to healthy individuals or is aligned with the disorder.
2. Materials and Methods
Study participants
A total of 56 ADHD subjects, including 30 combined subjects and 26 inattentive subjects, as well as 30 healthy subjects in the control group, with similar age and IQ range, underwent a session of MRI scanning. Table 1 presents the demographic information of each group. According to age, IQ, and handedness, no significant differences were observed among the groups (P<0.05).
.jpg)
Children in the ADHD group had to meet diagnostic criteria defined by the diagnostic and statistical manual, fourth edition, text revision (DSM-IV-TR) for ADHD as well as determine parent and child’s answers to the kiddie schedule for affective disorders and schizophrenia present and lifetime version (KSADS-PL) confirmed by a psychiatrist to be involved in this study. The exclusion criteria included comorbid mood or anxiety disorder, autistic or Asperger’s disorder, medical illness that was unstable or could cause psychiatric symptoms, or substance abuse within 2 months of participation.
Assessment of cognitive performance: intelligence quotient (IQ) scores
All participants completed the Wechsler abbreviated scale of intelligence (WASI) presented in Table 1 . The Wechsler abbreviated scale of intelligence (WASI) is general intelligence, or IQ test designed to measure overall cognitive abilities or a specific cognitive capability in individuals in the age range of 6-89 years. (American Psychological Association (APA PsycNet))
The experimental validation of our proposed method was performed on a dataset gathered from 4 data centers including Kennedy Krieger Institute (KKI), Oregon Health and Science University (OHSU), Peking University (PEK), and New York University (NYU). These data are related to ADHD-200 global completion and are publically.
Magnetic Resonance Imaging (MRI) data acquisition
All participants underwent a T1-weighted high-resolution MRI scanning using a siemens3T scanner system. The imaging data were collected using the following protocols, Kennedy Krieger Institute (KKI) (relaxation time [T1]: Contrast enhancement, echo time [TE]: Shortest, repetition time [TR]: Shortest, flip angle: 8°, field of view [FOV]: 256 mm, slice thickness: 1 mm); Oregon Health and Science University (OHSU) (T1: 900 ms, TE: 3.58 ms, TR: 2300 ms, flip angle: 10°, FOV: 256 mm, slice thickness: 1.1 mm); Peking University (PEK) (T1: 1100 ms, TE: 3.45 ms, TR:2530 ms, flip angle: 7°, FOV: 256mm, slice thickness: 1mm); New York University (NYU) (T1: 1100 ms, TE: 3.25 ms, TR: 2530 ms, flip angle: 7°, FOV: 256 mm, slice thickness: 1.33 mm).
MRI data processing
A standard processing pipeline was performed on the MRI data using the functional MRI of the brain (FMRIB) Software Library (FSL) and Analysis Of Functional Neuroimaging (AFNI). First, the brain MRI images were deobliqued, then re-oriented and the skull strip was removed. Then, images were registered to the MNI space. After that, MRI images were segmented into different tissue types and were parcellated into 392 regions of interest (ROI) using MNI normalized Craddock atlas (Craddock, James, Holtzheimer, Hu, & Mayberg, 2012) or 116 regions using Automated Anatomical Labeling (AAL) (Tzourio-Mazoyer et al., 2002). Results of the Craddock method are presented in the main text body and results of the AAL parcellation are provided in the supplementary materials.
Statistical analysis
Pearsons’ correlation was calculated between IQ and GM densities of separate brain regions and a significant relationship was recognized by setting a threshold of P<0.05. Then statistical analysis was performed using the statistical analysis toolbox of MATLAB 2015. Later, the group comparison of correlation values was performed using the cocor package (Diedenhofen & Musch, 2015). Then, the brain net viewer (Xia, Wang, & He, 2013) was used to visualize the significant results. The whole association pattern is also acquired using SPSS software. We also observed a differential pattern of GM density in groups as a second approach (Figure 1)
.jpg)
It should be mentioned that suggested regions of interest (ROIs) in the Craddock atlas were too fragmented (392 regions), so for the ease of presentation, the significant results were re-indexed to the frontal, occipital, temporal, parietal, cerebellum, and subcortical structures.
3. Results
Significant structural correlates of intelligence quotient (IQ)
The relationship between IQ scores and GM densities was found in several brain regions. Tables 2, 3, 4 and Figures 2, 3, 4 show the detailed information of significant results (P<0.05). Significant positive relationships between GM density and intelligence are shown in red and negative relationships are shown in blue.
In ADHD combined group, a positive correlation was observed between IQ scores and GM density in the frontal region (inferior operculum and orbitofrontal on the right side), temporal region (superior part in the left hemisphere, inferior part in the right hemisphere, and middle part bilaterally), occipital region (inferior part bilaterally), cerebellum (CRB 4-5 in the left hemisphere and CRB-Crus1, in the right side and CRB-Crus2 bilaterally), angular gyrus in the right side, fusiform in the right side, and the precentral region on the right side. In addition, negative correlations were observed in the left inferior temporal region and the left fusiform region (Figure 2, Table 2).
.jpg)
.jpg)
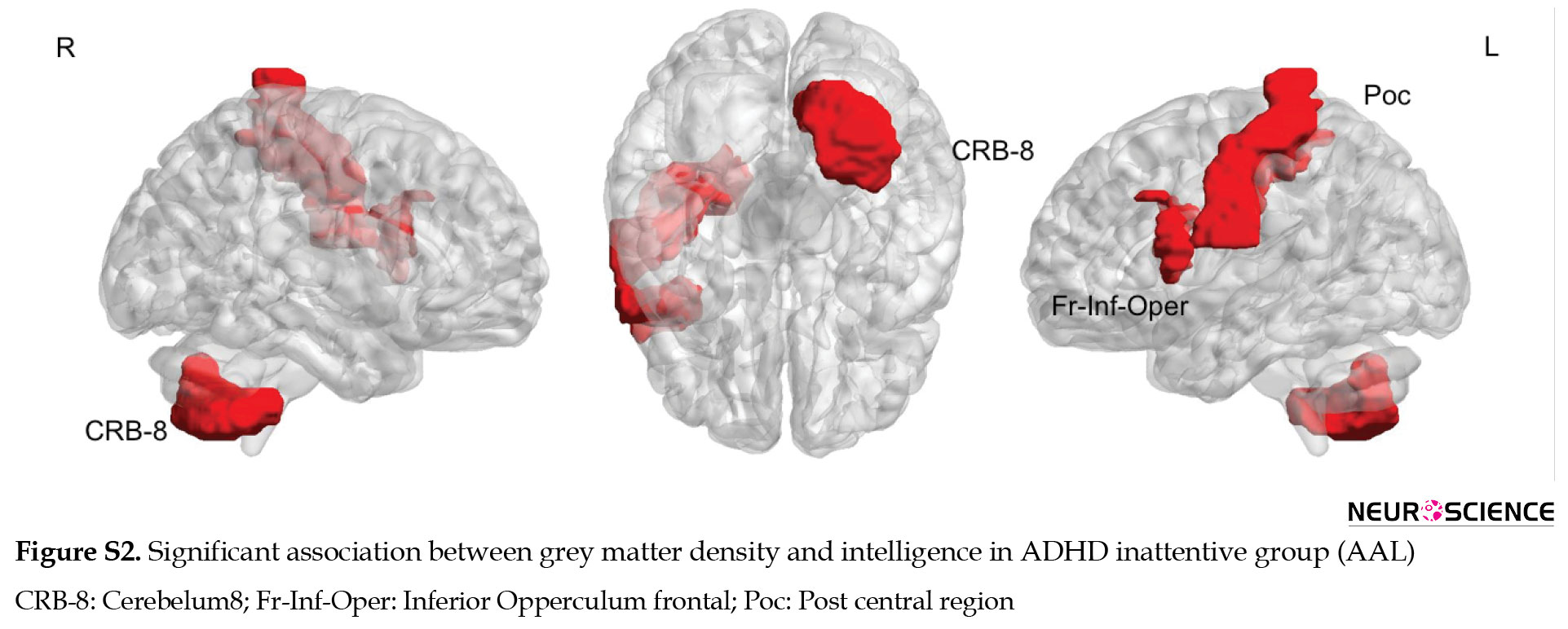
In ADHD inattentive group, no significant negative correlation was observed between IQ and GM density of the brain regions. While a positive correlation was identified in bilateral fusiform areas, caudate and cerebellum crus1, and the inferior parietal region in the right hemisphere (Figure 3, Table 3)
.jpg)
.jpg)
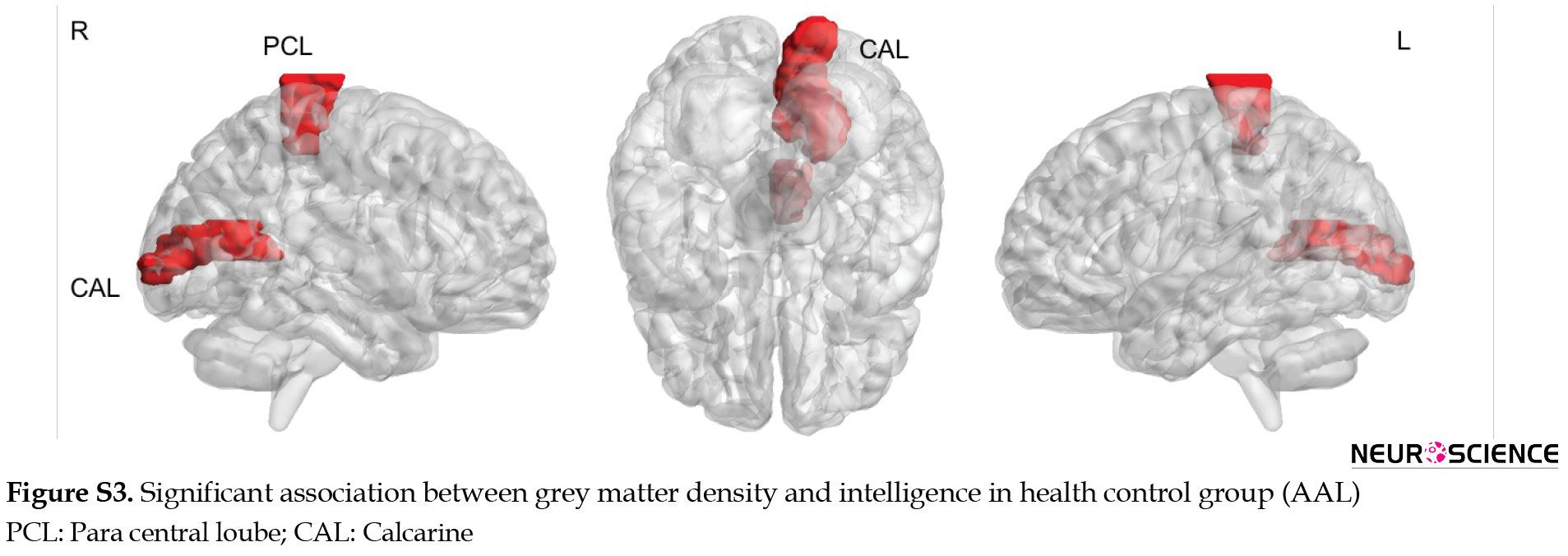
In the healthy control group, a positive and significant correlation was observed bilaterally in the precuneus, and fusiform. In the left hemisphere, a positive correlation was seen in temporal regions (inferior and superior temporal pole), calcarine, superior occipital, and middle frontal regions. In the right hemisphere, a positive correlation was observed at cerebellum 6, frontal inferior operculum, and cingulate cortex. Moreover, a negative correlation was identified in the left superior medial frontal cortex (Figure 4, Table 4).
.jpg)
.jpg)
Comparing the cluster size of each section showed that the most significant relationship for the ADHD combined group is located in the temporal, cerebellum, and frontal regions. In the ADHD inattentive group, the most significant relationship was observed in the temporal, subcortical region, and cerebellum. And in the healthy control group, the most significant relationship was observed in the temporal, frontal, and parietal regions (Table 5).
.jpg)
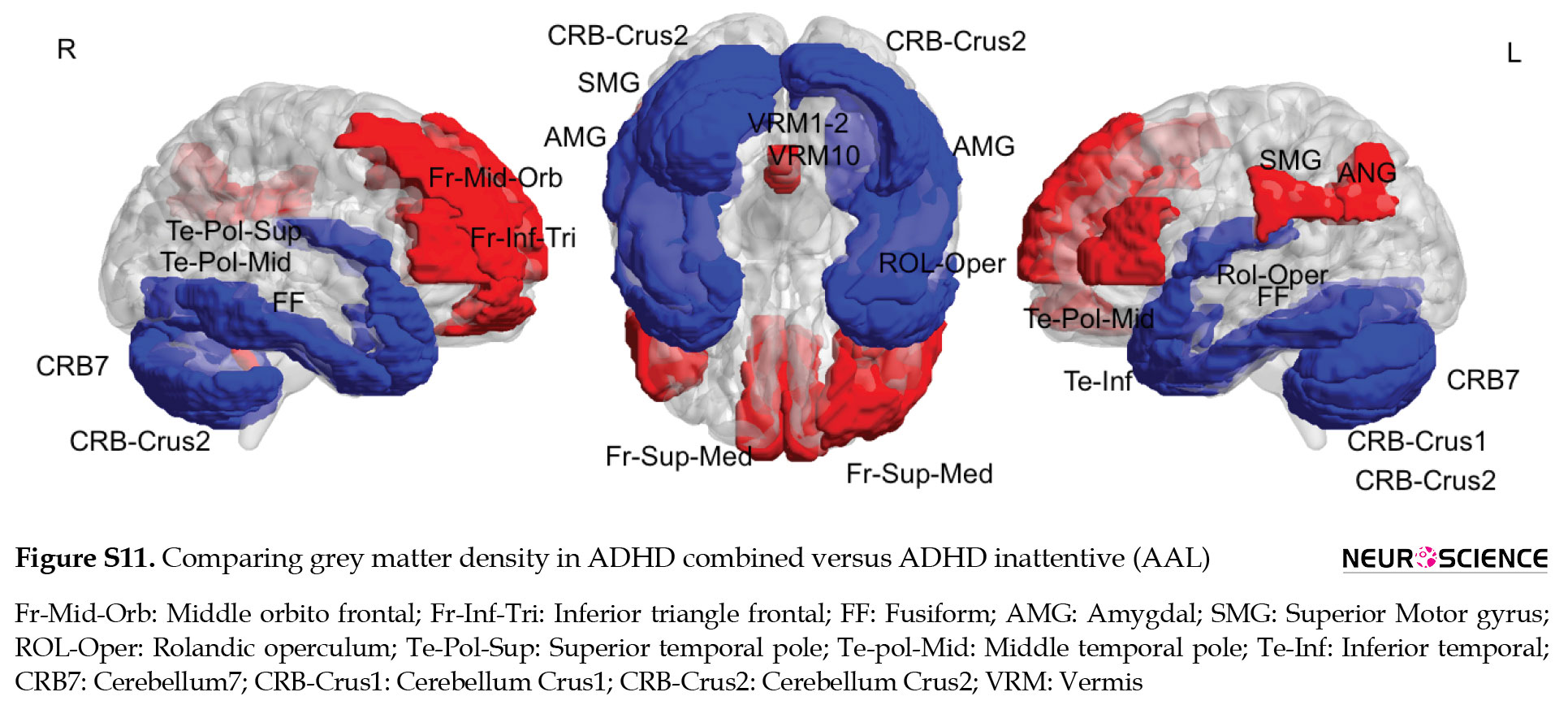
The results of the relationship between GM density and intelligence via AAL parcellations are presented in the supplementary materials (Figures S1, S2, S3, Table S1).


Group comparison
Figure 5, 6, 7 and Table 6 present significant results of comparing correlations observed in each group (significant P-values have been bolded in Table 6).
.jpg)
.jpg)
.jpg)
.jpg)
Significantly increased relationships between GM density and intelligence are shown in red and decreased relationships are shown in blue.
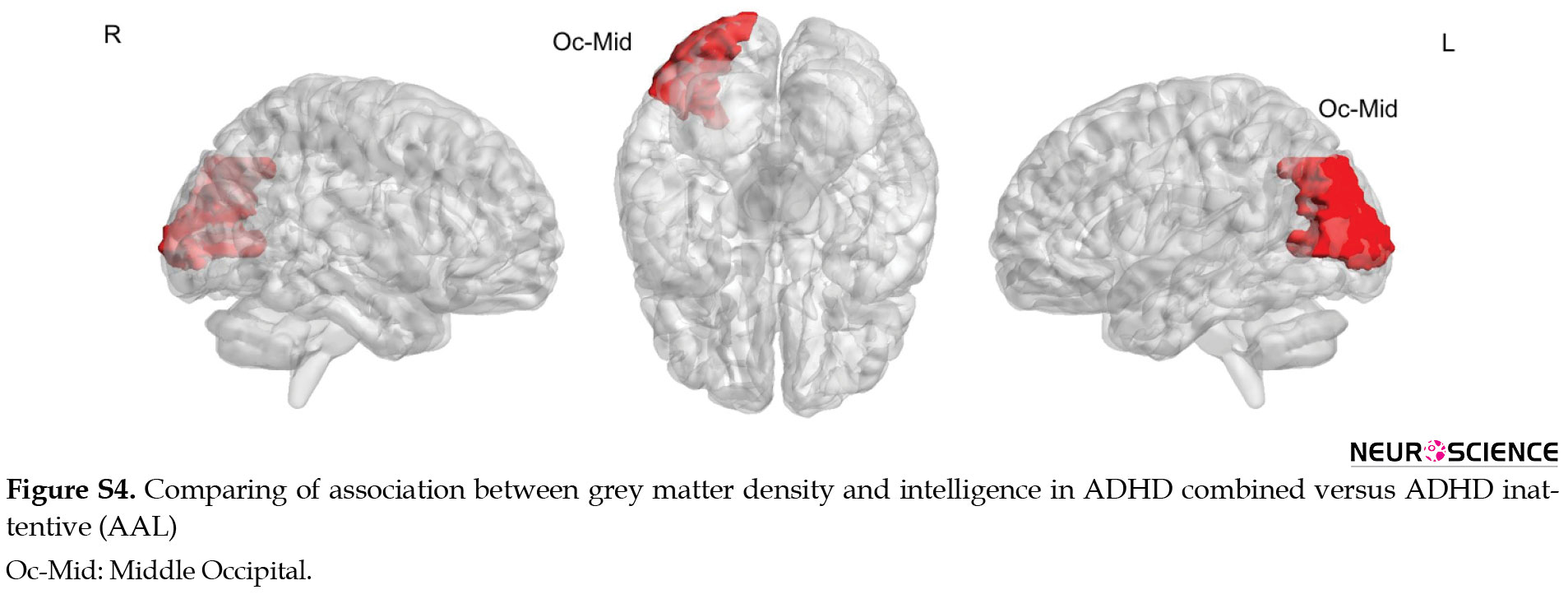
Comparing ADHD combined versus ADHD inattentive showed significantly increased associated GM density with intelligence at superior temporal and cerebellum-6 in the left hemisphere and angular region in the right hemisphere. Decreased associated grey matter density with intelligence was observed at the middle cingulate on the right side (Figure 5).
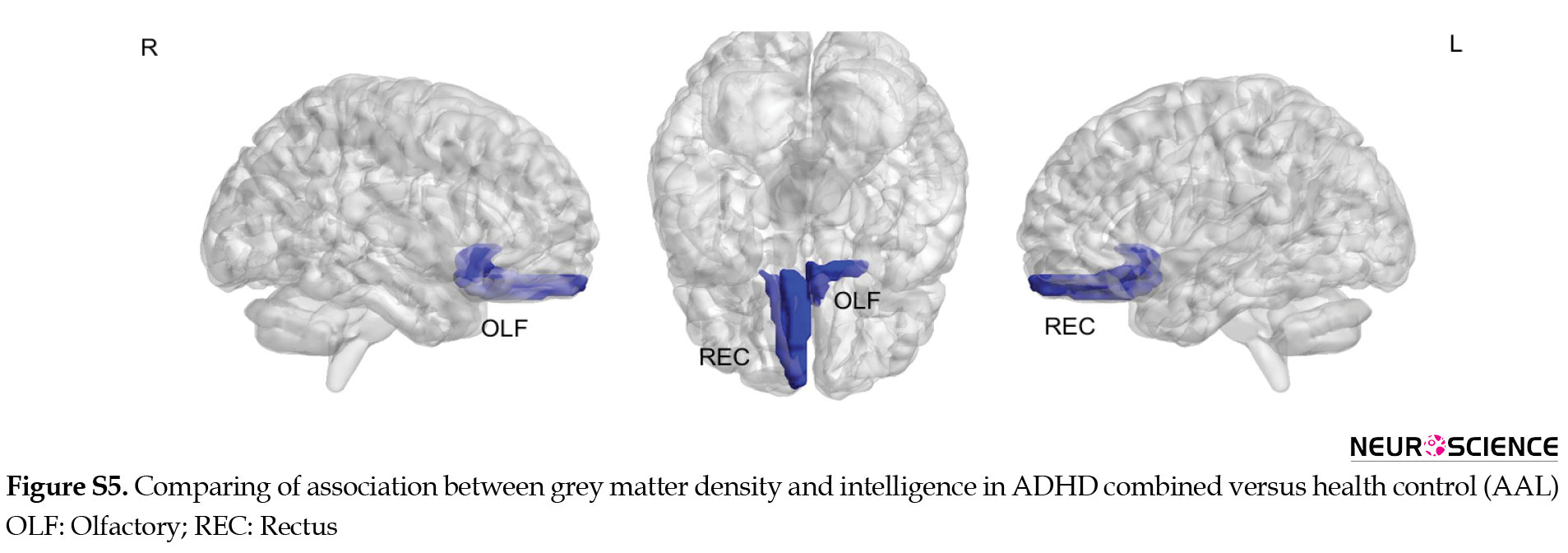
ADHD combined compared to the healthy control had an increased relationship between GM density and intelligence at middle temporal and orbitofrontal in the right hemisphere as well as rectus and superior parietal on the left side. Decreased relationship in GM density was seen at the inferior occipital, medial orbitofrontal, and superior frontal in the right hemisphere (Figure 6).
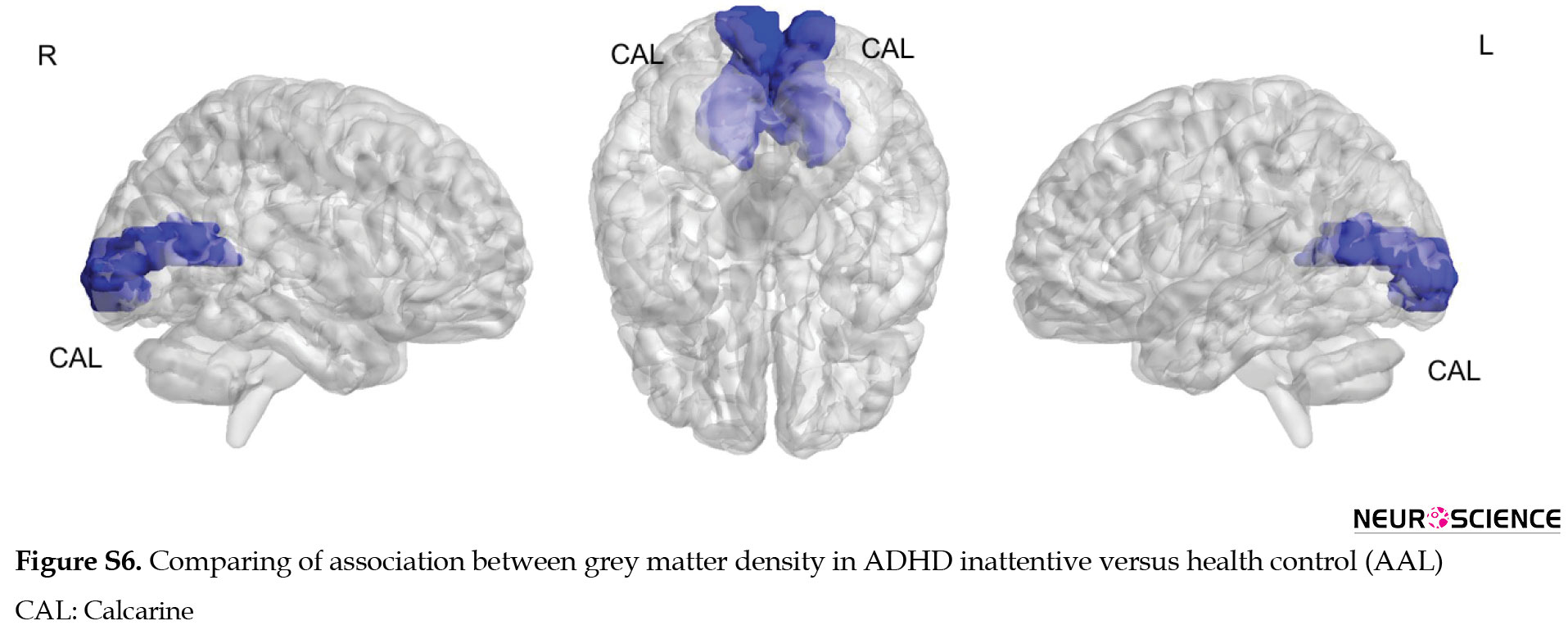
ADHD inattentive compared to the healthy control group showed increased associated GM density with intelligence in the middle and anterior cingulate and superior motor area in the right hemisphere as well as the precuneus, superior parietal, and rectus on the left side. Decreased relationship of GM density with intelligence was observed in the inferior occipital, putamen, caudate, medial orbitofrontal, and precentral region in the right hemisphere as well as middle occipital and cerebellum6 on the left side (Figure 7).
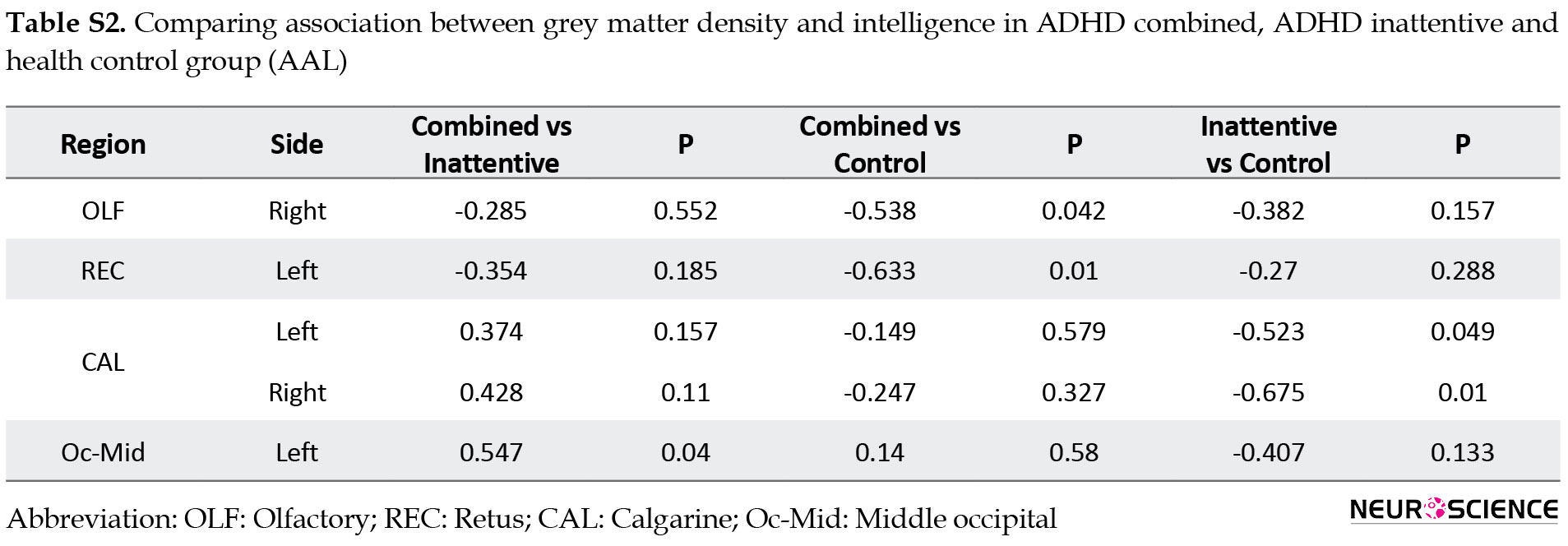
Compared to healthy control, ADHD groups (combined and inattentive) had increased associated GM density with intelligence at superior parietal and rectus in the left hemisphere, while decreased associated GM density with intelligence was seen at inferior occipital and medial frontal in the right side. The results of comparing the relationship between GM density and intelligence via AAL software are presented in supplementary materials (Figures S4, S5, S6, Table S2).
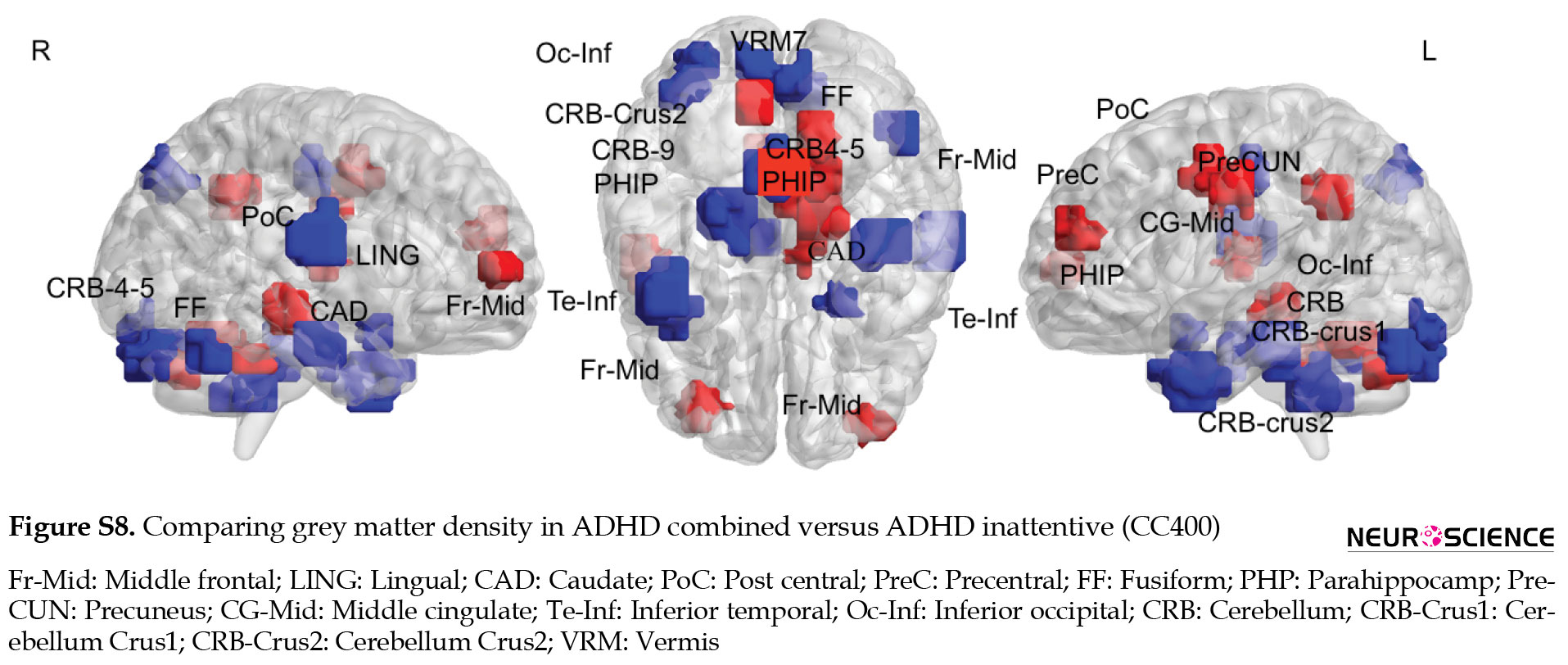
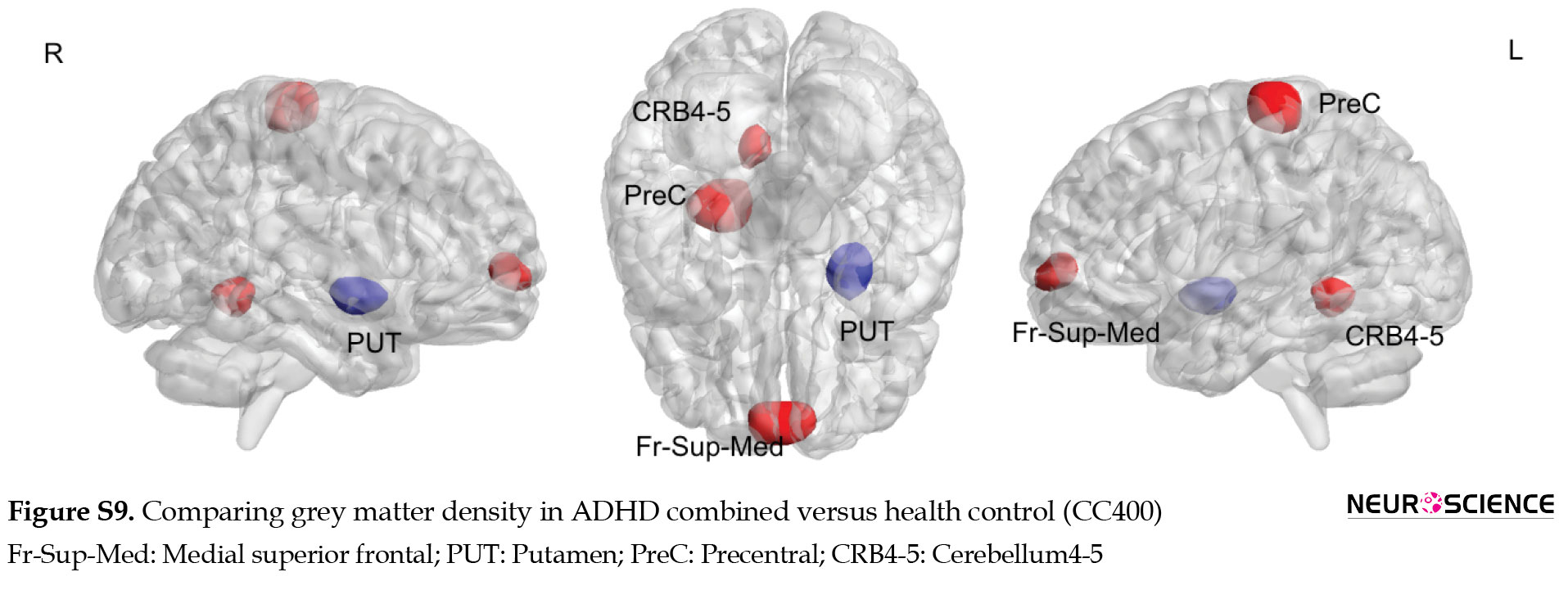
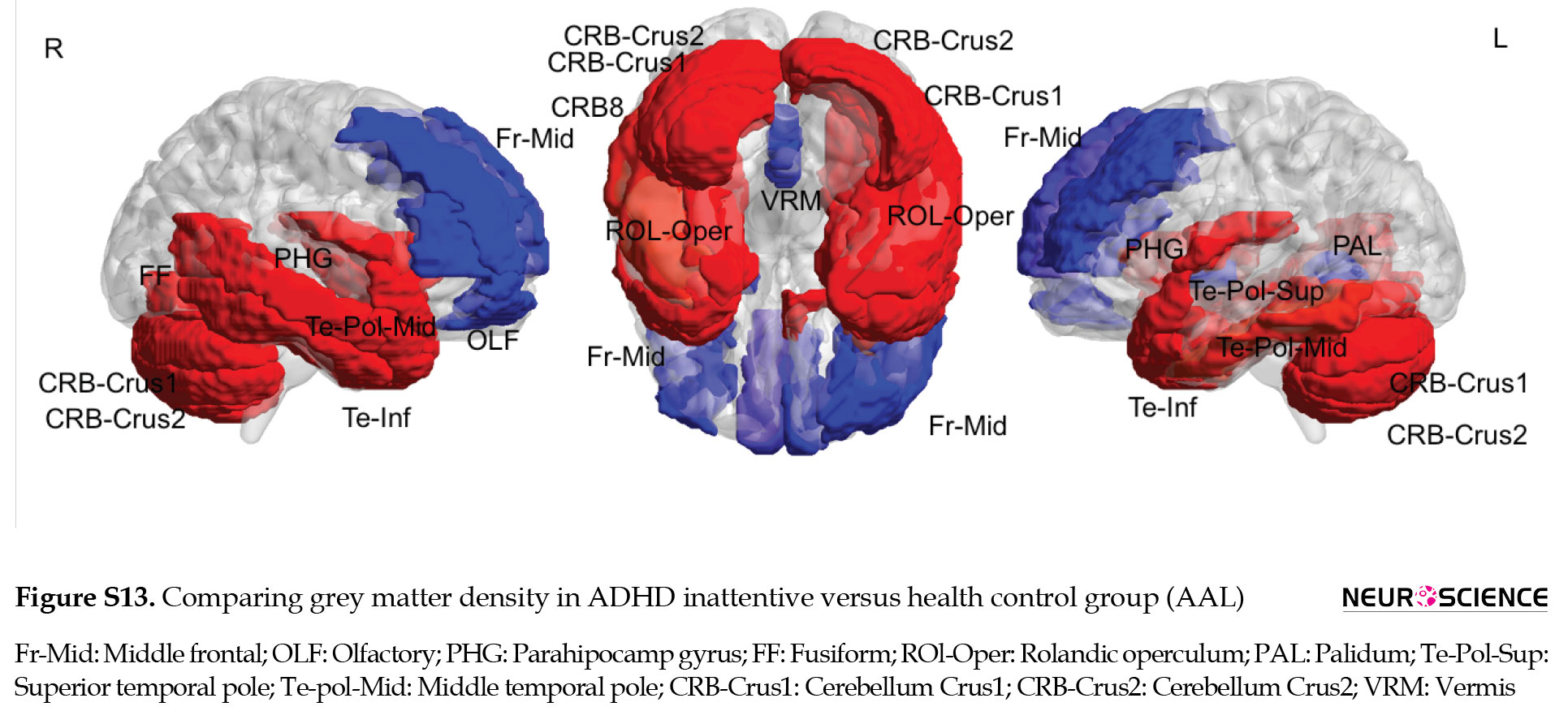
Multiple comparisons of whole brain association patterns also showed differences between groups. Although ADHD combined was significantly different from the other groups, this was not the case for ADHD inattentive group (Table 7), (Figure S14). We also compare raw data of grey matter density to show structural differences in terms of grey matter density whithin groups. Results from CC400 are shown in Table S3, Figures S8, S9, S10 and those of AAL116 are presented in Table S4, Figures S11, S12, S13).

.jpg)


4. Discussion
ADHD is a heritable phenotype that can be influenced by genetic factors. Therefore, genetics can influence brain morphometric properties in ADHD as reported in previous studies (Bellgrove et al., 2005; Swanson et al., 2007). Intelligence is also a heritable phenotype and overlapping effects of genetics on the intelligence score and ADHD also can be investigated. If IQ score is only related to brain morphometry properties, there should not be any significant differences between IQ-matched ADHD and control individuals. Otherwise, the relationship between IQ score and brain anatomy follows another mechanism that we investigate in this study. In this study, the regional GM density and full-scale IQ were compared between two groups of ADHD subtypes, including a combined and inattentive and a healthy control group. Two different parcellation strategies, including functionally separated regions (CC400) and anatomically separated regions (AAL116), were applied to detach the brain to 392 and 116 regions, respectively. After statistical analysis, it was observed that the relationship of IQ with regional GM density is mainly positive and located in the right hemisphere. The right hemisphere plays a vital role in cognition (Robertson, 2014), language (Gainotti, 2013), arithmetic (Knops & Willmes, 2014), and visuospatial attention (Longo, Trippier, Vagnoni, & Lourenco, 2015). These findings are not impressed by the parcellation technique and the results of CC400 were almost the same as AAL116.
The changes in GM density may alter behavior and cognitive functions discussed in the following. Our findings of the healthy control group showed a significant positive relationship between GM density and IQ score in the inferior and middle frontal, precuneus, fusiform, inferior temporal, calcarine, superior occipital, and middle cingulum, and cerebelum6. The inferior frontal is involved in selecting responses in uncertain situations (Frangou, Chitins, & Williams, 2004) and reasoning ability (Goel, Gold, Kapur, & Houle, 1997), and the relationship of frontal and parietal with intelligence has been shown in previous studies (Jung & Haier, 2007). Another finding was the inferior part of the temporal lobe which is involved in the analysis of visual form, motion, and representation of individuals and is directly related to the intelligence score (Frangou et al., 2004). Our results about fusiform may also reflect its role in language proficiency (Tan et al., 2011), recognition, and elaboration on the visual inputs (Colom et al., 2009). Furthermore, the significant relationship between cingulum GM density and IQ, observed in this study, in the healthy control group may reflect its role in spatial learning and memory (Aggleton, Neave, Nagle, & Sahgal, 1995). Last but not least, the relationship of cerebellum GM density with intelligence has also been reported in previous studies (Stoodley & Schmahmann, 2009). These findings confirm the results compared to previous studies.
On the other hand, ADHD showed a significantly different mechanism and pattern of association between GM density and intelligence, compared to the healthy control group. Comparing the whole association pattern, also suggest a discrepancy between the groups. The difference between ADHD combined and control group was more significant than the difference between ADHD inattentive and control, which seems rational because of more severe deficit symptoms in ADHD combined.
The IQ score represents a score for verbal and visual abilities, fluid reasoning, working memory, and processing speed (Wechsler, 1949). In previous studies, the strengths and weaknesses of ADHD individuals in these cognitive functions have been reported (Frazier, Demaree, & Youngstrom, 2004). Mayes’s group has tried to suggest a neurobiological basis for ADHD by emphasizing ADHD strengths in verbal, and visual reasoning and their weakness in attention, processing speed, and graph motor skill (Mayes & Calhoun, 2006). Given that the ADHD and healthy control groups in our study are matched for IQ, observed differences in associated GM density with IQ between ADHD and healthy control groups may suggest a compensatory mechanism in ADHD to maintain adequate performance. This mechanism can be discussed in our research findings. In ADHD groups, combined and inattentive, a decreased association between GM density and intelligence score was observed in the right inferior occipital and right medial frontal regions. The inferior occipital region is involved in visual imaging (Goldenberg et al., 1989) and the medial frontal is engaged in the switching tasks (Rushworth, Hadland, Paus, & Sipila, 2002). In contrast, an increased association was observed between the left superior parietal and the left rectus. The superior parietal region is involved in the manipulation of information in working memory (Koenigs, Barbey, Postle, & Grafman, 2009) and the rectus is engaged in the paradigm of reward learning (Kringelbach, 2005). Therefore, the lessened association between GM density and intelligence in the right hemisphere (inferior occipital and medial frontal) could be compensated by the increased association in the left hemisphere (superior parietal and rectus). This mechanism helps the ADHD subject to maintain an adequate level of cognitive performance.
Comparing row data of GM density in groups also reinforced the idea of a compensatory mechanism. In this regard, GM differences between ADHD inattentive and control group in our study was greater than between ADHD combined and control. This alteration in GM density may help ADHD inattentive individuals compensate for their brain deficiency and perform compared to ADHD combined.
Previous studies have shown that the cortical compensatory mechanisms can cope with the deficit in several cognitive processes in ADHD (Ma et al., 2012). Hence, a different association between regional GM density with the IQ score in the ADHD and control group may indicate that brain structure is not the only determiner of intelligence; rather, intelligence may also be underpinned by neural dynamics of the brain.
Limitation
The results of this study had several limitations; for example, the correlation was done for full-scale IQ and wechsler intelligence scale for children (WISC) subtests were not analyzed. Moreover, this study only focused on male participants in the age range of 8 to 13 years, which can be extended in future works. Therefore, a developmental study on both genders could provide additional insights into the mechanism of structural changes in the brain involved with intelligence.
5. Conclusion
To the best of our knowledge, this is the first study to investigate brain structural correlates of IQ in ADHD individuals. The study provides evidence that IQ may be closely related to GM density in specific brain regions but the pattern can be influenced by a disorder. The significant changes observed in the association scores of the ADHD individuals compared to the healthy individuals in the control group suggest a compensatory mechanism for having a suitable cognitive performance (IQ). Our findings indicate that IQ score may be affected by neural dynamics; therefore, the structural covariates can be a better alternative for the GM density. We hope that these findings can provide additional information for a better understanding of the relationship between brain morphometry and intelligence.
Ethical Considerations
Compliance with ethical guidelines
All ethical principals are considered in this article. The participants were informed of the purpose of the research and its implementation stages. They were also assured about the confidentiality of their information and were free to leave the study whenever they wished and if desired, the research results would be available to them. A written cosent has been obtain from the subjects. Priciples of the Helski convention was also observed.
Funding
This research did not receive any grant from funding agencies in the public commercial, or non-profit sectors.
Authors' contributions
All authors equally contributed to preparing this article.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the ADHD-200 consortium for generously sharing data.
References
Aggleton, J. P., Neave, N., Nagle, S., & Sahgal, A. (1995). A comparison of the effects of medial prefrontal, cingulate cortex, and cingulum bundle lesions on tests of spatial memory: Evidence of a double dissociation between frontal and cingulum bundle contributions. Journal of Neuroscience, 15(11), 7270-7281. [DOI:10.1523/JNEUROSCI.15-11-07270.1995] [PMID] [PMCID]
Anderson, J. R., Bothell, D., Byrne, M. D., Douglass, S., Lebiere, C., & Qin, Y. (2004). An integrated theory of the mind. Psychological Review, 111(4), 1036. [DOI:10.1037/0033-295X.111.4.1036] [PMID]
Basten, U., Hilger, K., & Fiebach, C. J. (2015). Where smart brains are different: A quantitative meta-analysis of functional and structural brain imaging studies on intelligence. Intelligence, 51, 10-27. [DOI:10.1016/j.intell.2015.04.009]
Bellgrove, M. A., Hawi, Z., Lowe, N., Kirley, A., Robertson, I. H., & Gill, M. (2005). DRD4 gene variants and sustained attention in Attention Deficit Hyperactivity Disorder (ADHD): Effects of associated alleles at the VNTR and− 521 SNP. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics, 136(1), 81-86. [DOI:10.1002/ajmg.b.30193] [PMID]
Burgaleta, M., MacDonald, P. A., Martínez, K., Roman, F. J., Álvarez-Linera, J., González, A. R., et al. (2014). Subcortical regional morphology correlates with fluid and spatial intelligence. Human Brain Mapping, 35(5), 1957-1968. [DOI:10.1002/hbm.22305] [PMID] [PMCID]
Burgess, N., Maguire, E. A., & O’Keefe, J. (2002). The human hippocampus and spatial and episodic memory. Neuron, 35(4), 625-641. [DOI:10.1016/S0896-6273(02)00830-9]
Colom, R., Haier, R. J., Head, K., Álvarez-Linera, J., Quiroga, M. Á., Shih, P. C., & Jung, R. E. (2009). Gray matter correlates of fluid, crystallized, and spatial intelligence: Testing the P-FIT model. Intelligence, 37(2), 124-135. [DOI:10.1016/j.intell.2008.07.007]
Craddock, R. C., James, G. A., Holtzheimer, P. E., Hu, X. P., & Mayberg, H. S. (2012). A whole brain fMRI atlas generated via spatially constrained spectral clustering. Human Brain Mapping, 33(8), 1914-1928. [DOI:10.1002/hbm.21333] [PMID] [PMCID]
Deary, I. J., Johnson, W., & Houlihan, L. M. (2009). Genetic foundations of human intelligence. Human Genetics, 126(1), 215-232. [DOI:10.1007/s00439-009-0655-4] [PMID]
Deary, I. J., Penke, L., & Johnson, W. (2010). The neuroscience of human intelligence differences: Nature reviews. Neuroscience, 11(3), 201. [DOI:10.1038/nrn2793] [PMID]
Diedenhofen, B., & Musch, J. (2015). cocor: A comprehensive solution for the statistical comparison of correlations. PloS one, 10(4), e0121945. [DOI:10.1371/journal.pone.0121945] [PMID] [PMCID]
Frangou, S., Chitins, X., & Williams, S. C. (2004). Mapping IQ and gray matter density in healthy young people. Neuroimage, 23(3), 800-805. [DOI:10.1016/j.neuroimage.2004.05.027] [PMID]
Frazier, T. W., Demaree, H. A., & Youngstrom, E. A. (2004). Meta-analysis of intellectual and neuropsychological test performance in Attention-Deficit/Hyperactivity Disorder. Neuropsychology, 18(3), 543-55. [DOI:10.1037/0894-4105.18.3.543] [PMID]
Gainotti, G. (2013). The contribution of language to the right-hemisphere conceptual representations: A selective survey. Journal of clinical and experimental neuropsychology, 35(6), 563-572. [DOI:10.1080/13803395.2013.798399] [PMID]
Goel, V., Gold, B., Kapur, S., & Houle, S. (1997). The seats of reason? An imaging study of deductive and inductive reasoning. NeuroReport, 8(5), 1305-1310. [DOI:10.1097/00001756-199703240-00049] [PMID]
Goldenberg, G., Podreka, I., Steiner, M., Willmes, K., Suess, E., & Deecke, L. (1989). Regional cerebral blood flow patterns in visual imagery. Neuropsychologia, 27(5), 641-664. [DOI:10.1016/0028-3932(89)90110-3]
Grazioplene, R. G.,g Ryman, S., Gray, J. R., Rustichini, A., Jung, R. E., & DeYoung, C. G. (2015). Subcortical intelligence: Caudate volume predicts IQ in healthy adults. Human Brain Mapping, 36(4), 1407-1416. [DOI:10.1002/hbm.22710] [PMID] [PMCID]
Jung, R. E., & Haier, R. J. (2007). The Parieto-Frontal Integration Theory (P-FIT) of intelligence: Converging neuroimaging evidence. Behavioral and Brain Sciences, 30(2), 135-154. [DOI:10.1017/S0140525X07001185] [PMID]
Karussis, D., Leker, R., & Abramsky, O. (2000). Cognitive dysfunction following thalamic stroke: A study of 16 cases and review of the literature. Journal of the Neurological Sciences, 172(1), 25-29. [DOI:10.1016/S0022-510X(99)00267-1]
Knops, A., & Willmes, K. (2014). Numerical ordering and symbolic arithmetic share frontal and parietal circuits in the right hemisphere. Neuroimage, 84, 786-795. [DOI:10.1016/j.neuroimage.2013.09.037] [PMID]
Koenigs, M., Barbey, A. K., Postle, B. R., & Grafman, J. (2009). Superior parietal cortex is critical for the manipulation of information in working memory. Journal of Neuroscience, 29(47), 14980-14986. [DOI:10.1523/JNEUROSCI.3706-09.2009] [PMID] [PMCID]
Kringelbach, M. L. (2005). The human orbitofrontal cortex: linking reward to hedonic experience. Nature Reviews Neuroscience, 6(9), 691-702. [DOI:10.1038/nrn1747] [PMID]
Li, C., & Tian, L. (2014). Association between resting-state coactivation in the parieto-frontal network and intelligence during late childhood and adolescence. American Journal of Neuroradiology, 35(6), 1150-1156. [DOI:10.3174/ajnr.A3850] [PMID] [PMCID]
Longo, M. R., Trippier, S., Vagnoni, E., & Lourenco, S. F. (2015). Right hemisphere control of visuospatial attention in near space. Neuropsychologia, 70, 350-357. [DOI:10.1016/j.neuropsychologia.2014.10.035] [PMID]
Ma, J., Lei, D., Jin, X., Du, X., Jiang, F., Li, F., et al. (2012). Compensatory brain activation in children with Attention Deficit/Hyperactivity Disorder during a simplified Go/No-go task. Journal of Neural Transmission, 119(5), 613-619. [DOI:10.1007/s00702-011-0744-0] [PMID]
Mayes, S. D., & Calhoun, S. L. (2006). WISC-IV and WISC-III profiles in children with ADHD. Journal of Attention Disorders, 9(3), 486-493. [DOI:10.1177/1087054705283616] [PMID]
McAlonan, G. M., Cheung, V., Cheung, C., Chua, S. E., Murphy, D. G., Suckling, J., et al. (2007). Mapping brain structure in attention deficit-hyperactivity disorder: A voxel-based MRI study of regional grey and white matter volume. Psychiatry Research: Neuroimaging, 154(2), 171-180. [DOI:10.1016/j.pscychresns.2006.09.006] [PMID]
McDaniel, M. A. (2005). Big-brained people are smarter: A meta-analysis of the relationship between in vivo brain volume and intelligence. Intelligence, 33(4), 337-346. [DOI:10.1016/j.intell.2004.11.005]
McNab, F., & Klingberg, T. (2008). Prefrontal cortex and basal ganglia control access to working memory. Nature neuroscience, 11(1), 103. [DOI:10.1038/nn2024] [PMID]
Pinter, J. D., Eliez, S., Schmitt, J. E., Capone, G. T., & Reiss, A. L. (2001). Neuroanatomy of Down’s syndrome: A high-resolution MRI study. American Journal of Psychiatry, 158(10), 1659-1665. [DOI:10.1176/appi.ajp.158.10.1659] [PMID]
Robertson, I. H. (2014). A right hemisphere role in cognitive reserve. Neurobiology of Aging, 35(6), 1375-1385. [DOI:10.1016/j.neurobiolaging.2013.11.028] [PMID]
Rushworth, M., Hadland, K., Paus, T., & Sipila, P. (2002). Role of the human medial frontal cortex in task switching: A combined fMRI and TMS study. Journal of Neurophysiology, 87(5), 2577-2592. [DOI:10.1152/jn.2002.87.5.2577] [PMID]
Stoodley, C. J., & Schmahmann, J. D. (2009). Functional topography in the human cerebellum: A meta-analysis of neuroimaging studies. Neuroimage, 44(2), 489-501. [DOI:10.1016/j.neuroimage.2008.08.039] [PMID]
Swanson, J. M., Kinsbourne, M., Nigg, J., Lanphear, B., Stefanatos, G. A., Volkow, N., et al. (2007). Etiologic subtypes of Attention-Deficit/Hyperactivity Disorder: Brain imaging, molecular genetic and environmental factors and the dopamine hypothesis. Neuropsychology Review, 17(1), 39-59. [DOI:10.1007/s11065-007-9019-9] [PMID]
Tan, L. H., Chen, L., Yip, V., Chan, A. H., Yang, J., Gao, J.-H., et al. (2011). Activity levels in the left hemisphere caudate-fusiform circuit predict how well a second language will be learned. Proceedings of the National Academy of Sciences, 108(6), 2540-2544. [DOI:10.1073/pnas.0909623108] [PMID] [PMCID]
Tang, Z., Chen, Z., Zhai, Q., Hao, Y., Zhang, Y., & Zeng, X. (2014). Correlation between interictal cerebral glucose hypometabolism and IQ in children with epilepsy. Epilepsy & Behavior, 31, 15-18. [DOI:10.1016/j.yebeh.2013.10.028] [PMID]
Thatcher, R. W., North, D., & Biver, C. (2005). EEG and intelligence: Relations between EEG coherence, EEG phase delay and power. Clinical Neurophysiology, 116(9), 2129-2141. [DOI:10.1016/j.clinph.2005.04.026] [PMID]
Tzourio-Mazoyer, N., Landeau, B., Papathanassiou, D., Crivello, F., Etard, O., Delcroix, N., et al. (2002). Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage, 15(1), 273-289. [DOI:10.1006/nimg.2001.0978] [PMID]
Vakhtin, A. A., Ryman, S. G., Flores, R. A., & Jung, R. E. (2014). Functional brain networks contributing to the parieto-frontal integration theory of intelligence. Neuroimage, 103, 349-354. [DOI:10.1016/j.neuroimage.2014.09.055] [PMID]
Vilgis, V., Sun, L., Chen, J., Silk, T. J., & Vance, A. (2016). Global and local grey matter reductions in boys with ADHD combined type and ADHD inattentive type. Psychiatry Research: Neuroimaging, 254, 119-126. [DOI:10.1016/j.pscychresns.2016.06.008] [PMID]
Wechsler, D. (1949). Wechsler intelligence scale for children. [Link]
Xia, M., Wang, J., & He, Y. (2013). BrainNet Viewer: A network visualization tool for human brain connectomics. PloS One, 8(7), e68910. [DOI:10.1371/journal.pone.0068910] [PMID] [PMCID]
Intelligence is a capacity involving planning, reasoning, comprehension, abstraction, and learning (Gottfredson, 1997) that can predict critical life outcomes (Deary, Johnson, & Houlihan, 2009). Therefore, understanding the biological systems taking apart intelligence can be necessary for psychological science (Deary et al., 2009). The neural basis of intelligence has been investigated by a range of neuroimaging studies, including magnetic resonance imaging (MRI) (McDaniel, 2005), electroencephalogram (EEG) (Thatcher, North & Biver, 2005), positron emission tomography (PET) (Tang et al., 2014), task-based functional magnetic resonance imaging (fMRI) (Jung & Haier, 2007) and resting-state fMRI (Li & Tian, 2014). Most structural studies associate intelligence quotient (IQ) with frontal and parietal lobes (Anderson et al., 2004; Basten, Hilger & Fiebach, 2015; Vakhtin, Ryman, Flores, & Jung, 2014; Jung & Haier, 2007). For instance, a relationship between grey matter (GM) volume in the posterior cingulate cortex (PCC) and intelligence has been reported (Deary, Penke, & Johnson, 2010). Moreover, the involvement of subcortical regions, such as the right striatum (Burgaleta et al., 2014), basal ganglia (McNab & Klingberg, 2008), hippocampus (Burgess, Maguire, & O’Keefe, 2002), and caudate (G: has been observed. In addition, the relationship between (GM) volume and cognitive deficits have been investigated in lesion studies (Karussis, Leker, & Abramsky, 2000), (Pinter, Eliez, Schmitt, Capone, & Reiss, 2001). Despite these findings, studies addressing the relationship between brain structures with intelligence in the attention deficit hyperactivity disorder (ADHD) population are rare (Vilgis, Sun, Chen, Silk, & Vance, 2016; McAlonan et al., 2007).
In this study, we hypothesized that the structural basis of intelligence should not be different in normal and ADHD individuals. Therefore, 56 ADHD individuals, including 30 individuals from the combined and 26 individuals from the inattentive subcategory, and 30 healthy individuals in the control group were scanned by MRI. Then, GM densities of anatomically separated brain regions were estimated from the brain images. Subsequently, Pearson’s correlation analysis was employed to detect the association between the GM densities of the brain regions and IQ. The whole pattern of association is also estimated. Then, analysis was performed at the group level and the correlation of IQ and GM densities in ADHD groups were compared with each other as well as the healthy control group. Furthermore, the differential pattern of GM density was observed in three groups. The main aim of the study was to find out whether the relationship between GM density and IQ in ADHD is similar to healthy individuals or is aligned with the disorder.
2. Materials and Methods
Study participants
A total of 56 ADHD subjects, including 30 combined subjects and 26 inattentive subjects, as well as 30 healthy subjects in the control group, with similar age and IQ range, underwent a session of MRI scanning. Table 1 presents the demographic information of each group. According to age, IQ, and handedness, no significant differences were observed among the groups (P<0.05).
.jpg)
Children in the ADHD group had to meet diagnostic criteria defined by the diagnostic and statistical manual, fourth edition, text revision (DSM-IV-TR) for ADHD as well as determine parent and child’s answers to the kiddie schedule for affective disorders and schizophrenia present and lifetime version (KSADS-PL) confirmed by a psychiatrist to be involved in this study. The exclusion criteria included comorbid mood or anxiety disorder, autistic or Asperger’s disorder, medical illness that was unstable or could cause psychiatric symptoms, or substance abuse within 2 months of participation.
Assessment of cognitive performance: intelligence quotient (IQ) scores
All participants completed the Wechsler abbreviated scale of intelligence (WASI) presented in Table 1 . The Wechsler abbreviated scale of intelligence (WASI) is general intelligence, or IQ test designed to measure overall cognitive abilities or a specific cognitive capability in individuals in the age range of 6-89 years. (American Psychological Association (APA PsycNet))
The experimental validation of our proposed method was performed on a dataset gathered from 4 data centers including Kennedy Krieger Institute (KKI), Oregon Health and Science University (OHSU), Peking University (PEK), and New York University (NYU). These data are related to ADHD-200 global completion and are publically.
Magnetic Resonance Imaging (MRI) data acquisition
All participants underwent a T1-weighted high-resolution MRI scanning using a siemens3T scanner system. The imaging data were collected using the following protocols, Kennedy Krieger Institute (KKI) (relaxation time [T1]: Contrast enhancement, echo time [TE]: Shortest, repetition time [TR]: Shortest, flip angle: 8°, field of view [FOV]: 256 mm, slice thickness: 1 mm); Oregon Health and Science University (OHSU) (T1: 900 ms, TE: 3.58 ms, TR: 2300 ms, flip angle: 10°, FOV: 256 mm, slice thickness: 1.1 mm); Peking University (PEK) (T1: 1100 ms, TE: 3.45 ms, TR:2530 ms, flip angle: 7°, FOV: 256mm, slice thickness: 1mm); New York University (NYU) (T1: 1100 ms, TE: 3.25 ms, TR: 2530 ms, flip angle: 7°, FOV: 256 mm, slice thickness: 1.33 mm).
MRI data processing
A standard processing pipeline was performed on the MRI data using the functional MRI of the brain (FMRIB) Software Library (FSL) and Analysis Of Functional Neuroimaging (AFNI). First, the brain MRI images were deobliqued, then re-oriented and the skull strip was removed. Then, images were registered to the MNI space. After that, MRI images were segmented into different tissue types and were parcellated into 392 regions of interest (ROI) using MNI normalized Craddock atlas (Craddock, James, Holtzheimer, Hu, & Mayberg, 2012) or 116 regions using Automated Anatomical Labeling (AAL) (Tzourio-Mazoyer et al., 2002). Results of the Craddock method are presented in the main text body and results of the AAL parcellation are provided in the supplementary materials.
Statistical analysis
Pearsons’ correlation was calculated between IQ and GM densities of separate brain regions and a significant relationship was recognized by setting a threshold of P<0.05. Then statistical analysis was performed using the statistical analysis toolbox of MATLAB 2015. Later, the group comparison of correlation values was performed using the cocor package (Diedenhofen & Musch, 2015). Then, the brain net viewer (Xia, Wang, & He, 2013) was used to visualize the significant results. The whole association pattern is also acquired using SPSS software. We also observed a differential pattern of GM density in groups as a second approach (Figure 1)
.jpg)
It should be mentioned that suggested regions of interest (ROIs) in the Craddock atlas were too fragmented (392 regions), so for the ease of presentation, the significant results were re-indexed to the frontal, occipital, temporal, parietal, cerebellum, and subcortical structures.
3. Results
Significant structural correlates of intelligence quotient (IQ)
The relationship between IQ scores and GM densities was found in several brain regions. Tables 2, 3, 4 and Figures 2, 3, 4 show the detailed information of significant results (P<0.05). Significant positive relationships between GM density and intelligence are shown in red and negative relationships are shown in blue.
In ADHD combined group, a positive correlation was observed between IQ scores and GM density in the frontal region (inferior operculum and orbitofrontal on the right side), temporal region (superior part in the left hemisphere, inferior part in the right hemisphere, and middle part bilaterally), occipital region (inferior part bilaterally), cerebellum (CRB 4-5 in the left hemisphere and CRB-Crus1, in the right side and CRB-Crus2 bilaterally), angular gyrus in the right side, fusiform in the right side, and the precentral region on the right side. In addition, negative correlations were observed in the left inferior temporal region and the left fusiform region (Figure 2, Table 2).
.jpg)
.jpg)
In ADHD inattentive group, no significant negative correlation was observed between IQ and GM density of the brain regions. While a positive correlation was identified in bilateral fusiform areas, caudate and cerebellum crus1, and the inferior parietal region in the right hemisphere (Figure 3, Table 3)
.jpg)
.jpg)
In the healthy control group, a positive and significant correlation was observed bilaterally in the precuneus, and fusiform. In the left hemisphere, a positive correlation was seen in temporal regions (inferior and superior temporal pole), calcarine, superior occipital, and middle frontal regions. In the right hemisphere, a positive correlation was observed at cerebellum 6, frontal inferior operculum, and cingulate cortex. Moreover, a negative correlation was identified in the left superior medial frontal cortex (Figure 4, Table 4).
.jpg)
.jpg)
Comparing the cluster size of each section showed that the most significant relationship for the ADHD combined group is located in the temporal, cerebellum, and frontal regions. In the ADHD inattentive group, the most significant relationship was observed in the temporal, subcortical region, and cerebellum. And in the healthy control group, the most significant relationship was observed in the temporal, frontal, and parietal regions (Table 5).
.jpg)
The results of the relationship between GM density and intelligence via AAL parcellations are presented in the supplementary materials (Figures S1, S2, S3, Table S1).




Group comparison
Figure 5, 6, 7 and Table 6 present significant results of comparing correlations observed in each group (significant P-values have been bolded in Table 6).
.jpg)
.jpg)
.jpg)
.jpg)
Significantly increased relationships between GM density and intelligence are shown in red and decreased relationships are shown in blue.
Comparing ADHD combined versus ADHD inattentive showed significantly increased associated GM density with intelligence at superior temporal and cerebellum-6 in the left hemisphere and angular region in the right hemisphere. Decreased associated grey matter density with intelligence was observed at the middle cingulate on the right side (Figure 5).
ADHD combined compared to the healthy control had an increased relationship between GM density and intelligence at middle temporal and orbitofrontal in the right hemisphere as well as rectus and superior parietal on the left side. Decreased relationship in GM density was seen at the inferior occipital, medial orbitofrontal, and superior frontal in the right hemisphere (Figure 6).
ADHD inattentive compared to the healthy control group showed increased associated GM density with intelligence in the middle and anterior cingulate and superior motor area in the right hemisphere as well as the precuneus, superior parietal, and rectus on the left side. Decreased relationship of GM density with intelligence was observed in the inferior occipital, putamen, caudate, medial orbitofrontal, and precentral region in the right hemisphere as well as middle occipital and cerebellum6 on the left side (Figure 7).
Compared to healthy control, ADHD groups (combined and inattentive) had increased associated GM density with intelligence at superior parietal and rectus in the left hemisphere, while decreased associated GM density with intelligence was seen at inferior occipital and medial frontal in the right side. The results of comparing the relationship between GM density and intelligence via AAL software are presented in supplementary materials (Figures S4, S5, S6, Table S2).




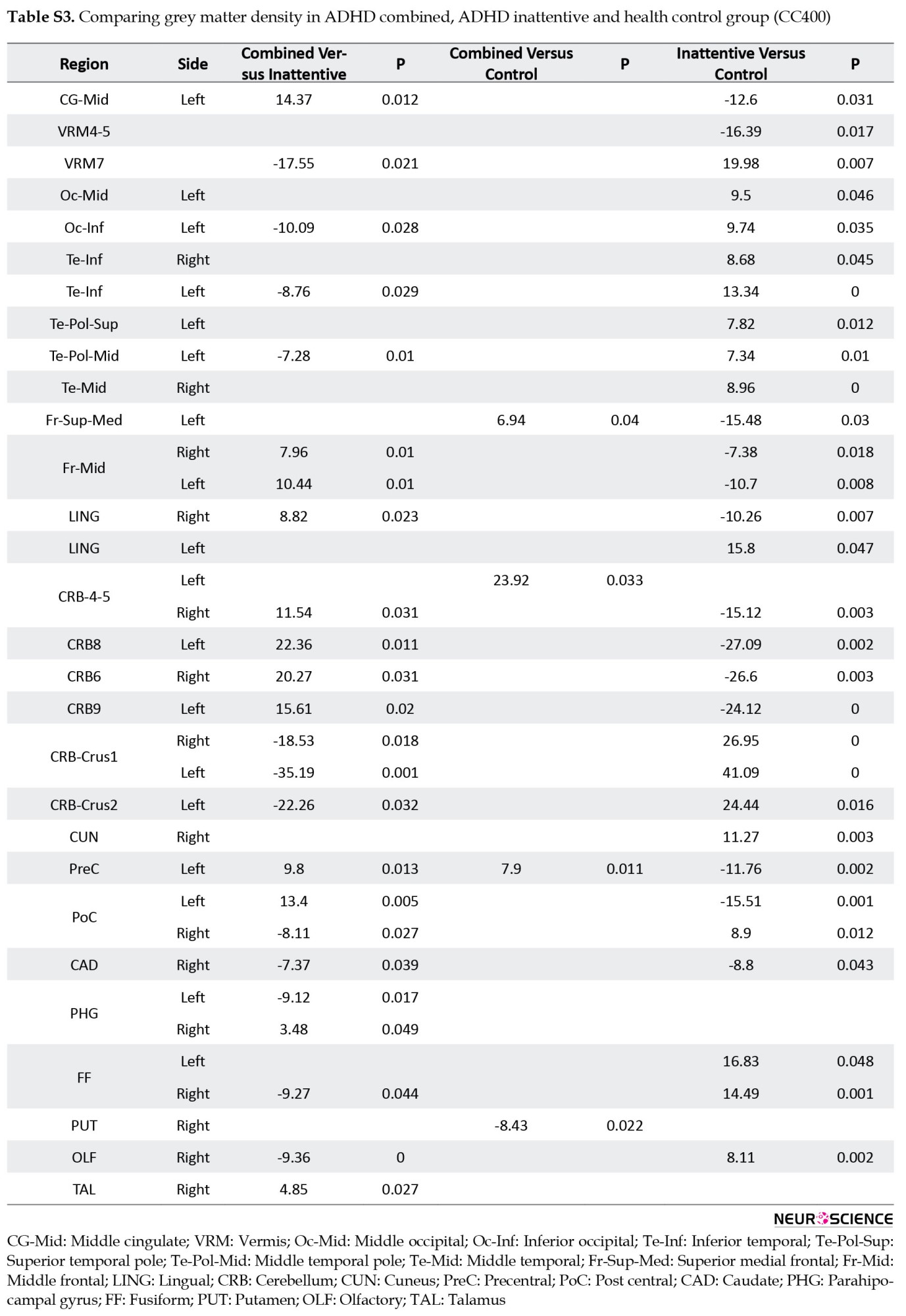
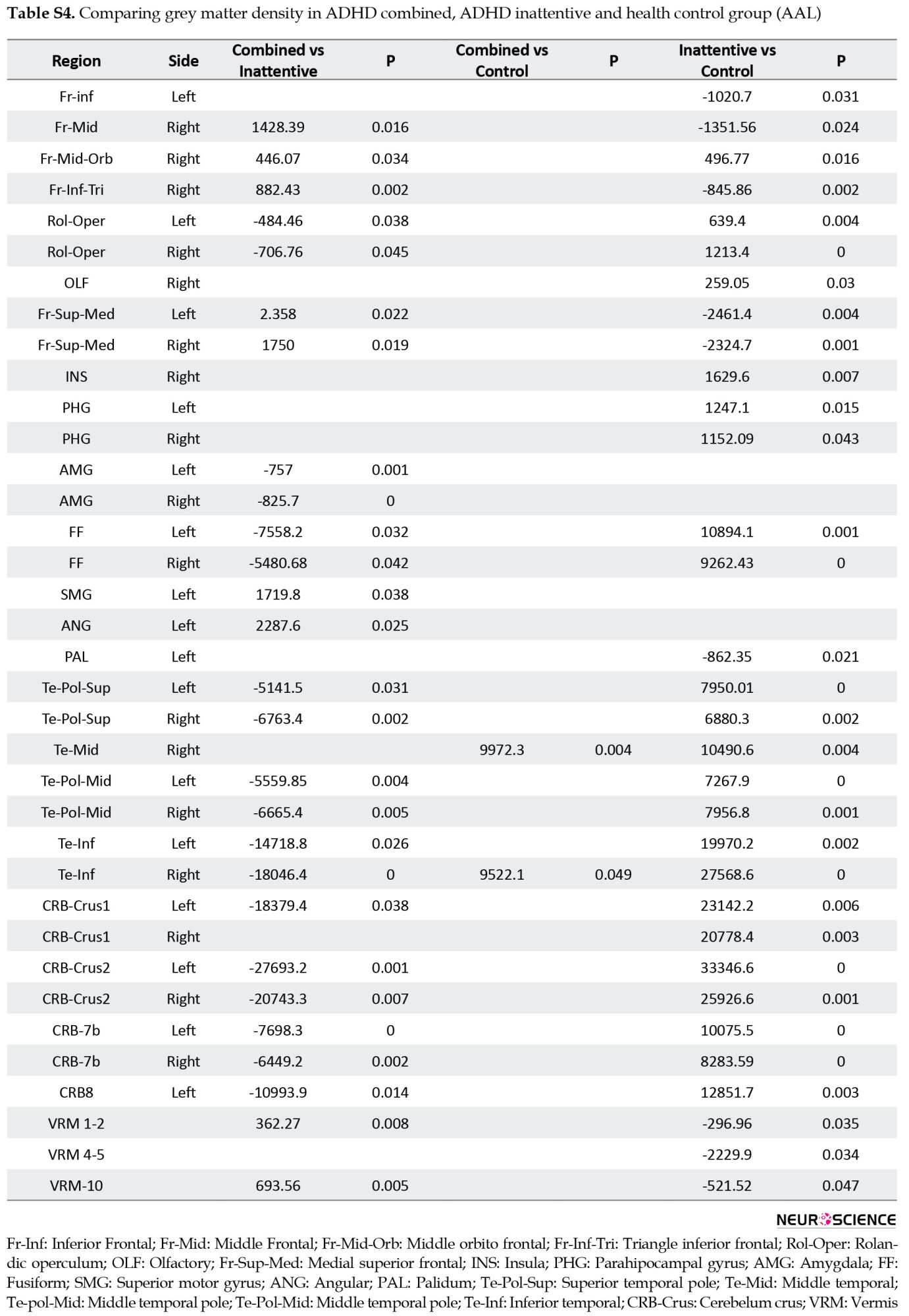
Multiple comparisons of whole brain association patterns also showed differences between groups. Although ADHD combined was significantly different from the other groups, this was not the case for ADHD inattentive group (Table 7), (Figure S14). We also compare raw data of grey matter density to show structural differences in terms of grey matter density whithin groups. Results from CC400 are shown in Table S3, Figures S8, S9, S10 and those of AAL116 are presented in Table S4, Figures S11, S12, S13).





.jpg)




4. Discussion
ADHD is a heritable phenotype that can be influenced by genetic factors. Therefore, genetics can influence brain morphometric properties in ADHD as reported in previous studies (Bellgrove et al., 2005; Swanson et al., 2007). Intelligence is also a heritable phenotype and overlapping effects of genetics on the intelligence score and ADHD also can be investigated. If IQ score is only related to brain morphometry properties, there should not be any significant differences between IQ-matched ADHD and control individuals. Otherwise, the relationship between IQ score and brain anatomy follows another mechanism that we investigate in this study. In this study, the regional GM density and full-scale IQ were compared between two groups of ADHD subtypes, including a combined and inattentive and a healthy control group. Two different parcellation strategies, including functionally separated regions (CC400) and anatomically separated regions (AAL116), were applied to detach the brain to 392 and 116 regions, respectively. After statistical analysis, it was observed that the relationship of IQ with regional GM density is mainly positive and located in the right hemisphere. The right hemisphere plays a vital role in cognition (Robertson, 2014), language (Gainotti, 2013), arithmetic (Knops & Willmes, 2014), and visuospatial attention (Longo, Trippier, Vagnoni, & Lourenco, 2015). These findings are not impressed by the parcellation technique and the results of CC400 were almost the same as AAL116.
The changes in GM density may alter behavior and cognitive functions discussed in the following. Our findings of the healthy control group showed a significant positive relationship between GM density and IQ score in the inferior and middle frontal, precuneus, fusiform, inferior temporal, calcarine, superior occipital, and middle cingulum, and cerebelum6. The inferior frontal is involved in selecting responses in uncertain situations (Frangou, Chitins, & Williams, 2004) and reasoning ability (Goel, Gold, Kapur, & Houle, 1997), and the relationship of frontal and parietal with intelligence has been shown in previous studies (Jung & Haier, 2007). Another finding was the inferior part of the temporal lobe which is involved in the analysis of visual form, motion, and representation of individuals and is directly related to the intelligence score (Frangou et al., 2004). Our results about fusiform may also reflect its role in language proficiency (Tan et al., 2011), recognition, and elaboration on the visual inputs (Colom et al., 2009). Furthermore, the significant relationship between cingulum GM density and IQ, observed in this study, in the healthy control group may reflect its role in spatial learning and memory (Aggleton, Neave, Nagle, & Sahgal, 1995). Last but not least, the relationship of cerebellum GM density with intelligence has also been reported in previous studies (Stoodley & Schmahmann, 2009). These findings confirm the results compared to previous studies.
On the other hand, ADHD showed a significantly different mechanism and pattern of association between GM density and intelligence, compared to the healthy control group. Comparing the whole association pattern, also suggest a discrepancy between the groups. The difference between ADHD combined and control group was more significant than the difference between ADHD inattentive and control, which seems rational because of more severe deficit symptoms in ADHD combined.
The IQ score represents a score for verbal and visual abilities, fluid reasoning, working memory, and processing speed (Wechsler, 1949). In previous studies, the strengths and weaknesses of ADHD individuals in these cognitive functions have been reported (Frazier, Demaree, & Youngstrom, 2004). Mayes’s group has tried to suggest a neurobiological basis for ADHD by emphasizing ADHD strengths in verbal, and visual reasoning and their weakness in attention, processing speed, and graph motor skill (Mayes & Calhoun, 2006). Given that the ADHD and healthy control groups in our study are matched for IQ, observed differences in associated GM density with IQ between ADHD and healthy control groups may suggest a compensatory mechanism in ADHD to maintain adequate performance. This mechanism can be discussed in our research findings. In ADHD groups, combined and inattentive, a decreased association between GM density and intelligence score was observed in the right inferior occipital and right medial frontal regions. The inferior occipital region is involved in visual imaging (Goldenberg et al., 1989) and the medial frontal is engaged in the switching tasks (Rushworth, Hadland, Paus, & Sipila, 2002). In contrast, an increased association was observed between the left superior parietal and the left rectus. The superior parietal region is involved in the manipulation of information in working memory (Koenigs, Barbey, Postle, & Grafman, 2009) and the rectus is engaged in the paradigm of reward learning (Kringelbach, 2005). Therefore, the lessened association between GM density and intelligence in the right hemisphere (inferior occipital and medial frontal) could be compensated by the increased association in the left hemisphere (superior parietal and rectus). This mechanism helps the ADHD subject to maintain an adequate level of cognitive performance.
Comparing row data of GM density in groups also reinforced the idea of a compensatory mechanism. In this regard, GM differences between ADHD inattentive and control group in our study was greater than between ADHD combined and control. This alteration in GM density may help ADHD inattentive individuals compensate for their brain deficiency and perform compared to ADHD combined.
Previous studies have shown that the cortical compensatory mechanisms can cope with the deficit in several cognitive processes in ADHD (Ma et al., 2012). Hence, a different association between regional GM density with the IQ score in the ADHD and control group may indicate that brain structure is not the only determiner of intelligence; rather, intelligence may also be underpinned by neural dynamics of the brain.
Limitation
The results of this study had several limitations; for example, the correlation was done for full-scale IQ and wechsler intelligence scale for children (WISC) subtests were not analyzed. Moreover, this study only focused on male participants in the age range of 8 to 13 years, which can be extended in future works. Therefore, a developmental study on both genders could provide additional insights into the mechanism of structural changes in the brain involved with intelligence.
5. Conclusion
To the best of our knowledge, this is the first study to investigate brain structural correlates of IQ in ADHD individuals. The study provides evidence that IQ may be closely related to GM density in specific brain regions but the pattern can be influenced by a disorder. The significant changes observed in the association scores of the ADHD individuals compared to the healthy individuals in the control group suggest a compensatory mechanism for having a suitable cognitive performance (IQ). Our findings indicate that IQ score may be affected by neural dynamics; therefore, the structural covariates can be a better alternative for the GM density. We hope that these findings can provide additional information for a better understanding of the relationship between brain morphometry and intelligence.
Ethical Considerations
Compliance with ethical guidelines
All ethical principals are considered in this article. The participants were informed of the purpose of the research and its implementation stages. They were also assured about the confidentiality of their information and were free to leave the study whenever they wished and if desired, the research results would be available to them. A written cosent has been obtain from the subjects. Priciples of the Helski convention was also observed.
Funding
This research did not receive any grant from funding agencies in the public commercial, or non-profit sectors.
Authors' contributions
All authors equally contributed to preparing this article.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors thank the ADHD-200 consortium for generously sharing data.
References
Aggleton, J. P., Neave, N., Nagle, S., & Sahgal, A. (1995). A comparison of the effects of medial prefrontal, cingulate cortex, and cingulum bundle lesions on tests of spatial memory: Evidence of a double dissociation between frontal and cingulum bundle contributions. Journal of Neuroscience, 15(11), 7270-7281. [DOI:10.1523/JNEUROSCI.15-11-07270.1995] [PMID] [PMCID]
Anderson, J. R., Bothell, D., Byrne, M. D., Douglass, S., Lebiere, C., & Qin, Y. (2004). An integrated theory of the mind. Psychological Review, 111(4), 1036. [DOI:10.1037/0033-295X.111.4.1036] [PMID]
Basten, U., Hilger, K., & Fiebach, C. J. (2015). Where smart brains are different: A quantitative meta-analysis of functional and structural brain imaging studies on intelligence. Intelligence, 51, 10-27. [DOI:10.1016/j.intell.2015.04.009]
Bellgrove, M. A., Hawi, Z., Lowe, N., Kirley, A., Robertson, I. H., & Gill, M. (2005). DRD4 gene variants and sustained attention in Attention Deficit Hyperactivity Disorder (ADHD): Effects of associated alleles at the VNTR and− 521 SNP. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics, 136(1), 81-86. [DOI:10.1002/ajmg.b.30193] [PMID]
Burgaleta, M., MacDonald, P. A., Martínez, K., Roman, F. J., Álvarez-Linera, J., González, A. R., et al. (2014). Subcortical regional morphology correlates with fluid and spatial intelligence. Human Brain Mapping, 35(5), 1957-1968. [DOI:10.1002/hbm.22305] [PMID] [PMCID]
Burgess, N., Maguire, E. A., & O’Keefe, J. (2002). The human hippocampus and spatial and episodic memory. Neuron, 35(4), 625-641. [DOI:10.1016/S0896-6273(02)00830-9]
Colom, R., Haier, R. J., Head, K., Álvarez-Linera, J., Quiroga, M. Á., Shih, P. C., & Jung, R. E. (2009). Gray matter correlates of fluid, crystallized, and spatial intelligence: Testing the P-FIT model. Intelligence, 37(2), 124-135. [DOI:10.1016/j.intell.2008.07.007]
Craddock, R. C., James, G. A., Holtzheimer, P. E., Hu, X. P., & Mayberg, H. S. (2012). A whole brain fMRI atlas generated via spatially constrained spectral clustering. Human Brain Mapping, 33(8), 1914-1928. [DOI:10.1002/hbm.21333] [PMID] [PMCID]
Deary, I. J., Johnson, W., & Houlihan, L. M. (2009). Genetic foundations of human intelligence. Human Genetics, 126(1), 215-232. [DOI:10.1007/s00439-009-0655-4] [PMID]
Deary, I. J., Penke, L., & Johnson, W. (2010). The neuroscience of human intelligence differences: Nature reviews. Neuroscience, 11(3), 201. [DOI:10.1038/nrn2793] [PMID]
Diedenhofen, B., & Musch, J. (2015). cocor: A comprehensive solution for the statistical comparison of correlations. PloS one, 10(4), e0121945. [DOI:10.1371/journal.pone.0121945] [PMID] [PMCID]
Frangou, S., Chitins, X., & Williams, S. C. (2004). Mapping IQ and gray matter density in healthy young people. Neuroimage, 23(3), 800-805. [DOI:10.1016/j.neuroimage.2004.05.027] [PMID]
Frazier, T. W., Demaree, H. A., & Youngstrom, E. A. (2004). Meta-analysis of intellectual and neuropsychological test performance in Attention-Deficit/Hyperactivity Disorder. Neuropsychology, 18(3), 543-55. [DOI:10.1037/0894-4105.18.3.543] [PMID]
Gainotti, G. (2013). The contribution of language to the right-hemisphere conceptual representations: A selective survey. Journal of clinical and experimental neuropsychology, 35(6), 563-572. [DOI:10.1080/13803395.2013.798399] [PMID]
Goel, V., Gold, B., Kapur, S., & Houle, S. (1997). The seats of reason? An imaging study of deductive and inductive reasoning. NeuroReport, 8(5), 1305-1310. [DOI:10.1097/00001756-199703240-00049] [PMID]
Goldenberg, G., Podreka, I., Steiner, M., Willmes, K., Suess, E., & Deecke, L. (1989). Regional cerebral blood flow patterns in visual imagery. Neuropsychologia, 27(5), 641-664. [DOI:10.1016/0028-3932(89)90110-3]
Grazioplene, R. G.,g Ryman, S., Gray, J. R., Rustichini, A., Jung, R. E., & DeYoung, C. G. (2015). Subcortical intelligence: Caudate volume predicts IQ in healthy adults. Human Brain Mapping, 36(4), 1407-1416. [DOI:10.1002/hbm.22710] [PMID] [PMCID]
Jung, R. E., & Haier, R. J. (2007). The Parieto-Frontal Integration Theory (P-FIT) of intelligence: Converging neuroimaging evidence. Behavioral and Brain Sciences, 30(2), 135-154. [DOI:10.1017/S0140525X07001185] [PMID]
Karussis, D., Leker, R., & Abramsky, O. (2000). Cognitive dysfunction following thalamic stroke: A study of 16 cases and review of the literature. Journal of the Neurological Sciences, 172(1), 25-29. [DOI:10.1016/S0022-510X(99)00267-1]
Knops, A., & Willmes, K. (2014). Numerical ordering and symbolic arithmetic share frontal and parietal circuits in the right hemisphere. Neuroimage, 84, 786-795. [DOI:10.1016/j.neuroimage.2013.09.037] [PMID]
Koenigs, M., Barbey, A. K., Postle, B. R., & Grafman, J. (2009). Superior parietal cortex is critical for the manipulation of information in working memory. Journal of Neuroscience, 29(47), 14980-14986. [DOI:10.1523/JNEUROSCI.3706-09.2009] [PMID] [PMCID]
Kringelbach, M. L. (2005). The human orbitofrontal cortex: linking reward to hedonic experience. Nature Reviews Neuroscience, 6(9), 691-702. [DOI:10.1038/nrn1747] [PMID]
Li, C., & Tian, L. (2014). Association between resting-state coactivation in the parieto-frontal network and intelligence during late childhood and adolescence. American Journal of Neuroradiology, 35(6), 1150-1156. [DOI:10.3174/ajnr.A3850] [PMID] [PMCID]
Longo, M. R., Trippier, S., Vagnoni, E., & Lourenco, S. F. (2015). Right hemisphere control of visuospatial attention in near space. Neuropsychologia, 70, 350-357. [DOI:10.1016/j.neuropsychologia.2014.10.035] [PMID]
Ma, J., Lei, D., Jin, X., Du, X., Jiang, F., Li, F., et al. (2012). Compensatory brain activation in children with Attention Deficit/Hyperactivity Disorder during a simplified Go/No-go task. Journal of Neural Transmission, 119(5), 613-619. [DOI:10.1007/s00702-011-0744-0] [PMID]
Mayes, S. D., & Calhoun, S. L. (2006). WISC-IV and WISC-III profiles in children with ADHD. Journal of Attention Disorders, 9(3), 486-493. [DOI:10.1177/1087054705283616] [PMID]
McAlonan, G. M., Cheung, V., Cheung, C., Chua, S. E., Murphy, D. G., Suckling, J., et al. (2007). Mapping brain structure in attention deficit-hyperactivity disorder: A voxel-based MRI study of regional grey and white matter volume. Psychiatry Research: Neuroimaging, 154(2), 171-180. [DOI:10.1016/j.pscychresns.2006.09.006] [PMID]
McDaniel, M. A. (2005). Big-brained people are smarter: A meta-analysis of the relationship between in vivo brain volume and intelligence. Intelligence, 33(4), 337-346. [DOI:10.1016/j.intell.2004.11.005]
McNab, F., & Klingberg, T. (2008). Prefrontal cortex and basal ganglia control access to working memory. Nature neuroscience, 11(1), 103. [DOI:10.1038/nn2024] [PMID]
Pinter, J. D., Eliez, S., Schmitt, J. E., Capone, G. T., & Reiss, A. L. (2001). Neuroanatomy of Down’s syndrome: A high-resolution MRI study. American Journal of Psychiatry, 158(10), 1659-1665. [DOI:10.1176/appi.ajp.158.10.1659] [PMID]
Robertson, I. H. (2014). A right hemisphere role in cognitive reserve. Neurobiology of Aging, 35(6), 1375-1385. [DOI:10.1016/j.neurobiolaging.2013.11.028] [PMID]
Rushworth, M., Hadland, K., Paus, T., & Sipila, P. (2002). Role of the human medial frontal cortex in task switching: A combined fMRI and TMS study. Journal of Neurophysiology, 87(5), 2577-2592. [DOI:10.1152/jn.2002.87.5.2577] [PMID]
Stoodley, C. J., & Schmahmann, J. D. (2009). Functional topography in the human cerebellum: A meta-analysis of neuroimaging studies. Neuroimage, 44(2), 489-501. [DOI:10.1016/j.neuroimage.2008.08.039] [PMID]
Swanson, J. M., Kinsbourne, M., Nigg, J., Lanphear, B., Stefanatos, G. A., Volkow, N., et al. (2007). Etiologic subtypes of Attention-Deficit/Hyperactivity Disorder: Brain imaging, molecular genetic and environmental factors and the dopamine hypothesis. Neuropsychology Review, 17(1), 39-59. [DOI:10.1007/s11065-007-9019-9] [PMID]
Tan, L. H., Chen, L., Yip, V., Chan, A. H., Yang, J., Gao, J.-H., et al. (2011). Activity levels in the left hemisphere caudate-fusiform circuit predict how well a second language will be learned. Proceedings of the National Academy of Sciences, 108(6), 2540-2544. [DOI:10.1073/pnas.0909623108] [PMID] [PMCID]
Tang, Z., Chen, Z., Zhai, Q., Hao, Y., Zhang, Y., & Zeng, X. (2014). Correlation between interictal cerebral glucose hypometabolism and IQ in children with epilepsy. Epilepsy & Behavior, 31, 15-18. [DOI:10.1016/j.yebeh.2013.10.028] [PMID]
Thatcher, R. W., North, D., & Biver, C. (2005). EEG and intelligence: Relations between EEG coherence, EEG phase delay and power. Clinical Neurophysiology, 116(9), 2129-2141. [DOI:10.1016/j.clinph.2005.04.026] [PMID]
Tzourio-Mazoyer, N., Landeau, B., Papathanassiou, D., Crivello, F., Etard, O., Delcroix, N., et al. (2002). Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage, 15(1), 273-289. [DOI:10.1006/nimg.2001.0978] [PMID]
Vakhtin, A. A., Ryman, S. G., Flores, R. A., & Jung, R. E. (2014). Functional brain networks contributing to the parieto-frontal integration theory of intelligence. Neuroimage, 103, 349-354. [DOI:10.1016/j.neuroimage.2014.09.055] [PMID]
Vilgis, V., Sun, L., Chen, J., Silk, T. J., & Vance, A. (2016). Global and local grey matter reductions in boys with ADHD combined type and ADHD inattentive type. Psychiatry Research: Neuroimaging, 254, 119-126. [DOI:10.1016/j.pscychresns.2016.06.008] [PMID]
Wechsler, D. (1949). Wechsler intelligence scale for children. [Link]
Xia, M., Wang, J., & He, Y. (2013). BrainNet Viewer: A network visualization tool for human brain connectomics. PloS One, 8(7), e68910. [DOI:10.1371/journal.pone.0068910] [PMID] [PMCID]
Type of Study: Original |
Subject:
Cognitive Neuroscience
Received: 2019/07/20 | Accepted: 2020/06/14 | Published: 2022/07/24
Received: 2019/07/20 | Accepted: 2020/06/14 | Published: 2022/07/24
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |








