BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://bcn.iums.ac.ir/article-1-1304-en.html
2- Division of Neurocognitive Sciences, Psychiatry and Behavioral Sciences Research Center, Mashhad University of Medical Sciences, Mashhad, Iran.
3- Neurogenic Inflammation Research Centre, Mashhad University of Medical Sciences, Mashhad, Iran.
Highlights
● Inactivation of the CnF by CoCl2 did not significantly affect basal cardiovascular responses.
● Hemorrhage significantly lowered blood pressure and increased heart rate.
● Inactivation of the CnF significantly attenuated hypotension and tachycardia induced by hemorrhage.
Plain Language Summary
The Hemorrhage (HEM) is a life-threatening condition produced by a significant loss of blood, leading to hypotension, decreased tissue perfusion, cellular hypoxia and possibly death. Following HEM, several systems, including the Central Nervous System (CNS) are activated to improve this condition. For example, CNS restores hypotension during HEM and increases tissue perfusion. Numerous brain areas, such as rostral ventrolateral medulla, nucleus tractus solitarius, paraventricular nucleus are involved in the regulation of cardiovascular responses during HEM. Also, other areas, like Cuneiform Nucleus (CnF) are involved in cardiovascular regulation; however, their cardiovascular effects during HEM are unclear. In this study, we evaluated the possible cardiovascular effect of CnF in HEM. The groups were as follows: vehicle, cobalt chloride (CoCl2, a reagent that blocks the activity of nucleus): microinjection of CoCl2 (1mM) into the CnF; HEM: blood withdrawal of about 15% of TBV, Hem+saline, and HEM+CoCl2 groups. In all groups, Blood Pressure (BP) and also Heart Rate (HR) were recorded by a PowerLab instrument and the changes were calculated and compared. The results indicated that HEM decreased BP and increased HR. However, these effects (decreased BP and increased HR) improved only after the inactivation of the CnF. Our results confirmed the involvement of CnF in cardiovascular regulation of BP and HR during HEM.
1. Introduction
Hemorrhage (HEM) is a life-threatening incident due to the loss of intravascular volume caused by hypotension with two compensatory and non-compensatory phases (Evans, Ventura, Dampney, & Ludbrook, 2001; Standl, Annecke, Cascorbi, Heller, Sabashnikov, & Teske, 2018). The compensatory phase is performed by the elicit of baroreceptors signaling hypotension to the nucleus of the solitary tract (NTS) followed by the activation of vasomotor neurons in Rostral Ventrolateral Medulla (RVLM), an important area in cardiovascular modulation, that excites the sympathetic neurons and maintains blood pressure in normal condition (Naves et al., 2018; Palkovits, 1999). If blood loss continues, the non-compensatory phase, as the next phase, is started, in which sympathetic drive abruptly reduces and arterial pressure falls. The underlying mechanism responsible for these phases of HEM is not completely understood (Evans et al., 2001) and it has suggested that in addition to NTS (Buller, Smith, & Day, 1999) and RVLM (Dean & Bago, 2002), other nuclei also are involved. For example, it has reported that the Paraventricular Nucleus of the Hypothalamus (PVN) (Fan & McIntosh, 1994) and parabrachial areas (Blair & Mickelsen, 2006) are associated with the cardiovascular regulation during HEM. Also, numerous nuclei, such as Cuneiform Nucleus (CnF) are found in the brain that play important role in cardiovascular regulation; however, their role in HEM condition has not yet defined (Verberne, 1995).
The CnF is a reticular nucleus in the midbrain located in the anterolateral of Periaqueductal Grey (PAG) matter and begins from caudate part of the midbrain and continues to the rostral part of pons (Gioia & Bianchi, 1987). The CnF is precipitated in several functions, including regulation of motor activity, pain and respiratory modulation, integration of sensory-motor movement related to pain, defense reactions, and sleep (Shafei, Nasimi, Alaei, Pourshanazari, & Hosseini, 2012; Xiang, Zhu, Guan, & Ye, 2013). The role of CnF in cardiovascular regulation has also been reported in previous studies. Verberne reported that electrical stimulation of CnF increased sympathetic vasomotor outflow and blood pressure (Verberne, 1995). The role of CnF in cardiovascular regulation has been evaluated (Shafei et al., 2012). For example, our previous study indicated that glutamate increased blood pressure (Nasimi, Shafei, & Alaei, 2012), and the cholinergic system decreased cardiovascular parameters (Shafei, Niazmand, Hosseini, & Daloee, 2013). Despite the role of CnF in cardiovascular regulation, we previously indicated that inactivation of this nucleus by Cobalt Chloride (CoCl2) did not change basic cardiovascular parameters (Shafei et al., 2012). Although CnF is not active in basal conditions, it may affect cardiovascular responses in conditions, such as defense reaction, exercise, pain, and HEM (Verberne, Lam, Owens, & Sartor, 1997). Similarly, i Ahlgren et al. reported that HEM increased Fos-Like Immunoreactivity (FLI), a marker of brain activity, in the CnF (Ahlgren, Porter, & Hayward, 2007). Also, the relationship between CnF and brain regions, such as RVLM (Verberne, 1995), NTS (Buller, Smith, & Day, 1999) and PGA mater (Netzer et al., 2011) that regulate cardiovascular response during HEM has been reported. Accordingly, it is conceivable that the CnF is involved in cardiovascular adjustment during HEM. Therefore, the present study was conducted to evaluate the possible role of CnF in cardiovascular regulation during HEM.
2. Metods
2.1. Animal and recording of blood pressure
In this experiment, 30 male Wistar rats were used. Animals anesthetized with urethane (1.5 g/kg, IP). A heating lamp was used to maintain a relatively constant body temperature of the animals. The left femoral artery also was catheterized with an angiocath filled with heparinized saline (60 u/ml) to record Blood Pressure (BP) and Heart Rate (HR) using a blood pressure transducer and a PowerLab system (ID instrument, Australia) (Shafei & Nasimi, 2011). In all groups, Systolic Blood Pressure (SBP), Mean Atrial Pressure (MAP), and HR were recorded throughout the trial period.
2.2. Surgical procedures
The rats were mounted on a stereotaxic apparatus (Stoelting, USA). The scalp was incised and the skull was leveled between the bregma and lambda, and a small hole drilled above the CnF in the skull. The CnF stereotaxic coordinates were -7.6 to -8.5 mm caudal to the bregma, -1.7 to -2.2 mm lateral to the midline suture, and -5.5 to -6.2 mm ventral to the bregma according to the Paxinos and Watson atlas (Paxinos & Watson 2005). CoCl2 (1mM) (Shafei, Alaei, & Farrokhi, 2011) was unilaterally microinjected into the CnF by a single-barreled micropipette (35-45 μm in diameter). For injection, the micropipette was connected through a PE-10 tube to a manual microinjector (Harvard) and carefully introduced into the CnF and injection was done during 30 s. The volume of the injected drug or saline was 100-150 nl in all groups (Shafei, Nikyar, Hosseini, Niazmand, & Paseban, 2017). The study protocol was approved by the Bioethics Committee of Mashhad University of Medical Sciences (Code: 931769).
2.3. Hemorrhage protocol
In groups subjected to hemorrhagic hypotension, the right femoral artery was cannulated by a polyethylene tube filled with heparinized saline (60 u/ml) to prevent clotting during blood withdrawal. Blood withdrawal was initiated 5 min after a microinjection of CoCl2 into the CnF. Blood withdrawal was performed from the femoral artery intermittently at a rate of 1 ml/100 g body weight for 10 min (Dean & Bago, 2002). In this method, about 15% of the Total Blood Volume (TBV) was withdrawn. The TBV of the rats was calculated previously as follows: 0.06 ml/g×body weight+0.77. This volume could induce an intermediate hemorrhage suitable to evaluate the role of cardiovascular regulation during HEM (Ahlgren et al., 2007).
At the end of each experiment, the animals’ brain was removed and 24 h after fixation with formalin 10%, serial sections (50-micron thickness) were prepared using a microtome, and the location of the injections verified based on the atlas of Paxinos and a light microscope (Shafei et al., 2012).
2.4. Drug
The urethane and CoCl2 were provided by the SigmaCo., USA. All drugs were dissolved in saline.
2.5. Animal groups
Animals were randomly divided into the five following groups (n=6 per group): 1. Vehicle: microinjection of saline into the CnF; 2. Cobalt chloride (CoCl2): microinjection of CoCl2 (1mM) into the CnF; 3. HEM: blood withdrawal of about 15% of TBV; 4. Hem+saline: saline microinjection into the CnF 5 min before HEM; and 5. HEM+CoCl2: CoCl2 microinjection into the CnF 5 min before HEM.
2.6. Data analysis
Changes (∆) in cardiovascular parameters (SBP, MAP, and HR) were calculated and expressed as mean±SEM. The ∆SBP, ∆MAP, and ∆HR parameters in all groups were computed at each 5 min and statistically compared with the saline and HEM groups (repeated measures ANOVA). Also, the peak ∆SBP, ∆MAP, and ∆HR of each group were separately calculated 30 min after HEM (40 min after initiation of HEM) and compared with peak changes of the vehicle and HEM groups (independent sample t-test). A P< 0.05 was considered significant.
3. Results
3.1. Effect of the vehicle and CoCl2 microinjection into the CnF on cardiovascular parameters in normotensive rats
In this study, vehicle (saline) and CoCl2 were separately injected into the CnF. Injection of saline into the CnF did not significantly change SBP, MAP, and HR compared with pre-injection. Inactivation of the CnF by microinjection of CoCl2 also did not significantly affect basal cardiovascular parameters than the saline group (Table 1 and Figure 1).
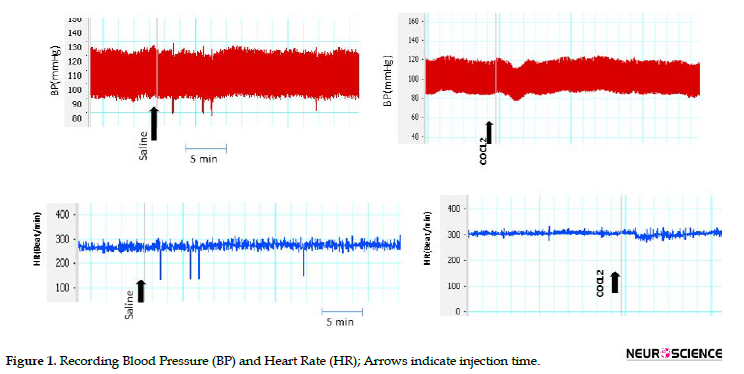
3.2. Effect of hemorrhage on cardiovascular parameters
In HEM, withdrawal of blood was done over 10 min and cardiovascular responses were evaluated 45 min later. Figure 2 indicates changes in blood pressure and HR during HEM.
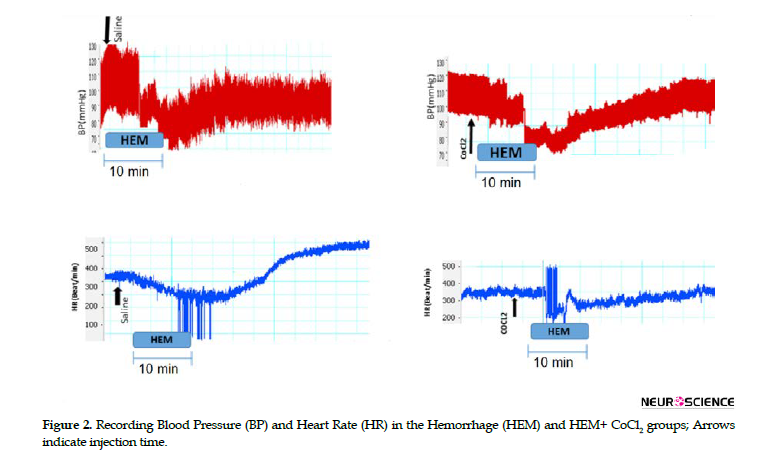
Time-course changes in the responses also were plotted in Figures 3-6. As can be seen, at the end of blood withdrawal (10 min after initiation of hemorrhage), MAP, SBP, and HR decreased and then slowly returned, and after 20 min, SBP and MAP stabilized below the baseline levels; however, HR showed an increase (Figure 2). ∆MAP and ∆SBP after blood withdrawal were significantly lower compared with the vehicle group over time (repeated measures ANOVA; P<0.01, n=6), whereas HR firstly decreased followed by a significant increase (repeated measures ANOVA; P<0.01, n=6).
Also, 30 min after blood withdrawal (40 min after initiation of blood withdrawal), the peak ∆MAP, ∆SBP, and ∆HR were calculated and compared with the vehicle group. Compared with vehicle group, ΔSBP (HEM: -27.2±2.8 mmHg vs. vehicle: -2.3±1.9 mmHg, P<0. 01) and ΔMAP (HEM: -21.6±3.4 mmHg vs. vehicle: -1.4±1.6 mmHg, P<0.01) significantly reduced, whereas ΔHR (HEM: 64.6±6.24 mmHg vs. vehicle: -6.7±3.8; P<0.001; independent sample t-test, Figures 3 and 4) significantly increased.
3.3. Inactivation of the CnF by CoCl2 and its effect on cardiovascular responses in hypotension caused by hemorrhage
In this study, CnF was first inactivated by the microinjection of CoCl2 and 5 min later, HEM performed over 10 min (Figure 3).
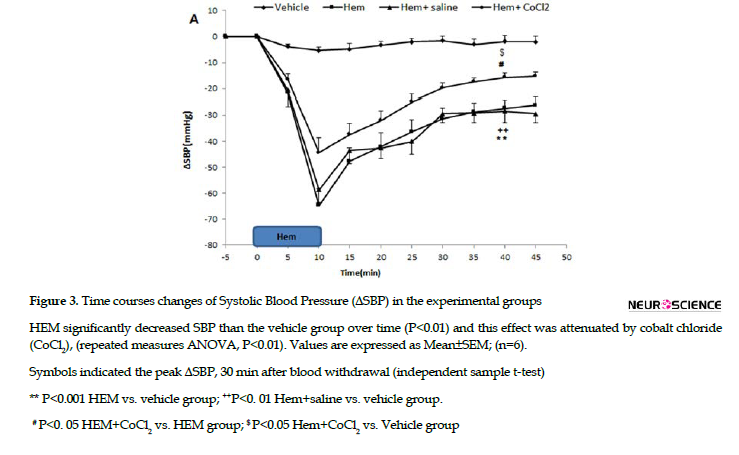
Time-course changes of SBP, MAP, and HR in the HEM, HEM+saline, and HEM+CoCl2 groups are shown in Figure 4.
.png)
As can be seen, in the HEM+saline group, all parameters were not significant compared with the HEM group; however, they were significant than the vehicle group (repeated measures ANOVA, P<0.01, Figure 4). In HEM+CoCl2 group, both ∆MAP and ∆SBP significantly increased, whereas ∆HR significantly decreased than the HEM group (repeated measures ANOVA, P<0. 01, Figure 4).
The peak ∆SBP, ∆MAP, and ΔHR, 30 min after blood withdrawal in the HEM+saline and HEM+CoCl2 groups were also evaluated. Comparing cardiovascular parameters 30 min after blood withdrawal (40th min) indicated that in the HEM+saline group, ∆SBP (-28.4±3.6 mmHg) and ∆MAP (-20.6±2.9 mmHg) were not significant than the HEM group; however, they were lower than that of the vehicle group (-2.3±1.9 mmHg and -1.4±1.6 mmHg, respectively) (P<0.01, Figure 4). ΔHR in the HEM+saline group was significant in comparison with the vehicle group (54.3±5.6 beats/min vs. -6.7±3.8 beats/min, respectively; P<0.001, Figure 4).
.png)
In the HEM+CoCl2 group, 30 min after termination of blood withdrawal, ΔSBP (-15.7±1.8 mmHg) and ΔMAP (12.2±1.4 mmHg) significantly increased than the HEM group (-27.2±2.8 mmHg and -21.6±3.4 mmHg, respectively); (P<0.05, Figure 4). In the HEM+CoCl2 group, ΔHR also significantly decreased compared with the HEM group (28.4±4.8 beats/min vs. 64.6±6.24 beats/min, respectively, P<0.01; Figure 4).
4. Discussion
We aimed at investigating whether the CnF is involved in returning the cardiovascular response to the normal level during hemorrhage. Therefore, the role of the CnF was studied in both normotensive and hemorrhagic hypotensive conditions. Our results indicated that in normotensive condition, inactivation of the CnF did not affect cardiovascular parameters. This effect confirms our previous experiment, in which CnF in the basal condition had no significant effect on the cardiovascular system (Shafei et al., 2012). Although CnF nucleus did not cause a cardiovascular response in basal condition; however, inactivation of this nucleus by CoCl2 attenuated hypotension and tachycardia, induced by HEM. This effect confirms the involvement of the CnF in cardiovascular response during HEM (Shafei et al., 2012). Our results are consistent with those reported by Ahlgren et al. that suggested the involvement of CnF in cardiovascular regulation during HEM. Algren et al. have shown that blood withdrawal of about 30% of TBV could initiate a non-compensatory phase and increase FLI in the CnF nucleus (Ahlgren et al., 2007).
Our experiment aimed to maintain the blood pressure of rats in the compensatory phase, i.e. blood withdrawal of 1 ml /100 g BW in 10 min. Loss of this volume of blood (about 15%) initially decreased both blood pressure and HR, and then HR significantly increased and blood pressure gradually returned and stabilized at about 30-40 mmHg lower than the baseline value. Reversible inactivation of CnF with CoCl2 before HEM could attenuate low blood pressure and tachycardia induced by HEM. This effect showed that the CnF has inhibitory and excitatory effects on blood pressure and HR during HEM, respectively.
The mechanism (s) of this effect of the CnF is unclear; however, several mechanisms have been proposed. The presence of serotonergic, GABAergic, glutamatergic, cholinergic, and nitrergic neurons have been shown in the CnF (Gioia & Bianchi, 1987; Shafei & Nasimi, 2011; Shafei et al., 2013; Shafei et al., 2017; Verberne et al., 1997), of which cholinergic and nitrergic neurons have the inhibitory effect on the cardiovascular system (Shafei et al., 2013; Shafei et al., 2017). We suggest that in HEM, these neurons of the CnF are active and can delay the recovery of blood pressure. However, inactivation of the CnF nucleus by CoCl2 (a synaptic blocker) blocked Ach release and accelerated the recovery of blood pressure. Other neurons present in the CnF and those involved in HEM have shown to be serotonergic neurons.
These neurons play an important role in the regulation of cardiovascular responses in normal and HEM conditions. For example, intracerebroventricular injection of serotonin increased both blood pressure and HR or 5-HT1A receptors of the NTS during HEM increased sympathetic activity. Serotonin neurons in CnF nucleus are associated with several functions CnF is involved, such as pain regulation. Currently, the role of CnF neurons in cardiovascular regulation in HEM is unknown and more studies are needed to indicate which neurotransmitter of the CnF is involved in this function (Gioia & Bianchi, 1987). Also, the relationship between CnF and RVLM, NTS, PVN, PAG, and Kölliker-Fuse nucleus (KF) areas has previously been reported (Korte, Jaarsma, Luiten, & Bohus, 1992).
The sympathoexcitatory effect of the CnF has shown to mostly be mediated by RVLM. However, the direct projection of CnF to RVLM is spare and indirect and mostly mediated by CnF-KF –RVLM pathway. Our previous study also indicated that the cardiovascular effect of the glutamatergic system of CnF is mediated via this pathway. As this pathway is excitatory and CnF has an inhibitory effect in HEM, it can be suggested that this pathway is not important in HEM. Also, CnF is associated with PAG and the cardiovascular effect of CnF is partly mediated by this Pathway. Due to the involvement of the PAG in the regulation of cardiovascular response during HEM, it is conceivable that this effect of the CnF can be mediated by the CnF-PAG pathway.
The CnF- raphe pathway can be another possible involved pathway. The relationship between CnF and raphe nuclei has been shown. Richter et al. indicated that this pathway plays a role in pain modulation (Richter & Behbehani, 1991). Therefore, it is possible that in addition to pain modulation, it could modulate cardiovascular response during HEM.
In HEM, chemo- and baroreceptors are active and can elicit chemo- and baroreflexes via projection to NTS. A projection from CnF to NTS has also been documented. Because NTS is an important area in the integration of chemo- and baroreflexes, it is conceivable that CnF via a relationship with NTS could modulate the activity of chemo- and baroreflexes. Also, Korte et al. indicated the connection of CnF with NTS and the dorsal motor nucleus of the vagus (Korte et al., 1992). Therefore, is can be suggested that the reduction of tachycardia induced by HEM after blockade of CnF with CoCl2 is mediated by vagal system or NTS. In this study, CnF was inactivated unilaterally, which its effect can be different from bilateral inactivation. However, future studies are needed to evaluate the mechanism(s) of the CnF on cardiovascular regulation during HEM.
In conclusion, our results showed that CnF nucleus is involved in the adjustment of cardiovascular responses in HEM. Further studies should be conducted to evaluate the mechanisms and neural circuits of CnF involved in the regulation of cardiovascular response during HEM.
Ethical Considerations
Compliance with ethical guidelines
This study was conducted in accordance with the protocols approved by the Ethical Committee of Mashhad University of Medical Sciences (Code: IR.MUMS.REC.1394.200).
Funding
This study was funded by the Research Council of Mashhad University of Medical Sciences, Mashhad, Iran.
Authors' contributions
Conceptualization: Mohammad Naser Shafei and Mahmoud Hosseini; Methodology: Reza Mohebbati and Majid Khazaei; Investigation and writing-original draft: Reza Mohebbati; Writing-review & editing, supervision, and funding acquisition: Mohammad Naser Shafei; Resources: All author.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors would like to thank the Research Council of Mashhad University of Medical Sciences for funding this study.
References
Ahlgren, J., Porter, K., & Hayward, L. F. (2007). Hemodynamic responses and c-Fos changes associated with hypotensive hemorrhage: Standardizing a protocol for severe hemorrhage in conscious rats. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 292(5), R1862-71. [DOI:10.1152/ajpregu.00325.2006] [PMID]
Blair, M. L., & Mickelsen, D. (2006). Activation of lateral parabrachial nucleus neurons restores blood pressure and sympathetic vasomotor drive after hypotensive hemorrhage. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 291(3), R742-50. [DOI:10.1152/ajpregu.00049.2006] [PMID]
Buller, K. M., Smith, D. W., & Day, T. A. (1999). NTS catecholamine cell recruitment by hemorrhage and hypoxia. NeuroReport, 10(18), 3853-6. [DOI:10.1097/00001756-199912160-00024] [PMID]
Dean, C., & Bago, M. (2002). Renal sympathoinhibition mediated by 5-HT 1A receptors in the RVLM during severe hemorrhage in rats. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 282(1), R122-30. [DOI:10.1152/ajpregu.2002.282.1.R122] [PMID]
Evans, R. G., Ventura, S., Dampney, R. A. l., & Ludbrook, J. (2001). John Ludbrook APPS symposium neural mechanisms in the cardiovascular responses to acute central hypovolaemia. Clinical and Experimental Pharmacology and Physiology, 28(5‐6), 479-87. [DOI:10.1046/j.1440-1681.2001.3473.x] [PMID]
Fan, L., & McIntosh, T. K. (1994). Effect of dynorphin microinjection in the paraventricular nucleus on the hemodynamic response to hemorrhage in the rat. Circulatory Shock, 42(4), 197-203. [PMID]
Gioia, M., & Bianchi, R. (1987). The cytoarchitecture of the nucleus cuneiformis. A Nissl and Golgi study. Journal of Anatomy, 155, 165-76. [PMID] [PMCID]
Korte, S. M., Jaarsma, D., Luiten, P. G. M., & Bohus, B. (1992). Mesencephalic cuneiform nucleus and its ascending and descending projections serve stress-related cardiovascular responses in the rat. Journal of the Autonomic Nervous System, 41(1-2), 157-76. [DOI:10.1016/0165-1838(92)90137-6]
Nasimi, A., Shafei, M. N., & Alaei, H. (2012). Glutamate injection into the cuneiform nucleus in rat, produces correlated single unit activities in the Kolliker-Fuse nucleus and cardiovascular responses. Neuroscience, 223, 439-46. [DOI:10.1016/j.neuroscience.2012.07.041] [PMID]
Naves, L. M., Marques, S. M., Mourão, A. A., Fajemiroye, J. O., Xavier, C. H., & de Castro, C. H., et al. (2018). Involvement of median preoptic nucleus and medullary noradrenergic neurons in cardiovascular and sympathetic responses of hemorrhagic rats. Scientific Reports, 8(1), 11276. [DOI:10.1038/s41598-018-29310-z] [PMID] [PMCID]
Netzer, F., Bernard, J. F., Verberne, A. J., Hamon, M., Camus, F., & Benoliel, J. J., et al. (2011). Brain circuits mediating baroreflex bradycardia inhibition in rats: An anatomical and functional link between the cuneiform nucleus and the periaqueductal grey. The Journal of Physiology, 589(Pt 8), 2079-91. [DOI:10.1113/jphysiol.2010.203737] [PMID] [PMCID]
Palkovits, M. (1999). Interconnections between the neuroendocrine hypothalamus and the central autonomic system: Geoffrey Harris Memorial Lecture, Kitakyushu, Japan, October 1998. Frontiers in Neuroendocrinology, 20(4), 270-95. [DOI:10.1006/frne.1999.0186] [PMID]
Paxinos G., & Watson, C. (2005). The rat brain in stereotaxic coordinates. Burlington, MA: Elsevier Science. https://books.google.com/books?id=LKJqAAAAMAAJ&dq
Richter, R. C., & Behbehani, M. M. (1991). Evidence for glutamic acid as a possible neurotransmitter between the mesencephalic nucleus cuneiformis and the medullary nucleus raphe magnus in the lightly anesthetized rat. Brain Research, 544(2), 279-86. [DOI:10.1016/0006-8993(91)90065-4]
Shafei, M. N., Alaei, H., & Farrokhi, E. (2011). Effect of reversible inactivation of the Kolliker fuse nucleus on basal blood pressure and heart rate in anesthetized rat. Basic and Clinical Neuroscience, 3(1), 4-8. http://bcn.iums.ac.ir/article-1-189-en.html
Shafei, M. N., & Nasimi, A. (2011). Effect of glutamate stimulation of the cuneiform nucleus on cardiovascular regulation in anesthetized rats: Role of the pontine Kolliker-Fuse nucleus. Brain Research, 1385, 135-43. [DOI:10.1016/j.brainres.2011.02.046] [PMID]
Shafei, M. N., Nasimi, A., Alaei, H., Pourshanazari, A. A., & Hosseini, M. (2012). Role of cuneiform nucleus in regulation of sympathetic vasomotor tone in rats. Pathophysiology, 19(3), 151-5. [DOI:10.1016/j.pathophys.2011.11.001] [PMID]
Shafei, M. N., Niazmand, S., Enayatfard, L., Hosseini, M., & Hasanzade Daloee, M. (2013). Pharmacological study of cholinergic system on cardiovascular regulation in the cuneiform nucleus of rat. Neuroscience Letters, 549, 12-7. [DOI:10.1016/j.neulet.2013.05.046] [PMID]
Shafei, M. N., Nikyar, T., Hosseini, M., Niazmand, S., & Paseban, M. (2017). Cardiovascular effects of nitrergic system of the pedunculopontine tegmental nucleus in anesthetized rats. Iranian Journal of Basic Medical Sciences, 20(7), 776-82. [DOI:10.22038/IJBMS.2017.9009] [PMID] [PMCID]
Standl, T., Annecke, T., Cascorbi, I., Heller, A. R., Sabashnikov, A., & Teske, W. (2018). The nomenclature, definition and distinction of types of shock. Deutsches Ärzteblatt International, 115(45), 757-68. [DOI:10.3238/arztebl.2018.0757] [PMID] [PMCID]
Verberne, A. J. (1995). Cuneiform nucleus stimulation produces activation of medullary sympathoexcitatory neurons in rats. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 268(3), R752-8. [DOI:10.1152/ajpregu.1995.268.3.R752] [PMID]
Verberne, A. J., Lam, W., Owens, N. C., & Sartor, D. (1997). Supramedullary modulation of sympathetic vasomotor function. Clinical and Experimental Pharmacology and Physiology, 24(9-10), 748-54. [DOI:10.1111/j.1440-1681.1997.tb02126.x] [PMID]
Xiang, H. B., Zhu, W. Z., Guan, X. H., & Ye, D. W. (2013). The cuneiform nucleus may be involved in the regulation of skeletal muscle tone by motor pathway: A virally mediated trans-synaptic tracing study in surgically sympathectomized mice. Brain, 136(Pt 10), e251. [DOI:10.1093/brain/awt123] [PMID]
Received: 2018/08/8 | Accepted: 2019/11/10 | Published: 2020/05/1
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |









.JPG)
