Volume 12, Issue 3 (May & June 2021)
BCN 2021, 12(3): 383-394 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Jameie S B, Pirasteh A, Naseri A, Jameie M S, Farhadi M, Fahanik Babaee J et al . β-Amyloid Formation, Memory, and Learning Decline Following Long-term Ovariectomy and Its Inhibition by Systemic Administration of Apigenin and β-Estradiol. BCN 2021; 12 (3) :383-394
URL: http://bcn.iums.ac.ir/article-1-1820-en.html
URL: http://bcn.iums.ac.ir/article-1-1820-en.html
Seyed Behnamedin Jameie *1 

 , Abbas Pirasteh2
, Abbas Pirasteh2 
 , Ali Naseri2
, Ali Naseri2 
 , Melika Sadat Jameie3
, Melika Sadat Jameie3 

 , Mona Farhadi4
, Mona Farhadi4 

 , Javad Fahanik Babaee5
, Javad Fahanik Babaee5 

 , Leila Elyasi6
, Leila Elyasi6 




 , Abbas Pirasteh2
, Abbas Pirasteh2 
 , Ali Naseri2
, Ali Naseri2 
 , Melika Sadat Jameie3
, Melika Sadat Jameie3 

 , Mona Farhadi4
, Mona Farhadi4 

 , Javad Fahanik Babaee5
, Javad Fahanik Babaee5 

 , Leila Elyasi6
, Leila Elyasi6 


1- Neuroscience Research Center, Iran University of Medical Sciences, Tehran, Iran.
2- Department of Psychology, Faculty of Humanities, Firoozabad Branch, Islamic Azad University, Fars, Iran.
3- Department of Anatomy, Faculty of Medicine, Shahid Behshti University of Medical Sciences, Tehran, Iran.
4- Department of Microbiology, Karaj Branch, Islamic Azad University, Karaj, Iran.
5- Department of Physiology, School of Medicine, Iran University of Medical Sciences, Tehran, Iran.
6- Department of Anatomy, Neuroscience Research Center, School of Medicine, Golestan University of Medical Sciences, Gorgan, Iran.
2- Department of Psychology, Faculty of Humanities, Firoozabad Branch, Islamic Azad University, Fars, Iran.
3- Department of Anatomy, Faculty of Medicine, Shahid Behshti University of Medical Sciences, Tehran, Iran.
4- Department of Microbiology, Karaj Branch, Islamic Azad University, Karaj, Iran.
5- Department of Physiology, School of Medicine, Iran University of Medical Sciences, Tehran, Iran.
6- Department of Anatomy, Neuroscience Research Center, School of Medicine, Golestan University of Medical Sciences, Gorgan, Iran.
Full-Text [PDF 11099 kb]
| Abstract (HTML)
Full-Text:
1. Introduction
Neurodegenerative Diseases (ND) are age-dependent. Other factors, including genetics and gender, also affect the incidence, severity, response to treatment, and mortality. Cellular and molecular changes have been reported previously. The formation of β-amyloid plaques, changes of dendritic and synaptic structures, and neuronal death has been reported in AD (Cheng & Li, 2014; Lee, Park, Koh & Choi, 2017). AD starts around 60- 65 years old with clinical symptoms, such as memory and speech impairment, psychological and psychiatric changes. Studies have emphasized the role of β-amyloid plaques in the cortex and hippocampus, leading to AD symptoms (Hatashita & Wakebe, 2019; Boumenir et al., 2019; Wang et al., 2011; Prakash & Kumar, 2014). Memory impairment and cognitive dysfunction in AD are associated with cellular and molecular changes in the hippocampus (Krishnan, Slavin, Tran, Murali Doraiswamy & Petrella, 2006; Giffard, Desgranges & Eustache, 2005). Other factors, including oxidative stress, vascular, metabolic, hormonal, and neurotransmitter changes, influence the formation of β-amyloid plaques and Neurofibrillary Tangles (NFTs). The neuroprotective effect of ovarian sex steroids has been mentioned in several studies (Jameei, Nouyan-Ashraf & Behzadi, 2004; Jameie et al., 2016). These hormones easily pass through the blood-brain barrier and affect nervous tissue (Sandoval & Witt, 2011). The level of circulating sex hormones affects physiopathology, symptoms, severity, and treatment of ND (Vest & Pike, 2013; Pike, Carroll, Rosario & Barron, 2009). Epidemiological studies have revealed that the incidence of ND, such as Parkinson Disease (PD), Multiple Sclerosis (MS), and Alzheimer Diseases (AD), are associated with changes in the circulating sex hormones, i.e., they are more prevalent in menopause women (Vegeto, Benedusi & Maggi, 2008; Bhavnani, 2003). Therefore, the neuroprotective role of these hormones for the treatment of the ND such as AD has been highlighted (Zandi et al., 2002; Henderson et al., 2005). Regarding the role of free radicals and oxidative stress in the pathophysiology of ND, attention has paid recently to the importance of the use of antioxidants agents in ND therapy (Giordano, Darley-Usmar & Zhang, 2014; Calabrese et al., 2009). Apigenin, is an effective ingredient of the chamomile plant and a neuroprotective flavonoid that has antioxidant and anti-inflammatory properties. Regarding the neuroprotective role of the ovarian sex hormones and the antioxidant effects of apigenin, the present study was designed to find the combined effects of exogenous β-estradiol hormone and apigenin on the formation of β-amyloid plaques in the hippocampus and memory and learning deficiency in adult menopause rats.
2. Methods
2.1. Animal model
Forty-two adult female rats (weight: 200-250 g) were used in this study and randomly divided into seven groups: 1) ovariectomy (OVX), 2) OVX with β-estradiol (OVX + E2), 3) OVX with apigenin (OVX + API), 4) OVX with apigenin and β-estradiol (OVX + E2 + API), 5 &6) vehicle shams for E2 and API, and 7) surgical sham. All study procedures were done by the ethical code (IR.IUMS.REC 1396.327 24) of research and animal rights approved by Neuroscience Research Center.
2.2. Ovariectomy (OVX)
Bilateral ovariectomy was performed under anesthesia and sterile condition similar to previous studies (Baeza, De Castro, Giménez-Llort & De la Fuente, 2010). Histological study of the ovaries and the plasma level of ovarian hormones was performed to verify the model.
2.3. Administration of β-estradiol and apigenin
We administered 10 μL β-Estradiol (Sigma E8875) containing 20 μg of β-estradiol soluble in sesame oil intramuscularly three weeks after ovariectomy. Sesame oil injection in this group was performed only once a week after ovariectomy and the first stage of the Morris Wter Maze test (MWM). The steps were as follows: 1) ovariectomy, 2) the first stage of the Morris test, 3) injection for 5 weeks, and 4) the second stage of the Morris test.
We used 33 μL apigenin (Sigma A3145) containing 2 mg of apigenin, solved in ethanol, and injected intraperitoneally for 5 weeks with an interval of 3 days. The OVX + E2 + Api group received the compounds simultaneously. The vehicle sham group received only the solvents. The surgical sham group underwent surgery without removing ovaries. Immediately after the end of the injection period, the second stage of the MWM was performed for memory evaluation. Afterward, the animals were transcardially perfused and fixed (4% paraformaldehyde in PBS 0.1 M/ pH of 7.4); their brains were removed and cross-sectioned with a thickness of 10 µ prepared using freezing microtome based on the Paxinos Rat Brain Atlas.
2.4. Microscopic study
Nissl staining was used to study the neuronal count and density in the hippocampus area. To count and identify β-amyloid plaques, the Congo red staining was used. Using the ImageJ software, counting of neurons and β-amyloid plaques was done in the right and left hippocampus areas.
2.5. Behavioral study
MWM was used to study learning and memory. The first stage was done three weeks after the ovariectomy for 4 consecutive days, comprising three days of training and one day of the test. The first three days of the test, for training and learning, were performed on all animals. Then the animals were grouped, and the fourth day of the test, which was the first stage of remembering (probe 1), was performed on animals of other groups. After administering the drug and at the end of the treatment period, the second stage of the MWM (probe 2) was carried out.
2.6. Molecular study
2.6.1. Western blotting
The cortex and the hippocampus areas were homogenized by an ice-cold buffer (0.1% SDS, 0.1% Na deoxycholate), 10 mM Tris–HCl (pH 7.4), 1 mM EDTA, 1% NP-40 with protease inhibitors (2.5 mg/mL of leupeptin, 1 mM phenylmethylsulfonyl fluoride, 10 mg/mL of aprotinin), and 1 mM sodium orthovanadate. The homogenized solution was centrifuged at 14000 rpm at 4ºC for 15 min. The next supernatant, as the cell fraction, was retained. The Bradford method was used to measure protein concentrations (Bio-Rad Laboratories, München, Germany). The same quantities of protein were resolved electrophoretically on a 9% SDS-PAGE gel and transferred to nitrocellulose membranes (Hybond ECL, GE Healthcare Bio-Sciences Corp., Piscataway, NJ, USA). After blocking (overnight at 4ºC) with 5% non-fat dried milk in Tris-buffered saline with Tween 20 (blocking buffer, TBS-T, 150 mM NaCl, 20 mM Tris-HCl, pH 7.5, 0.1% Tween 20), the membranes were explored with caspase-3 rabbit polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA), 1:500 for 1 h at room temperature. After being washed in TBS-T (three times, 5 min), the blots were incubated at room temperature for 60 min with HRP conjugated secondary antibody (1:15000, GE Healthcare Bio-Sciences Corp.). The antibody was diluted in the blocking buffer. The antibody-antigen complexes were discovered by the ECL system and exposed to Lumi-Film Chemiluminescent detection film (Roche Applied Science, Mannheim, Germany). For exploring the intensity of expression, the Lab Work analyzing software was used (UVP, Cambridge, UK). β-Actin immunoblotting (Cell Signaling Technology, Inc.; 1:1000) was used as a loading control.
2.6.2. Statistical analysis
The obtained data were analyzed in SPSS v. 21 and Prism 5. Two-way Analysis of Variance (ANOVA) was used to assess memory and learning in animals, and 1-way ANOVA to analyze the results of the research hypotheses. Density values of caspase-3 and β-actin proteins were measured by band densitometry. For each sample, these values were calculated from the ratio of the desired protein to β-actin. The results were analyzed by 1-way ANOVA and then by the Student-Newman-Keuls test. The study findings were presented as Mean±Standard Deviation (SD), and P values less than 0.05 are considered significant.
3. Results
3.1. β-Amyloid plaque count
β-Amyloid plaques formation in the right and left hippocampus is shown in Figures 1 & 2.
.png)
.png)
The number of plaques in the right and left hippocampus in the OVX+Api, OVX+ E2, and OVX+E2+Api groups were significantly lower than OVX animals (P<0.05) (Figure 3 A & B).
.png)
In Figure 4, the results were compared in two areas of the left and right hippocampus in all groups. Accordingly, the best therapeutic results were observed in OVX+ Api+ E2 (P<0.05).
.png)
3.2. Neuronal count
Live and necrotic neurons in the hippocampus were studied by Nissl staining. As it is shown in Figure 5, the decreased number of live neurons and increased number of necrotic neurons in the hippocampus was significant following OVX.
.png)
Comparing the results for OVX to OVX+ Api, OVX+ E2, OVX+ E2 + Api showed significant effects of administration of OVX+ E2 + Api. The best results were seen in the animals treated with apigenin and β-estradiol simultaneously (Figure 6A & B).
.png)
3.3. Results of Morris Water Maze (MWM) test
The distance traveled and the time spent to reach the hidden platform has been declined significantly in all OVX groups comparing to surgical sham animals within three days (P<0.05, Figure 7A & B).
.png)
Analysis of data from probe 2 of remembering in OVX, OVX+Api, OVX+ E2, OVX+E2+Api, OVX+ E2 vehicle, and OVX+ Api vehicle showed no difference between the OVX and E2 groups in terms of time duration in the target quadrant. However, a comparison of OVX+ Api and OVX+Api+E2 groups with the OVX group showed that the time elapsed in the target quadrant increased under the influence of treatment. However, this increase was only significant for the combined apigenin and β-estradiol group (P<0.05, Figure 8A). Figure 8B shows the comparison between the times elapsed in the target quadrant between the first and second steps of remembering. As the Figure shows, the OVX+Api+E2 animals in the second stage spent more time in the target quarter, indicating a better effect of the combination of E2 and apigenin on memory and learning than OVX, OVX+E2, and OVX+ Api groups (P <0.05, Figure 8A).

3.4. Molecular results
Figure 9 shows a representative Western blot of caspase-3 expression in the hippocampus of different groups.
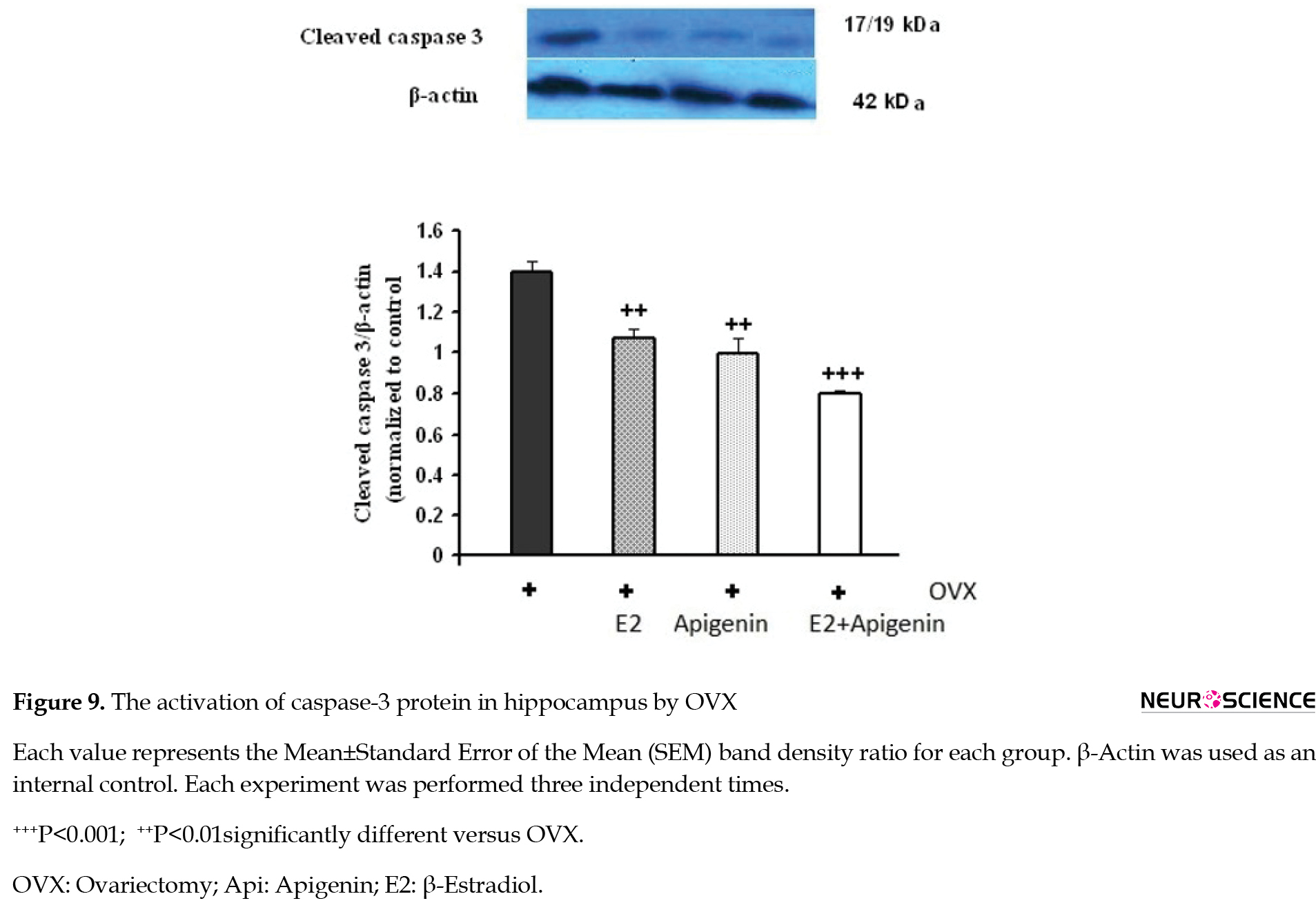
OVX activated caspase-3 in the hippocampus, and administration of E2 + apigenin inhibited caspase-3 activation. As this Figure shows, the amount of cleaved caspase-3 in the hippocampus of ovariectomized animals was higher than the treated groups (P<0.05). In the E2 group, as well as the apigenin, the amount of caspase-3 decreased significantly. In the E2 + Api group, the caspase-3 decrease rate was the highest (P<0.001).
4. Discussion
The significant difference in the formation of β-amyloid plaques in the hippocampus was seen between OVX and treated groups. These results indicate that β-estradiol and apigenin may inhibit the formation of β-amyloid plaques with better results in E2 + Api treated animals. Our study showed that ovarian hormones deprivation plays an essential role in the formation of β-amyloid plaques. Nilsen et al. (2006) reported that the treatment with β-estradiol significantly reduced β-amyloid plaques in the hippocampus area (Nilsen, Chen, Irwin, Iwamoto & Brinton, 2006). In a study on 82 postmenopausal women to examine their sexual hormones balance, the incidence of dementia increased in whom that the level of hormones had decreased more (Robusto-Leitao & Ferreira, 2006). The long-term decline in ovarian function reduces the choline acetyltransferase function. In other words, β-amyloid prevents acetylcholine secretion and choline absorption in different parts of the hippocampus. The results of various studies indicate that β-amyloid can worsen cholinergic secretion due to long-term sex steroid deprivation that leads to β-amyloid plaque formation (Baeza et al., 2010). Clinical studies confirm that estrogen therapy in postmenopausal women decreases or delays the risk of developing AD. In 1999, Yamada et al. reported the role of estrogen therapy in improving AD. They showed that estrogen reduces memory and learning impairment in ovariectomized rats and reduces the neuronal death and number of β-amyloid plaques (Yamada et al., 1999). AD may be more destructive in women, which can be due to gender differences. The plasma estrogen level in women with AD was reported to be less than women who do not suffer from this disease and are matched in age. Therefore, short-term use of β-estradiol can reduce the risk of β-amyloid formation among menopausal women (Craig, Hong & McDonald, 2011). The well-known neuroprotective effect of sex steroids could consider the hypothesis in reducing the incidence of AD. Pike et al. showed that β-estradiol inhibits apoptosis via reducing cell death induced by glutamate. Jameie et al. showed that neural changes following sex steroid deprivation, including neuronal death and changes in dendritic structures, are reversible (Jameie et al., 2016). Moreover, estrogen reduces the formation of the primary amyloid protein through the protein kinase, a potent activator of the non-amyloid process (Pike et al., 2009). β-Estradiol reduces the production of β-amyloid plaques via acting against oxidative stress by regulating mitochondrial function (Amtul, Wang, Westaway & Rozmahel, 2010). The insulin-degrading enzyme is an essential factor in the degradation of β-amyloid plaques, which is regulated via the PI3K/AKT/mTOR pathway (Guo, Xi & Li, 2019). The presence of β-estradiol is essential for regulating brain-derived neurotrophic factor which is widely distributed in the brain, especially in the hippocampus that plays a role in memory (Zhao, Zheng, Xu & Lin, 2019). Apigenin is well known for its anti-oxidant, anti-inflammatory, anti-carcinogenic, and inhibitory effects for pro-inflammatory mediators (Venigalla, Sonego, Gyengesi & Münch, 2015). Apigenin improves the cholinergic transmission and protects the strength and integrity of the blood-brain barrier against β-amyloid injection. In addition, apigenin plays a vital role in reducing the level of the primary β-amyloid protein and preventing the accumulation of β-amyloid in different brain areas (Zhao, Woody & Chhibber, 2015). Electrophysiological studies have shown that apigenin regulates the neuronal transfer of GABA and glutamate. It affects synaptic receptors and modulates the function of GABA receptors. Apigenin also acts against glutamate-induced poisoning (Losi, Puia, Garzon, de Vuono & Baraldi, 2004). It also enhances the antioxidant activity of the superoxide enzyme. Zhao et al. (2013) argued that apigenin protects neurons against direct poisoning with β-amyloid through the mechanism of the antioxidative reaction, maintaining the mitochondrial function, and preventing cell death (Zhao, Wang, Wang & Fa, 2013). Venigalla et al. (2015) reported the anti-inflammatory role of apigenin. Regarding anti-inflammatory effects, apigenin inhibits inflammatory proteins such as Interleukin (IL)-6, IL-1β, and tumor necrosis factor-α by deactivating NF-κB. By inhibiting the cytochrome C release, apigenin protects against apoptosis induced by caspase-3 and caspase-7.
5. Conclusion
Concurrent use of hormone therapy and antioxidants can adjust the cognitive dysfunction in AD during menopause, and it is the promising result of this study that requires more research.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of Iran University of Medical Sciences (Code: IR.IUMS.REC.1396.327.24); Also this study was approved by the animal rights of Neuroscience Research Center.
Funding
The present research was supported by the Neuroscience Research Center of Iran University of Medical Sciences and a research grant by Iran University of Medical Sciences.
Authors' contributions
Principal Investigator and study design: Seyed Behnamedin Jameie & Mona Farhadi; Methodology (microscopic & behavioral study): Abbas Pirasteh, Seyed Behnamedin Jameie, Javad Fahanik Babaee & Ali Naseri; Methodology (Molecular study): Mona Farhadi, Leila Elyasi & Melika Sadat Jameie; Data analysis: Melika Sadat Jameie & Leila Elyasi; Writing and preparing manuscript: Seyed Behnamedin Jameie, Melika Sadat Jameie & Mona Farhadi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors would like to thank the Neurohistology and Behavioral Lab of Neuroscience Research Center, Islamic Azad University, Firozabad Branch, and Department of Psychology for their collaboration.
References
Amtul, Z., Wang, L., Westaway, D., & Rozmahel, R. F. (2010). Neuroprotective mechanism conferred by 17beta-estradiol on the biochemical basis of Alzheimer’s disease. Neuroscience, 169(2), 781-6. [DOI:10.1016/j.neuroscience.2010.05.031] [PMID]
Baeza, I., De Castro, N. M., Giménez-Llort, L., & De la Fuente, M. (2010). Ovariectomy, a model of menopause in rodents, causes a premature aging of the nervous and immune systems. Journal of Neuroimmunology, 219(1-2), 90-9. [DOI:10.1016/j.jneuroim.2009.12.008] [PMID]
Bhavnani, B. R. (2003). Estrogens and menopause: Pharmacology of conjugated equine estrogens and their potential role in the prevention of neurodegenerative diseases such as Alzheimer’s. The Journal of Steroid Biochemistry and Molecular Biology, 85(2-5), 473-82. [DOI:10.1016/S0960-0760(03)00220-6]
Boumenir, A., Cognat, E., Sabia, S., Hourregue, C., Lilamand, M., & Dugravot, A., et al. (2019). CSF level of β-amyloid peptide predicts mortality in Alzheimer’s disease. Alzheimer’s Research & Therapy, 11(1), 29. [DOI:10.1186/s13195-019-0481-4] [PMID] [PMCID]
Calabrese, V., Cornelius, C., Mancuso, C., Barone, E., Calafato, S., & Bates, T., et al. (2009). Vitagenes, dietary antioxidants and neuroprotection in neurodegenerative diseases. Frontiers in Bioscience-Landmark, 14(1), 376-97. [DOI:10.2741/3250] [PMID]
Cheng, X. L., & Li, M. K. (2014). Effect of topiramate on apoptosis-related protein expression of hippocampus in model rats with Alzheimers disease. European Review for Medical and Pharmacological Sciences, 18(6), 761-8. [PMID]
Craig, L. A., Hong, N. S., & McDonald, R. J. (2011). Revisiting the cholinergic hypothesis in the development of Alzheimer’s disease. Neuroscience & Biobehavioral Reviews, 35(6), 1397-409. [DOI:10.1016/j.neubiorev.2011.03.001] [PMID]
Giffard, B., Desgranges, B., & Eustache, F. (2005). Semantic memory disorders in Alzheimer’s disease: Clues from semantic priming effects. Current Alzheimer Research, 2(4), 425-34. [DOI:10.2174/156720505774330582] [PMID]
Giordano, S., Darley-Usmar, V., & Zhang, J. (2014). Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biology, 2, 82-90. [DOI:10.1016/j.redox.2013.12.013] [PMID] [PMCID]
Guo, Sh., Xi, X., & Li, J. (2019). Notoginsenoside R1: A systematic review of its pharmacological properties. Die Pharmazie - An International Journal of Pharmaceutical Sciences, 74(11), 641-7. [DOI:10.1691/ph.2019.9534] [PMID]
Hatashita, Sh., & Wakebe, D. (2019). Amyloid β deposition and glucose metabolism on the long-term progression of preclinical Alzheimer’s disease. Future Science OA, 5(3), FSO356. [DOI:10.4155/fsoa-2018-0069] [PMID] [PMCID]
Henderson, V. W., Benke, K. S., Green, R. C., Cupples, L. A., Farrer, L. A., & MIRAGE Study Group. (2005). Postmenopausal hormone therapy and Alzheimer’s disease risk: Interaction with age. Journal of Neurology, Neurosurgery & Psychiatry, 76(1), 103-5. [DOI:10.1136/jnnp.2003.024927] [PMID] [PMCID]
Jameei, S. B., Nouyan-Ashraf, M. H., & Behzadi, G. (2004). Ovariectomy reduces the dendritic spine density of the dorsal raphe neurons in the adult rat. Archives of Iranian Medicine, 7(2), 122-7. https://www.sid.ir/en/journal/ViewPaper.aspx?ID=4729
Jameie, B., Mehdinejadiani, Sh., Farhadi, M., Solimani, M., Hassanzadeh, S., & Kerdari, M., et al. (2016). Effects of exogenous estrogen treatment on hippocampal neurogenesis of diabetic ovariectomized rats. International Clinical Neuroscience Journal, 3(2), 99-108. [DOI:10.22037/icnj.v3i2.13766]
Krishnan, S., Slavin, M. J., Tran, T. T. T., Murali Doraiswamy, P., & Petrella, J. R. (2006). Accuracy of spatial normalization of the hippocampus: Implications for fMRI research in memory disorders. NeuroImage, 31(2), 560-71. [DOI:10.1016/j.neuroimage.2005.12.061] [PMID]
Lee, J., Park, H. H., Koh, S. H., & Choi, H. (2017). Neural stem cell death mechanisms induced by amyloid beta. Dementia and Neurocognitive Disorders, 16(4), 121-7. [DOI:10.12779/dnd.2017.16.4.121] [PMID] [PMCID]
Losi, G., Puia, G., Garzon, G., de Vuono, M. C., & Baraldi, M. (2004). Apigenin modulates GABAergic and glutamatergic transmission in cultured cortical neurons. European Journal of Pharmacology, 502(1-2), 41-6. [DOI:10.1016/j.ejphar.2004.08.043] [PMID]
Nilsen, J., Chen, Sh., Irwin, R. W., Iwamoto, S., & Brinton, R. D. (2006). Estrogen protects neuronal cells from amyloid beta-induced apoptosis via regulation of mitochondrial proteins and function. BMC Neuroscience, 7, 74. [DOI:10.1186/1471-2202-7-74] [PMID] [PMCID]
Pike, Ch. J., Carroll, J. C., Rosario, E. R., & Barron, A. M. (2009). Protective actions of sex steroid hormones in Alzheimer’s disease. Frontiers in Neuroendocrinology, 30(2), 239-58. [DOI:10.1016/j.yfrne.2009.04.015] [PMID] [PMCID]
Prakash, A., & Kumar, A. (2014). Implicating the role of lycopene in restoration of mitochondrial enzymes and BDNF levels in β-amyloid induced Alzheimer׳s disease. European Journal of Pharmacology, 741, 104-11. [DOI:10.1016/j.ejphar.2014.07.036] [PMID]
Robusto-Leitao, O., & Ferreira, H. (2006). Hormones and dementia - a comparative study of hormonal impairment in post-menopausal women, with and without dementia. Neuropsychiatric Disease and Treatment, 2(2), 199-206. [DOI:10.2147/nedt.2006.2.2.199] [PMID] [PMCID]
Sandoval, K. E., & Witt, K. A. (2011). Age and 17β-estradiol effects on blood-brain barrier tight junction and estrogen receptor proteins in ovariectomized rats. Microvascular Research, 81(2), 198-205. [DOI:10.1016/j.mvr.2010.12.007] [PMID]
Vegeto, E., Benedusi, V., & Maggi, A. (2008). Estrogen anti-inflammatory activity in brain: A therapeutic opportunity for menopause and neurodegenerative diseases. Frontiers in Neuroendocrinology, 29(4), 507-19. [DOI:10.1016/j.yfrne.2008.04.001] [PMID] [PMCID]
Venigalla, M., Sonego, S., Gyengesi, E., & Münch, G. (2015). Curcumin and Apigenin - novel and promising therapeutics against chronic neuroinflammation in Alzheimer’s disease. Neural Regeneration Research, 10(8), 1181-5. [DOI:10.4103/1673-5374.162686] [PMID] [PMCID]
Vest, R. S., & Pike, Ch. J. (2013). Gender, sex steroid hormones, and Alzheimer’s disease. Hormones and Behavior, 63(2), 301-7. [DOI:10.1016/j.yhbeh.2012.04.006] [PMID] [PMCID]
Wang, F., Zhou, X. L., Yang, Q. G., Xu, W. H., Wang, F., & Chen, Y. P., et al. (2011). A peptide that binds specifically to the β-amyloid of Alzheimer’s disease: Selection and assessment of anti-β-amyloid neurotoxic effects. PLoS One, 6(11), e27649. [DOI:10.1371/journal.pone.0027649] [PMID] [PMCID]
Yamada, K., Tanaka, T., Zou, L. B., Senzaki, K., Yano, K., & Osada, T., et al. (1999). Long‐term deprivation of oestrogens by ovariectomy potentiates β‐amyloid‐induced working memory deficits in rats. British Journal of Pharmacology, 128(2), 419-27. [DOI:10.1038/sj.bjp.0702811] [PMID] [PMCID]
Zandi, P. P., Carlson, M. C., Plassman, B. L., Welsh-Bohmer, K. A., Mayer, L. S., & Steffens, D. C., et al. (2002). Hormone replacement therapy and incidence of Alzheimer disease in older women: The Cache County Study. JAMA, 288(17), 2123-9. [DOI:10.1001/jama.288.17.2123] [PMID]
Zhao, Y., Zheng, H. X., Xu, Y., & Lin, N. (2019). Estrogenic effect of the extract of QingYan formula on reproductive tissues in immature mice. Evidence-Based Complementary and Alternative Medicine, 2019, 5493714. [DOI:10.1155/2019/5493714] [PMID] [PMCID]
Zhao, L., Woody, S. K., & Chhibber, A. (2015). Estrogen receptor β in Alzheimer’s disease: From mechanisms to therapeutics. Ageing Research Reviews, 24(Pt B), 178-90. [DOI:10.1016/j.arr.2015.08.001] [PMID] [PMCID]
Zhao, L., Wang, J. L., Wang, Y. R., & Fa, X. Z. (2013). Apigenin attenuates copper-mediated β-amyloid neurotoxicity through antioxidation, mitochondrion protection and MAPK signal inactivation in an AD cell model. Brain Research, 1492, 33-45. [DOI:10.1016/j.brainres.2012.11.019] [PMID]
Neurodegenerative Diseases (ND) are age-dependent. Other factors, including genetics and gender, also affect the incidence, severity, response to treatment, and mortality. Cellular and molecular changes have been reported previously. The formation of β-amyloid plaques, changes of dendritic and synaptic structures, and neuronal death has been reported in AD (Cheng & Li, 2014; Lee, Park, Koh & Choi, 2017). AD starts around 60- 65 years old with clinical symptoms, such as memory and speech impairment, psychological and psychiatric changes. Studies have emphasized the role of β-amyloid plaques in the cortex and hippocampus, leading to AD symptoms (Hatashita & Wakebe, 2019; Boumenir et al., 2019; Wang et al., 2011; Prakash & Kumar, 2014). Memory impairment and cognitive dysfunction in AD are associated with cellular and molecular changes in the hippocampus (Krishnan, Slavin, Tran, Murali Doraiswamy & Petrella, 2006; Giffard, Desgranges & Eustache, 2005). Other factors, including oxidative stress, vascular, metabolic, hormonal, and neurotransmitter changes, influence the formation of β-amyloid plaques and Neurofibrillary Tangles (NFTs). The neuroprotective effect of ovarian sex steroids has been mentioned in several studies (Jameei, Nouyan-Ashraf & Behzadi, 2004; Jameie et al., 2016). These hormones easily pass through the blood-brain barrier and affect nervous tissue (Sandoval & Witt, 2011). The level of circulating sex hormones affects physiopathology, symptoms, severity, and treatment of ND (Vest & Pike, 2013; Pike, Carroll, Rosario & Barron, 2009). Epidemiological studies have revealed that the incidence of ND, such as Parkinson Disease (PD), Multiple Sclerosis (MS), and Alzheimer Diseases (AD), are associated with changes in the circulating sex hormones, i.e., they are more prevalent in menopause women (Vegeto, Benedusi & Maggi, 2008; Bhavnani, 2003). Therefore, the neuroprotective role of these hormones for the treatment of the ND such as AD has been highlighted (Zandi et al., 2002; Henderson et al., 2005). Regarding the role of free radicals and oxidative stress in the pathophysiology of ND, attention has paid recently to the importance of the use of antioxidants agents in ND therapy (Giordano, Darley-Usmar & Zhang, 2014; Calabrese et al., 2009). Apigenin, is an effective ingredient of the chamomile plant and a neuroprotective flavonoid that has antioxidant and anti-inflammatory properties. Regarding the neuroprotective role of the ovarian sex hormones and the antioxidant effects of apigenin, the present study was designed to find the combined effects of exogenous β-estradiol hormone and apigenin on the formation of β-amyloid plaques in the hippocampus and memory and learning deficiency in adult menopause rats.
2. Methods
2.1. Animal model
Forty-two adult female rats (weight: 200-250 g) were used in this study and randomly divided into seven groups: 1) ovariectomy (OVX), 2) OVX with β-estradiol (OVX + E2), 3) OVX with apigenin (OVX + API), 4) OVX with apigenin and β-estradiol (OVX + E2 + API), 5 &6) vehicle shams for E2 and API, and 7) surgical sham. All study procedures were done by the ethical code (IR.IUMS.REC 1396.327 24) of research and animal rights approved by Neuroscience Research Center.
2.2. Ovariectomy (OVX)
Bilateral ovariectomy was performed under anesthesia and sterile condition similar to previous studies (Baeza, De Castro, Giménez-Llort & De la Fuente, 2010). Histological study of the ovaries and the plasma level of ovarian hormones was performed to verify the model.
2.3. Administration of β-estradiol and apigenin
We administered 10 μL β-Estradiol (Sigma E8875) containing 20 μg of β-estradiol soluble in sesame oil intramuscularly three weeks after ovariectomy. Sesame oil injection in this group was performed only once a week after ovariectomy and the first stage of the Morris Wter Maze test (MWM). The steps were as follows: 1) ovariectomy, 2) the first stage of the Morris test, 3) injection for 5 weeks, and 4) the second stage of the Morris test.
We used 33 μL apigenin (Sigma A3145) containing 2 mg of apigenin, solved in ethanol, and injected intraperitoneally for 5 weeks with an interval of 3 days. The OVX + E2 + Api group received the compounds simultaneously. The vehicle sham group received only the solvents. The surgical sham group underwent surgery without removing ovaries. Immediately after the end of the injection period, the second stage of the MWM was performed for memory evaluation. Afterward, the animals were transcardially perfused and fixed (4% paraformaldehyde in PBS 0.1 M/ pH of 7.4); their brains were removed and cross-sectioned with a thickness of 10 µ prepared using freezing microtome based on the Paxinos Rat Brain Atlas.
2.4. Microscopic study
Nissl staining was used to study the neuronal count and density in the hippocampus area. To count and identify β-amyloid plaques, the Congo red staining was used. Using the ImageJ software, counting of neurons and β-amyloid plaques was done in the right and left hippocampus areas.
2.5. Behavioral study
MWM was used to study learning and memory. The first stage was done three weeks after the ovariectomy for 4 consecutive days, comprising three days of training and one day of the test. The first three days of the test, for training and learning, were performed on all animals. Then the animals were grouped, and the fourth day of the test, which was the first stage of remembering (probe 1), was performed on animals of other groups. After administering the drug and at the end of the treatment period, the second stage of the MWM (probe 2) was carried out.
2.6. Molecular study
2.6.1. Western blotting
The cortex and the hippocampus areas were homogenized by an ice-cold buffer (0.1% SDS, 0.1% Na deoxycholate), 10 mM Tris–HCl (pH 7.4), 1 mM EDTA, 1% NP-40 with protease inhibitors (2.5 mg/mL of leupeptin, 1 mM phenylmethylsulfonyl fluoride, 10 mg/mL of aprotinin), and 1 mM sodium orthovanadate. The homogenized solution was centrifuged at 14000 rpm at 4ºC for 15 min. The next supernatant, as the cell fraction, was retained. The Bradford method was used to measure protein concentrations (Bio-Rad Laboratories, München, Germany). The same quantities of protein were resolved electrophoretically on a 9% SDS-PAGE gel and transferred to nitrocellulose membranes (Hybond ECL, GE Healthcare Bio-Sciences Corp., Piscataway, NJ, USA). After blocking (overnight at 4ºC) with 5% non-fat dried milk in Tris-buffered saline with Tween 20 (blocking buffer, TBS-T, 150 mM NaCl, 20 mM Tris-HCl, pH 7.5, 0.1% Tween 20), the membranes were explored with caspase-3 rabbit polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA), 1:500 for 1 h at room temperature. After being washed in TBS-T (three times, 5 min), the blots were incubated at room temperature for 60 min with HRP conjugated secondary antibody (1:15000, GE Healthcare Bio-Sciences Corp.). The antibody was diluted in the blocking buffer. The antibody-antigen complexes were discovered by the ECL system and exposed to Lumi-Film Chemiluminescent detection film (Roche Applied Science, Mannheim, Germany). For exploring the intensity of expression, the Lab Work analyzing software was used (UVP, Cambridge, UK). β-Actin immunoblotting (Cell Signaling Technology, Inc.; 1:1000) was used as a loading control.
2.6.2. Statistical analysis
The obtained data were analyzed in SPSS v. 21 and Prism 5. Two-way Analysis of Variance (ANOVA) was used to assess memory and learning in animals, and 1-way ANOVA to analyze the results of the research hypotheses. Density values of caspase-3 and β-actin proteins were measured by band densitometry. For each sample, these values were calculated from the ratio of the desired protein to β-actin. The results were analyzed by 1-way ANOVA and then by the Student-Newman-Keuls test. The study findings were presented as Mean±Standard Deviation (SD), and P values less than 0.05 are considered significant.
3. Results
3.1. β-Amyloid plaque count
β-Amyloid plaques formation in the right and left hippocampus is shown in Figures 1 & 2.
.png)
.png)
The number of plaques in the right and left hippocampus in the OVX+Api, OVX+ E2, and OVX+E2+Api groups were significantly lower than OVX animals (P<0.05) (Figure 3 A & B).
.png)
In Figure 4, the results were compared in two areas of the left and right hippocampus in all groups. Accordingly, the best therapeutic results were observed in OVX+ Api+ E2 (P<0.05).
.png)
3.2. Neuronal count
Live and necrotic neurons in the hippocampus were studied by Nissl staining. As it is shown in Figure 5, the decreased number of live neurons and increased number of necrotic neurons in the hippocampus was significant following OVX.
.png)
Comparing the results for OVX to OVX+ Api, OVX+ E2, OVX+ E2 + Api showed significant effects of administration of OVX+ E2 + Api. The best results were seen in the animals treated with apigenin and β-estradiol simultaneously (Figure 6A & B).
.png)
3.3. Results of Morris Water Maze (MWM) test
The distance traveled and the time spent to reach the hidden platform has been declined significantly in all OVX groups comparing to surgical sham animals within three days (P<0.05, Figure 7A & B).
.png)
Analysis of data from probe 2 of remembering in OVX, OVX+Api, OVX+ E2, OVX+E2+Api, OVX+ E2 vehicle, and OVX+ Api vehicle showed no difference between the OVX and E2 groups in terms of time duration in the target quadrant. However, a comparison of OVX+ Api and OVX+Api+E2 groups with the OVX group showed that the time elapsed in the target quadrant increased under the influence of treatment. However, this increase was only significant for the combined apigenin and β-estradiol group (P<0.05, Figure 8A). Figure 8B shows the comparison between the times elapsed in the target quadrant between the first and second steps of remembering. As the Figure shows, the OVX+Api+E2 animals in the second stage spent more time in the target quarter, indicating a better effect of the combination of E2 and apigenin on memory and learning than OVX, OVX+E2, and OVX+ Api groups (P <0.05, Figure 8A).

3.4. Molecular results
Figure 9 shows a representative Western blot of caspase-3 expression in the hippocampus of different groups.

OVX activated caspase-3 in the hippocampus, and administration of E2 + apigenin inhibited caspase-3 activation. As this Figure shows, the amount of cleaved caspase-3 in the hippocampus of ovariectomized animals was higher than the treated groups (P<0.05). In the E2 group, as well as the apigenin, the amount of caspase-3 decreased significantly. In the E2 + Api group, the caspase-3 decrease rate was the highest (P<0.001).
4. Discussion
The significant difference in the formation of β-amyloid plaques in the hippocampus was seen between OVX and treated groups. These results indicate that β-estradiol and apigenin may inhibit the formation of β-amyloid plaques with better results in E2 + Api treated animals. Our study showed that ovarian hormones deprivation plays an essential role in the formation of β-amyloid plaques. Nilsen et al. (2006) reported that the treatment with β-estradiol significantly reduced β-amyloid plaques in the hippocampus area (Nilsen, Chen, Irwin, Iwamoto & Brinton, 2006). In a study on 82 postmenopausal women to examine their sexual hormones balance, the incidence of dementia increased in whom that the level of hormones had decreased more (Robusto-Leitao & Ferreira, 2006). The long-term decline in ovarian function reduces the choline acetyltransferase function. In other words, β-amyloid prevents acetylcholine secretion and choline absorption in different parts of the hippocampus. The results of various studies indicate that β-amyloid can worsen cholinergic secretion due to long-term sex steroid deprivation that leads to β-amyloid plaque formation (Baeza et al., 2010). Clinical studies confirm that estrogen therapy in postmenopausal women decreases or delays the risk of developing AD. In 1999, Yamada et al. reported the role of estrogen therapy in improving AD. They showed that estrogen reduces memory and learning impairment in ovariectomized rats and reduces the neuronal death and number of β-amyloid plaques (Yamada et al., 1999). AD may be more destructive in women, which can be due to gender differences. The plasma estrogen level in women with AD was reported to be less than women who do not suffer from this disease and are matched in age. Therefore, short-term use of β-estradiol can reduce the risk of β-amyloid formation among menopausal women (Craig, Hong & McDonald, 2011). The well-known neuroprotective effect of sex steroids could consider the hypothesis in reducing the incidence of AD. Pike et al. showed that β-estradiol inhibits apoptosis via reducing cell death induced by glutamate. Jameie et al. showed that neural changes following sex steroid deprivation, including neuronal death and changes in dendritic structures, are reversible (Jameie et al., 2016). Moreover, estrogen reduces the formation of the primary amyloid protein through the protein kinase, a potent activator of the non-amyloid process (Pike et al., 2009). β-Estradiol reduces the production of β-amyloid plaques via acting against oxidative stress by regulating mitochondrial function (Amtul, Wang, Westaway & Rozmahel, 2010). The insulin-degrading enzyme is an essential factor in the degradation of β-amyloid plaques, which is regulated via the PI3K/AKT/mTOR pathway (Guo, Xi & Li, 2019). The presence of β-estradiol is essential for regulating brain-derived neurotrophic factor which is widely distributed in the brain, especially in the hippocampus that plays a role in memory (Zhao, Zheng, Xu & Lin, 2019). Apigenin is well known for its anti-oxidant, anti-inflammatory, anti-carcinogenic, and inhibitory effects for pro-inflammatory mediators (Venigalla, Sonego, Gyengesi & Münch, 2015). Apigenin improves the cholinergic transmission and protects the strength and integrity of the blood-brain barrier against β-amyloid injection. In addition, apigenin plays a vital role in reducing the level of the primary β-amyloid protein and preventing the accumulation of β-amyloid in different brain areas (Zhao, Woody & Chhibber, 2015). Electrophysiological studies have shown that apigenin regulates the neuronal transfer of GABA and glutamate. It affects synaptic receptors and modulates the function of GABA receptors. Apigenin also acts against glutamate-induced poisoning (Losi, Puia, Garzon, de Vuono & Baraldi, 2004). It also enhances the antioxidant activity of the superoxide enzyme. Zhao et al. (2013) argued that apigenin protects neurons against direct poisoning with β-amyloid through the mechanism of the antioxidative reaction, maintaining the mitochondrial function, and preventing cell death (Zhao, Wang, Wang & Fa, 2013). Venigalla et al. (2015) reported the anti-inflammatory role of apigenin. Regarding anti-inflammatory effects, apigenin inhibits inflammatory proteins such as Interleukin (IL)-6, IL-1β, and tumor necrosis factor-α by deactivating NF-κB. By inhibiting the cytochrome C release, apigenin protects against apoptosis induced by caspase-3 and caspase-7.
5. Conclusion
Concurrent use of hormone therapy and antioxidants can adjust the cognitive dysfunction in AD during menopause, and it is the promising result of this study that requires more research.
Ethical Considerations
Compliance with ethical guidelines
This study was approved by the Ethics Committee of Iran University of Medical Sciences (Code: IR.IUMS.REC.1396.327.24); Also this study was approved by the animal rights of Neuroscience Research Center.
Funding
The present research was supported by the Neuroscience Research Center of Iran University of Medical Sciences and a research grant by Iran University of Medical Sciences.
Authors' contributions
Principal Investigator and study design: Seyed Behnamedin Jameie & Mona Farhadi; Methodology (microscopic & behavioral study): Abbas Pirasteh, Seyed Behnamedin Jameie, Javad Fahanik Babaee & Ali Naseri; Methodology (Molecular study): Mona Farhadi, Leila Elyasi & Melika Sadat Jameie; Data analysis: Melika Sadat Jameie & Leila Elyasi; Writing and preparing manuscript: Seyed Behnamedin Jameie, Melika Sadat Jameie & Mona Farhadi.
Conflict of interest
The authors declared no conflict of interest.
Acknowledgments
The authors would like to thank the Neurohistology and Behavioral Lab of Neuroscience Research Center, Islamic Azad University, Firozabad Branch, and Department of Psychology for their collaboration.
References
Amtul, Z., Wang, L., Westaway, D., & Rozmahel, R. F. (2010). Neuroprotective mechanism conferred by 17beta-estradiol on the biochemical basis of Alzheimer’s disease. Neuroscience, 169(2), 781-6. [DOI:10.1016/j.neuroscience.2010.05.031] [PMID]
Baeza, I., De Castro, N. M., Giménez-Llort, L., & De la Fuente, M. (2010). Ovariectomy, a model of menopause in rodents, causes a premature aging of the nervous and immune systems. Journal of Neuroimmunology, 219(1-2), 90-9. [DOI:10.1016/j.jneuroim.2009.12.008] [PMID]
Bhavnani, B. R. (2003). Estrogens and menopause: Pharmacology of conjugated equine estrogens and their potential role in the prevention of neurodegenerative diseases such as Alzheimer’s. The Journal of Steroid Biochemistry and Molecular Biology, 85(2-5), 473-82. [DOI:10.1016/S0960-0760(03)00220-6]
Boumenir, A., Cognat, E., Sabia, S., Hourregue, C., Lilamand, M., & Dugravot, A., et al. (2019). CSF level of β-amyloid peptide predicts mortality in Alzheimer’s disease. Alzheimer’s Research & Therapy, 11(1), 29. [DOI:10.1186/s13195-019-0481-4] [PMID] [PMCID]
Calabrese, V., Cornelius, C., Mancuso, C., Barone, E., Calafato, S., & Bates, T., et al. (2009). Vitagenes, dietary antioxidants and neuroprotection in neurodegenerative diseases. Frontiers in Bioscience-Landmark, 14(1), 376-97. [DOI:10.2741/3250] [PMID]
Cheng, X. L., & Li, M. K. (2014). Effect of topiramate on apoptosis-related protein expression of hippocampus in model rats with Alzheimers disease. European Review for Medical and Pharmacological Sciences, 18(6), 761-8. [PMID]
Craig, L. A., Hong, N. S., & McDonald, R. J. (2011). Revisiting the cholinergic hypothesis in the development of Alzheimer’s disease. Neuroscience & Biobehavioral Reviews, 35(6), 1397-409. [DOI:10.1016/j.neubiorev.2011.03.001] [PMID]
Giffard, B., Desgranges, B., & Eustache, F. (2005). Semantic memory disorders in Alzheimer’s disease: Clues from semantic priming effects. Current Alzheimer Research, 2(4), 425-34. [DOI:10.2174/156720505774330582] [PMID]
Giordano, S., Darley-Usmar, V., & Zhang, J. (2014). Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biology, 2, 82-90. [DOI:10.1016/j.redox.2013.12.013] [PMID] [PMCID]
Guo, Sh., Xi, X., & Li, J. (2019). Notoginsenoside R1: A systematic review of its pharmacological properties. Die Pharmazie - An International Journal of Pharmaceutical Sciences, 74(11), 641-7. [DOI:10.1691/ph.2019.9534] [PMID]
Hatashita, Sh., & Wakebe, D. (2019). Amyloid β deposition and glucose metabolism on the long-term progression of preclinical Alzheimer’s disease. Future Science OA, 5(3), FSO356. [DOI:10.4155/fsoa-2018-0069] [PMID] [PMCID]
Henderson, V. W., Benke, K. S., Green, R. C., Cupples, L. A., Farrer, L. A., & MIRAGE Study Group. (2005). Postmenopausal hormone therapy and Alzheimer’s disease risk: Interaction with age. Journal of Neurology, Neurosurgery & Psychiatry, 76(1), 103-5. [DOI:10.1136/jnnp.2003.024927] [PMID] [PMCID]
Jameei, S. B., Nouyan-Ashraf, M. H., & Behzadi, G. (2004). Ovariectomy reduces the dendritic spine density of the dorsal raphe neurons in the adult rat. Archives of Iranian Medicine, 7(2), 122-7. https://www.sid.ir/en/journal/ViewPaper.aspx?ID=4729
Jameie, B., Mehdinejadiani, Sh., Farhadi, M., Solimani, M., Hassanzadeh, S., & Kerdari, M., et al. (2016). Effects of exogenous estrogen treatment on hippocampal neurogenesis of diabetic ovariectomized rats. International Clinical Neuroscience Journal, 3(2), 99-108. [DOI:10.22037/icnj.v3i2.13766]
Krishnan, S., Slavin, M. J., Tran, T. T. T., Murali Doraiswamy, P., & Petrella, J. R. (2006). Accuracy of spatial normalization of the hippocampus: Implications for fMRI research in memory disorders. NeuroImage, 31(2), 560-71. [DOI:10.1016/j.neuroimage.2005.12.061] [PMID]
Lee, J., Park, H. H., Koh, S. H., & Choi, H. (2017). Neural stem cell death mechanisms induced by amyloid beta. Dementia and Neurocognitive Disorders, 16(4), 121-7. [DOI:10.12779/dnd.2017.16.4.121] [PMID] [PMCID]
Losi, G., Puia, G., Garzon, G., de Vuono, M. C., & Baraldi, M. (2004). Apigenin modulates GABAergic and glutamatergic transmission in cultured cortical neurons. European Journal of Pharmacology, 502(1-2), 41-6. [DOI:10.1016/j.ejphar.2004.08.043] [PMID]
Nilsen, J., Chen, Sh., Irwin, R. W., Iwamoto, S., & Brinton, R. D. (2006). Estrogen protects neuronal cells from amyloid beta-induced apoptosis via regulation of mitochondrial proteins and function. BMC Neuroscience, 7, 74. [DOI:10.1186/1471-2202-7-74] [PMID] [PMCID]
Pike, Ch. J., Carroll, J. C., Rosario, E. R., & Barron, A. M. (2009). Protective actions of sex steroid hormones in Alzheimer’s disease. Frontiers in Neuroendocrinology, 30(2), 239-58. [DOI:10.1016/j.yfrne.2009.04.015] [PMID] [PMCID]
Prakash, A., & Kumar, A. (2014). Implicating the role of lycopene in restoration of mitochondrial enzymes and BDNF levels in β-amyloid induced Alzheimer׳s disease. European Journal of Pharmacology, 741, 104-11. [DOI:10.1016/j.ejphar.2014.07.036] [PMID]
Robusto-Leitao, O., & Ferreira, H. (2006). Hormones and dementia - a comparative study of hormonal impairment in post-menopausal women, with and without dementia. Neuropsychiatric Disease and Treatment, 2(2), 199-206. [DOI:10.2147/nedt.2006.2.2.199] [PMID] [PMCID]
Sandoval, K. E., & Witt, K. A. (2011). Age and 17β-estradiol effects on blood-brain barrier tight junction and estrogen receptor proteins in ovariectomized rats. Microvascular Research, 81(2), 198-205. [DOI:10.1016/j.mvr.2010.12.007] [PMID]
Vegeto, E., Benedusi, V., & Maggi, A. (2008). Estrogen anti-inflammatory activity in brain: A therapeutic opportunity for menopause and neurodegenerative diseases. Frontiers in Neuroendocrinology, 29(4), 507-19. [DOI:10.1016/j.yfrne.2008.04.001] [PMID] [PMCID]
Venigalla, M., Sonego, S., Gyengesi, E., & Münch, G. (2015). Curcumin and Apigenin - novel and promising therapeutics against chronic neuroinflammation in Alzheimer’s disease. Neural Regeneration Research, 10(8), 1181-5. [DOI:10.4103/1673-5374.162686] [PMID] [PMCID]
Vest, R. S., & Pike, Ch. J. (2013). Gender, sex steroid hormones, and Alzheimer’s disease. Hormones and Behavior, 63(2), 301-7. [DOI:10.1016/j.yhbeh.2012.04.006] [PMID] [PMCID]
Wang, F., Zhou, X. L., Yang, Q. G., Xu, W. H., Wang, F., & Chen, Y. P., et al. (2011). A peptide that binds specifically to the β-amyloid of Alzheimer’s disease: Selection and assessment of anti-β-amyloid neurotoxic effects. PLoS One, 6(11), e27649. [DOI:10.1371/journal.pone.0027649] [PMID] [PMCID]
Yamada, K., Tanaka, T., Zou, L. B., Senzaki, K., Yano, K., & Osada, T., et al. (1999). Long‐term deprivation of oestrogens by ovariectomy potentiates β‐amyloid‐induced working memory deficits in rats. British Journal of Pharmacology, 128(2), 419-27. [DOI:10.1038/sj.bjp.0702811] [PMID] [PMCID]
Zandi, P. P., Carlson, M. C., Plassman, B. L., Welsh-Bohmer, K. A., Mayer, L. S., & Steffens, D. C., et al. (2002). Hormone replacement therapy and incidence of Alzheimer disease in older women: The Cache County Study. JAMA, 288(17), 2123-9. [DOI:10.1001/jama.288.17.2123] [PMID]
Zhao, Y., Zheng, H. X., Xu, Y., & Lin, N. (2019). Estrogenic effect of the extract of QingYan formula on reproductive tissues in immature mice. Evidence-Based Complementary and Alternative Medicine, 2019, 5493714. [DOI:10.1155/2019/5493714] [PMID] [PMCID]
Zhao, L., Woody, S. K., & Chhibber, A. (2015). Estrogen receptor β in Alzheimer’s disease: From mechanisms to therapeutics. Ageing Research Reviews, 24(Pt B), 178-90. [DOI:10.1016/j.arr.2015.08.001] [PMID] [PMCID]
Zhao, L., Wang, J. L., Wang, Y. R., & Fa, X. Z. (2013). Apigenin attenuates copper-mediated β-amyloid neurotoxicity through antioxidation, mitochondrion protection and MAPK signal inactivation in an AD cell model. Brain Research, 1492, 33-45. [DOI:10.1016/j.brainres.2012.11.019] [PMID]
Type of Study: Original |
Subject:
Behavioral Neuroscience
Received: 2020/06/12 | Accepted: 2020/08/9 | Published: 2021/05/1
Received: 2020/06/12 | Accepted: 2020/08/9 | Published: 2021/05/1
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





